Abstract
The presence of chlorophylls in phenolic extracts may limit their use in food products due to undesirable coloration. This project seeks to minimize the co-extraction of chlorophylls during the ultrasonic-assisted extraction (UAE) of polyphenols from sugar beet leaves (SBLs) by optimizing the extraction condition using response surface methodology (RSM). Optimization aimed to minimize the chlorophyll yield (CY) of the extraction while maximizing the total phenolic content (TPC) and total flavonoid content (TFC) with the lowest possible amount of sample. Optimized extraction parameters were 25% ethanol as solvent, 8 min extraction time, and 3.98% (w/v) solid:liquid ratio. The experimental values at optimized condition were 11.49 ± 0.66 mmol gallic acid equivalent (GAE)/L TPC, 2.09 ± 0.06 mmol quercetin equivalent/L TFC, and 0.05 ± 0.01 mg/g CY. In the optimized extract, the ferric reducing antioxidant power (FRAP) was 3.16 ± 0.25 mmol trolox equivalent/L, and trans-ferulic acid had the highest concentration (123.39 ± 4.13 µmol/L) among the detected phenolic compounds. The optimized extract, at a phenolic concentration of 300 µmol GAE/L, chelated cupric and ferrous ions by 48.95 ± 1.06%, and 66.18 ± 1.31%, respectively. Adding the optimized extract to vegetable oils increased their oxidative stability significantly (p < 0.05). The antioxidant activity index of the optimized extract in all the cases was comparable to butylated hydroxytoluene (BHT), confirming that the obtained extract could be a reliable substitute for synthetic antioxidants. The results of this study are important in enhancing the utilization of extracts recovered from food by-products in the food industry.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Oxidative deterioration is a major issue associated with edible oils rich in polyunsaturated fatty acids, which causes a sequence of unfavorable changes in the sensorial properties of oil and the formation of toxic compounds causing health problems (Bayram & Decker, 2023a). As an efficient and cheap solution to this problem, synthetic antioxidants, such as butylated hydroxytoluene (BHT), are commonly used in the food industry (S. Sharma et al., 2023). However, the growing demand for clean-label food and awareness of consumers regarding the health concerns of synthetic antioxidants have prompted the food industry to explore alternative methods for inhibiting lipid oxidation (Culler et al., 2022). One potential solution to address this issue is the utilization of natural antioxidants (Ebrahimi & Lante, 2021, 2022). However, despite the identification and characterization of numerous natural antioxidants, only a small fraction is actually sold and employed as antioxidants on an industrial level. This underscores the immediate need to enhance the efficacy of authorized natural antioxidants (Villeneuve et al., 2023).
Numerous studies indicated that food by-products could serve as a valuable and cost-effective source of natural antioxidants with applications in the food industry. Furthermore, employing food by-products as a source of bioactive compounds presents a promising opportunity to implement a circular economic approach and mitigate the escalating production of organic waste and its associated environmental issues (Ferrando et al., 2023; Palos-Hernández et al., 2022). The leaves of the sugar beet plant (Beta Vulgaris L.) are often overlooked and discarded as by-products or utilized in animal feed (Akyüz & Ersus, 2021; Nutter et al., 2020), while they are a rich source for the recovery of bioactive compounds such as polyphenols (Ebrahimi et al., 2022).
The recovery of polyphenols requires efficient and affordable extraction strategies with environmentally friendly properties (Kumar et al. 2021). Ultrasound-assisted extraction (UAE) as an environmentally friendly technology has been proven to be a potential alternative for the recovery of polyphenols from food by-products compared to conventional extraction methods as this method gives maximum extraction yield in a shorter time with minimal solvent (Ebrahimi & Lante, 2022; Kumar et al. 2021). Nevertheless, the use of organic solvents, like ethanol, in the extraction process from green plants leads to the simultaneous extraction of chlorophylls, which can potentially affect the accuracy of analytical measurement and complicate the use of the extract in food product development by altering sensory characteristics (Ebrahimi et al., 2023; Ebrahimi, Hoxha, et al., 2024b; Skendi et al., 2022). Moreover, the inclusion of chlorophyll-containing extracts in food products can induce lipid photo-oxidation upon exposure to light (Tzima et al., 2020). One solution to solve this issue could be using water as an extracting solvent. However, not all phenolic compounds are soluble in water, emphasizing the importance of determining the optimal extraction condition using an alcoholic solvent that can effectively solubilize fewer chlorophylls.
