Abstract
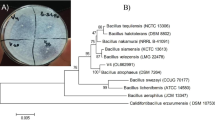
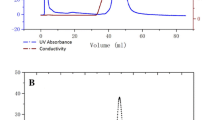
In this study, Mirabilis jalapa tuber powder (MJTP) was used as a new complex organic substrate for the growth and production of fibrinolytic enzymes by a newly isolated Bacillus amyloliquefaciens An6. Maximum protease activity (1,057 U/ml) with casein as a substrate was obtained when the strain was grown in medium containing (grams per liter) MJTP 30, yeast extract 6, CaCl2 1, K2HPO4 0.1, and K2HPO4 0.1. The strain was also found to grow and produce extracellular proteases in a medium containing only MJTP, indicating that it can obtain its carbon, nitrogen, and salts requirements directly from MJTP. The B. amyloliquefaciens An6 fibrinase (BAF1) was partially purified, and fibrinolytic activity was assayed in a test tube with an artificial fibrin clot. The molecular weight of the partially purified BAF1 fibrinolytic protease was estimated to be 30 kDa by sodium dodecyl sulfate polyacrylamide gel electrophoresis and gel filtration. The optimum temperature and pH for the caseinolytic activity were 60 °C and 9.0, respectively. The enzyme was highly stable from pH 6.0 to 11.0 and retained 62% of its initial activity after 1 h incubation at 50 °C. However, the enzyme was inactivated at higher temperatures. The activity of the enzyme was totally lost in the presence of phenylmethylsulfonyl fluoride, suggesting that BAF1 is a serine protease.





Similar content being viewed by others
References
Kim, W., Choi, K., Kim, Y., Park, H., Choi, J., Lee, Y., et al. (1996). Applied and Environmental Microbiology, 62, 2482–2488.
Kim, S. H., & Choi, N. S. (2000). Bioscience, Biotechnology, and Biochemistry, 64, 1722–1725.
Mine, Y., Wong, A. H. K., & Jiang, B. (2005). Food Research International, 38, 243–250.
Sherry, S. (1987). American Journal of Cardiology, 59, 984–989.
Sumi, H., Hamada, H., Tsushima, H., Mihara, H., & Muraki, H. (1987). Experientia, 43, 1110–1111.
Mihara, H., Sumi, H., Yoneta, T., Mizumoto, H., Ikeda, R., Seiki, M., et al. (1991). Japanese Journal of Physiology, 41, 461–472.
Nakamura, T., Yamagata, Y., & Ichishima, E. (1992). Bioscience, Biotechnology, and Biochemistry, 56, 1869–1871.
Fujita, M., Nomura, K., Hong, K., Ito, Y., Asada, A., & Nishimuro, S. (1993). Biochemical and Biophysical Research Communications, 197, 1340–1347.
Sumi, H., Hamada, H., Nakanishi, K., & Hiratani, H. (1990). Acta Haematologica, 84, 139–143.
Peng, Y., Huang, Q., Zhang, R. H., & Zhang, Y. Z. (2003). Comparative Biochemistry and Physiology Part B, 134, 45–52.
Johnvesly, B., & Naik, G. R. (2001). Process Biochemistry, 37, 139–144.
Puri, S., Beg, Q. K., & Gupta, R. (2002). Current Microbiology, 44, 286–290.
Frankena, J., Koningstein, G. M., Van Verseveld, H. W., & Stouthamer, A. H. (1986). Applied Microbiology and Biotechnology, 24, 106–112.
Giesecke, U. E., Bierbaum, G., Rudde, H., Spohn, U., & Wandrey, C. (1991). Applied Microbiology and Biotechnology, 35, 720–724.
Drucker, H. (1972). Journal of Bacteriology, 110, 1041–1049.
Ferrero, M. A., Castro, G. R., Abate, C. M., Baigori, M. D., & Sineriz, F. (1996). Applied Microbiology and Biotechnology, 45, 327–332.
Gessesse, A., & Gashe, B. A. (1997). Biotechnological Letters, 19, 479–481.
Mehrotra, S., Pandey, P. K., Gaur, R., & Darmwal, N. S. (1999). Bioresource Technology, 67, 201–203.
Ghorbel-Frikha, B., Sellami-Kamoun, A., Fakhfakh, N., Haddar, A., Manni, L., & Nasri, M. (2005). Journal of Industrial Microbiology & Biotechnology, 32, 186–194.