To obtain the optimal extraction condition, the input factors (i.e., extraction parameters), including temperature, incubation time, particle size, pressure, concentration and type of extracting solvent, and solid:liquid ratio, must be optimized. Response surface methodology (RSM) is a crucial tool for this purpose, as it allows for statistical and mathematical model building to optimize the level of these independent variables (Kumar et al. 2021; Mehmood et al., 2018). Although RSM has been widely used in the literature to enhance the recovery of phenolic compounds from plant leaves, the co-extraction of chlorophylls has often been overlooked, with most studies primarily focusing on maximizing phenolic yield (Bengardino et al., 2019; Cassiana Frohlich et al., 2022; Derrien et al., 2017; González-Silva et al., 2022; Miao et al., 2022). This study uniquely addresses this gap by not only optimizing the extraction of phenolic compounds but also actively minimizing chlorophyll content, thus enhancing the extract's suitability for use in food products, particularly vegetable oils.
Therefore, the primary objective of this study was to optimize the extraction of phenolic compounds from sugar beet leaves (SBLs) to obtain the highest phenolic content from the least possible amount of sample while minimizing chlorophyll content. To accomplish this, total phenolic content (TPC), total flavonoid content (TFC), and chlorophyll yield (CY) were measured as key optimization responses. Additionally, the study aimed to enhance the oxidative stability of various vegetable oils by selecting the optimal concentration of the SBL extract based on metal chelating activity tests.
Materials and methods
Chemicals and materials
The leaves were of a sugar beet plant (Beta vulgaris L., var. SMART DJERBA-KWS) granted by a local farm in Padova, Italy. All the vegetable oils were purchased from local markets in Padova, Italy. Sodium hydroxide, hydrochloric acid, ethanol, iron(III) chloride, 2,4,6-tri(2- pyridyl)-s-triazine (TPTZ), aluminum nitrate, potassium acetate, BHT, sodium carbonate, Folin–Ciocalteu reagent, gallic acid, trolox, 4-dihydroxybenzoic acid, caffeic acid, trans-ferulic acid, imidazole, sodium acetate anhydrous, pyrocatechol violet, copper(II) sulfate, iron(II) chloride, and 3-(2-pyridyl)-5,6-diphenyl-1,2,4-triazine-4′,4″-disulfonic acid sodium salt (ferrozine) were obtained from Sigma-Aldrich (St. Louis, MO, USA). All chemicals and solvents utilized in this study were of analytical grade.
Sample preparation
The SBLs were transported to the laboratory in an insulated box. They were rinsed with distilled water to remove the soil, and the stems were separated manually. The leaves were freeze-dried at − 50 ℃ using a freeze dryer (Modulyo, Edwards, West Sussex, UK) and then ground using a mortar and pestle. The powdered leaves were vacuum-packed and stored at − 80 ℃ until the following analyses.
Extraction of polyphenols
The extraction of polyphenols from SBLs was performed using a UAE method, according to Ebrahimi, Mihaylova, et al. (2022), with minor modifications. The extract was obtained using an ultrasonic homogenizer (HD 2200.2, Bandelin, Berlin, Germany) operating at 200 W output power, and 20 kHz frequency, equipped with a 6 mm titanium probe (KE 76, Bandelin, Berlin, Germany). The extraction was done by immersing the probe for a length of 2 mm in the mixture of freeze-dried SBLs and 10 mL of extracting solvent. The initial temperature of the extraction mixtures was 25 ℃, and to prevent high temperatures during ultrasonication, the extraction cell was placed in an ice bucket. The extraction parameters to be optimized were solid:liquid ratio (1–5% w/v), ethanol concentration (0–100%), amplitude (20–40%), and extraction time (2–10 min). The apparatus was set to 60-s ultrasonication cycles with 10-s pauses in the middle to avoid overheating in the probe. The mixture obtained from ultrasonication was centrifuged at 4 ℃ and 4000 × g for 10 min. The supernatant was filtered through Whatman No. 1 filter papers (GE Healthcare, Chalfont St. Giles, UK) and 0.45-µm cellulose acetate syringe filters (Test Scientific, Perugia, Italy) and stored at − 80 ℃ until the following analyses.
Optimization of extraction
To optimize the extraction condition, the effect of solid:liquid ratio, ethanol concentration, amplitude, and extraction time on TPC, TFC, and CY of extracts was analyzed through RSM to fit the second-order polynomial equation described as follows (Eq. 1) (Derrien et al., 2018):
where Y is response variables (i.e., TPC (Y1), TFC (Y2), and CY (Y3) of extracts), β0 is the constant coefficient, βi is the linear coefficient, βij is the two-factor interaction coefficient, βii is the quadratic coefficients, and Xi and Xj are the independent variables, including ethanol concentration (X1), extraction time (X2), amplitude (X3) and solid:liquid ratio (X4). The experiment was designed using central composite design (CCD). The type and range of independent variables were selected based on literature (Table S1, Supplementary material) and preliminary experimental findings (Data not shown). In accordance with CCD, the independent variables were set at five levels (-α, -1, 0, + 1, + α). Table 1 presents the RSM design, including coded and actual levels. Thirty treatments, comprising eight axial points, sixteen fractional factorial points, and six central points, were randomly executed based on CCD. The optimization criteria for the output factors were to minimize chlorophyll content while maximizing TPC and TFC. For the input factors, the criteria focused on minimizing the sample quantity used in the extraction process to align with sustainability principles and improve the eco-efficiency of the extraction.
Total phenolic content (TPC)
The TPC test was conducted based on the Folin–Ciocalteu colorimetric method as reported by Ebrahimi, Mihaylova, et al. (2022). The test mixture consisted of 0.5 mL of the extract, 0.25 mL of a two-time diluted Folin–Ciocalteu reagent, and 1.25 mL of a 7.5% (w/v) sodium carbonate solution containing 1 M sodium hydroxide. In the blank sample, 0.5 mL of the extracting solvent was used instead of the extract. After incubation for 30 min at room temperature in the dark, the absorbance was recorded at 650 nm using a Varian Carry 50 Bio UV/Vis spectrophotometer (Agilent Technologies, Santa Clara, CA, USA). A standard curve of gallic acid (R2 = 0.996) was used to calculate the results in mmol gallic acid equivalent (GAE)/L extract.
Total flavonoid Content (TFC)
The TFC test was conducted according to the method described by Mihaylova et al. (2021) with some modifications. The test mixture consisted of 0.22 mL of the extract, 45 µL of a 10% (w/v) aluminum nitrate solution, 45 µL of a 1 M potassium acetate solution, and 1.7 mL of a 70% ethanol. In the blank sample, 0.22 mL of extracting solvent was used instead of the extract. After incubation at room temperature for 40 min, the absorbance was measured at 415 nm. A standard curve of quercetin (R2 = 0.999) was used to calculate the results in mmol quercetin equivalent (QE)/L extract.
Determination of chlorophyll and carotenoid content
The chlorophyll content of the extracts was monitored spectrophotometrically, according to Lichtenthaler and Buschmann, (2001). First, the extracts were diluted using 95% ethanol, depending on their greenness, to keep the absorbance values between 0.2 and 0.8. Next, they were filtered through 0.22 µm cellulose acetate syringe filters (Test Scientific, Perugia, Italy) to eliminate any possible turbidity. Absorbance measurements were done at 664.1, 648.6, 470 and 750 nm. As chlorophyll a and b exhibit no absorbance at 750 nm, absorbance values at 664.1 and 648.6 nm were adjusted by subtracting them from the absorbance at 750 nm. Equations 2, 3, and 4 were used to calculate the concentration of chlorophyll a, b, and carotenoids, respectively. Considering the weight of freeze-dried SBLs and the volume of the solvent in the extraction process, the CY in mg/g was calculated based on the sum of chlorophyll a and b to be utilized in the optimization process.
where Chla is chlorophyll a, Chlb is chlrophyll b, Ct is carotenoid content, and A is absorbance.
Ferric reducing antioxidant power (FRAP)
The FRAP assay was carried out based on the method described by Ebrahimi, Mihaylova, et al. (2022). The FRAP reagent was prepared by combining sodium acetate buffer (0.04 M, pH 3.6), 20 mM iron(III) chloride solution, and 0.01 M TPTZ in 0.04 M hydrochloric acid in a ratio of 10:1:1. The test mixture consisted of 0.9 mL of the prepared FRAP reagent, and 0.1 mL of the extract. In the blank sample, the extracting solvent was added instead of the extract. After incubation for 30 min at 37 ℃, the absorbance was recorded at 593 nm. A standard curve of trolox (R2 = 0.999) was used to calculate the results in mmol Trolox equivalent (TE)/L extract.