Ellouz, Y., Bayoudh, A., Kammoun, S., Gharsallah, N., & Nasri, M. (2001). Bioresource Technology, 80, 49–51.
Joo, H. S., & Chang, C. S. (2005). Process Biochemistry, 40, 1263–1270.
Naidu, K. S. B., & Devi, K. L. (2005). African Journal of Biotechnology, 4, 724–726.
Tari, C., Genckal, H., & Tokatli, F. (2006). Process Biochemistry, 41, 659–665.
Vivanco, J. M., Querci, M., & Salazar, L. F. (1999). Plant Disease, 83, 1116–1121.
Yang, S. W., Ubillas, R., McAlpine, J., Stafford, A., Ecker, D. M., Talbot, M. K., et al. (2001). Journal of Natural Products, 64, 313–317.
DeBolle, M., Eggermont, K., Duncan, R., Osborn, R., Terras, F., & Broekaert, W. (1995). Plant Molecular Biology, 28, 713–721.
Wang, Y., Chen, J., Yang, Y., Zheng, Y., Tang, S., & Luo, S. (2002). Helvetica Chimica Acta, 85, 2342–2348.
Miller, J. H. (1972). Experiments in molecular genetics (pp. 431–435). Cold Spring Harbor: Cold Spring Harbor Laboratory.
Hajji, M., Rebai, A., Gharsallah, N., & Nasri, M. (2008). Applied Microbiology and Biotechnology, 79, 915–923.
Jellouli, K., Bayoudh, A., Manni, L., Agrebi, R., & Nasri, M. (2008). Applied Microbiology and Biotechnology, 79, 989–999.
Kembhavi, A. A., Kulharni, A., & Pant, A. A. (1993). Applied Biochemistry and Biotechnology, 38, 83–92.
Bradford, M. M. (1976). Analytical Biochemistry, 72, 248–254.
Laemmli, U. K. (1972). Nature, 227, 680–685.
Garcia-Carreno, F. L., Dimes, L. F., & Haard, N. F. (1993). Analytical Biochemistry, 214, 65–69.
North, M. J. (1982). Experimental Mycology, 6, 345–352.
Hinman, R. L. (1994). Chemtech, 24, 45–48.
Ko, J. H., Yan, J. P., Zhu, L., & Qi, Y. P. (2004). Comparative Biochemistry and Physiology. Part C. Toxicology & Pharmacology, 137, 65–74.
Choi, N. S., & Kim, S. H. (2001). Journal of Biochemistry and Molecular Biology, 34, 134–138.
Kho, C. W., Park, S. G., Cho, S., Lee, D. H., Myung, P. K., & Park, B. C. (2005). Protein Expression and Purification, 39, 1–7.
Chang, C. T., Fan, M. H., Kuo, F. C., & Sung, H. Y. (2000). Journal of Agricultural and Food Chemistry, 48, 3210–3216.
Lee, S. K., Bae, D. H., Kwon, T. J., Lee, S. B., Lee, H. H., Park, J. H., et al. (2001). Journal of Microbiology and Biotechnology, 11, 845–852.
Jeong, Y. K., Park, J. U., Baek, H., Park, S. H., & Kong, I. S. (2001). World Journal of Microbiology & Biotechnology, 17, 89–92.
Wang, C., Ji, B., Li, B., & Ji, H. (2006). World Journal of Microbiology & Biotechnology, 22, 1365–1371.
Agrebi, R., Haddar, A., Hmidet, N., Jellouli, K., Manni, L., & Nasri, M. (2009). Process Biochemistry, 44, 1252–1259.
Wang, S. H., Cheng, Z., Yang, Y. L., Miao, D., & Bai, M. F. (2008). World Journal of Microbiology & Biotechnology, 24, 475–482.
Liang, X., Jia, S., Sun, Y., Chen, M., Chen, X., Zhong, J., et al. (2007). Molecular Biotechnology, 37, 187–194.
Acknowledgment
This work was funded by “Ministry of Higher Education, Scientific Research and Technology—Tunisia”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Agrebi, R., Hmidet, N., Hajji, M. et al. Fibrinolytic Serine Protease Isolation from Bacillus amyloliquefaciens An6 Grown on Mirabilis jalapa Tuber Powders. Appl Biochem Biotechnol 162, 75–88 (2010). https://doi.org/10.1007/s12010-009-8800-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-009-8800-z