Metal chelating activity
The cupric and ferrous ion chelating activity of the optimized extract were determined using different concentrations based on the TPC. This involved freeze-drying the optimized extract of SBLs and then reconstituting it in 75% ethanol, yielding a concentrated extract for the analysis. Subsequently, diverse dilutions were made using sodium acetate-imidazole buffer (10 mM, pH 7.0), resulting in final concentrations ranging from 50 to 300 µmol GAE/L. The cupric (Cu2+) chelating activity was determined according to Liu et al. (2015) with some modifications. The test mixture consisted of 50 µL of a 5 mM copper (II) sulfate solution, and 2.5 mL of each concentration of extract. In the blank sample, the buffer was used instead of extracts. After incubation for 30 min at room temperature, 50 µL of 4 mM pyrocatechol violet was added, and absorbance was recorded at 632 nm. The percentage of inhibition of Cu2+-pyrocatechol violet complex formation was calculated according to Eq. 5.
The ferrous (Fe2⁺) chelating activity was assayed according to Bayram et al. (2023) with some modifications. The test mixture consisted of 10 µL of 10 mM ferrous chloride solution, 50 μL of 0.05 M ferrozine solution, and 2.5 mL of each concentration of extract. In the blank sample, the buffer was used instead of extracts. After incubation in the dark at room temperature for 10 min, the absorbance was recorded at 562 nm. The percentage of inhibition of Fe2+-ferrozine complex formation was calculated according to Eq. 5.
where A0 is the absorbance of the blank, and A1 is the absorbance of the extracts.
HPLC–PDA analysis of phenolic compounds
The individual phenolic compounds of the optimized extract were detected according to Ebrahimi, Bayram, et al. (2024). An HPLC apparatus (LC-2030C 3D, Shimadzu, Tokyo, Japan) equipped with a UV–Vis photodiode array (PDA) detector was used to separate and quantify the phenolic compounds using an Ascentis Express 90 Å C18 reversed-phase column (5 μm i.d., 250 × 4.6 mm; Supelco, Bellefonte, PA, USA). The column and guard column were maintained at a constant temperature of 40 ℃. The extract and standards were eluted using a mobile phase composed of ultrapure water acidified with 0.1% acetic acid (designated as solvent A) and acetonitrile (designated as solvent B) at a flow rate of 1 mL/min. The solvent gradient, expressed in volumetric ratios of solvents A and B, was as follows: 10–30% B for 30 min and 30–10% B for 10 min. After each analysis, the column was conditioned by running the HPLC system for 15 min with the mobile phase composition set at 10% B. The injection volume was 20 μL, and the wavelength spectrum was recorded from 200 to 400 nm to differentiate phenolic compounds based on their maximum absorbance wavelength. The detection wavelength used for monitoring phenolic compounds was 280 nm. Standard stock solutions (5 mM) of gallic acid, 3,4-dihydroxybenzoic acid, caffeic acid, and trans-ferulic acid were prepared in 75% ethanol. Calibration curves for each compound were established at concentrations ranging from 2 to 100 μM to determine the final concentration of detected phenolic compounds.
Fortification of different oils using the optimized extract
Different vegetable oils, including soybean oil, corn oil, sunflower oil, rapeseed oil, and peanut oil were enriched with the optimized extract of SBLs to assess its ability to increase the oxidative stability of the oils, compared to BHT. Briefly, the optimized extract was freeze-dried and resuspended in 75% ethanol to obtain a 10-times concentrated extract for the analysis. The TPC of the oils was then set to 300 µmol GAE/L using the concentrated optimized extract. This TPC was selected based on the maximum metal-chelating activity of the extract to prevent the prooxidative action of free metal ions. As a benchmark, BHT was utilized as a synthetic antioxidant at a concentration of 200 ppm (≈900 µmol/L), aligning with the maximum permissible level established by Codex Alimentarius (2023). The oil-antioxidant mixture underwent homogenization at 1000 × g while placed in an ice bucket, using a homogenizer (T 25 digital ULTRA-TURRAX®, IKA- Werke GmbH and Co. KG, Staufen, Germany) for 5 min.
Oxidative stability assay using Rancimat method
The Rancimat method, as described by Cisneros-Yupanqui et al. (2021), was used to assess the oxidative stability of the different vegetable oils after adding BHT or the optimized extract of SBLs. Firstly, 3 g of control/antioxidant added oil was loaded into the Rancimat instrument (Metrohm, model 743, Herisau, Switzerland) for evaluating the oxidative stability over time based on the formation of volatile acids produced by the free radical chain reaction. The temperature and airflow were set at 120 ℃ and 20 L/h, respectively. The oxidative stability is reported as the induction period (IP), which refers to the time (h) at which the water conductivity (µS/min) starts to increase as a result of the production of volatile compounds involved in the lipid oxidation. The antioxidant activity index (AAI) was calculated using Eq. 6.
Statistical analysis
All the analyses were carried out in triplicate. Design expert software (version. 13.0.1.0) was used to prepare the RSM design and perform analysis of variance (ANOVA) for studying model fit, coefficients of determinations, factors, and interactions with a significant effect on responses. Based on the model selection criteria (Sect. 3.1), a quadratic model was initially employed to process the data. After specifying the significant terms according to ANOVA, the quadratic model was simplified to fit the predicting equations. The data obtained in the Rancimat test and metal chelating activity assays were subjected to ANOVA using IBM SPSS Statistics (Version 20.0, SPSS Inc, Chicago, IL, USA), and the comparisons were made by Tukey’s test with a significance level and confidence level of 0.05 and 95%, respectively. The data on the antioxidant activity index was analyzed using a t test. Origin Pro 2022 (Northampton, MA, USA) was used for graphing the data.
Results and discussions
Optimization of extraction
In the present paper, the effect of independent variables (i.e., extraction parameters) on TPC (Y1), TFC (Y2), and CY (Y3) was evaluated to find the most optimum condition for the extraction of polyphenols using RSM. The results of all the extraction conditions are detailed in Table 1.
Table 2 presents the summary of the analysis of variance (ANOVA) and fit statistics. The quadratic polynomial model demonstrated high F-values and significant p-values (p < 0.001) for all independent variables, indicating a highly significant model, as supported by Mehmood et al. (2018). Moreover, compared to the other fitted models (i.e., linear, two-factor interaction, cubic), the quadratic model had a higher R2, adjusted R2, and predicted R2 values for the dataset in the present study. The coefficient of determination (R2) for all responses was greater than 0.90, indicating that a high percentage of the variation was explained by the model. In the quadratic model, the values for adequate precision were well above 4, indicating an adequate signal. The non-significant lack of fit F-values (p > 0.05) suggests that the model fits the data well. Moreover, the low coefficient of variation values indicate good precision and reliability of the model. Additionally, the PRESS value for all responses in the quadratic model was lower than in other models, demonstrating its superior predictive performance. These indicators collectively demonstrate that the quadratic model is appropriate for explaining the variability in TPC, TFC, and CY with high accuracy, precision, and predictive capability.
Equations 7, 8, and 9 represent the initial predicting equations based on the selected quadratic model, before removing the insignificant terms. To stabilize variance and improve the normality of residuals, an inverse square root transformation was applied to TFC, and a square root transformation was applied to CY.
Among the input factors, solid:liquid ratio and ethanol concentration significantly affected all dependent variables (p < 0.0001). The extraction time had a significant effect only on TFC (p < 0.05). However, based on the ANOVA results (Table 2), the effect of amplitude in the model for all the responses was insignificant (p > 0.05). This might happen due to the combined effect of multiple variables interacting with each other, which might overshadow the individual effects. In the literature, there are some examples where the effect of amplitude or time was not individually significant in the extraction (Derrien et al., 2017; Papoutsis et al., 2018; Zhang et al., 2024).
To improve the significance of the model, a simplification was conducted by removing the insignificant terms, which led to the re-estimation of coefficients in the final fitted model (Rebollo-Hernanz et al., 2021). The individual and interaction effects of amplitude were excluded from all models, while the effects of time were removed from all models except for TFC. This approach preserved the hierarchy of the models. Equations 10, 11, and 12 represent the final equations used as prediction models. Therefore, the final responses were predicted by considering only the significant terms in the simplified models, resulting in an accurate, and precise prediction. These observations were further supported by the actual vs. predicted plot (Fig. 1), where the points are close to the fitted line.
Figure 2 indicates the response surface plots showing the effect of significant independent variables on TPC (a), CY (b), and TFC (c, d, e). The results show that the extraction condition, especially the solid:liquid ratio and the concentration of ethanol is prominent to achieve the optimum results. Decreasing the ethanol concentration significantly reduced CY. Marcheafave et al. (2019) reported that the high concentration of ethanol can increase the content of chlorophyll b in the extraction. This is because chlorophylls are not soluble in water, but they are soluble in organic solvents. The chlorophyll molecules constitute a hydrophilic (porphyrin) head group and a lipophilic hydrocarbon tail (phytol group). Due to their lipophilic hydrocarbon chains as a phytol tail, they are generally considered insoluble in water (M. Sharma et al., 2021). Therefore, it is preferred to have a lower concentration of ethanol for the extraction of polyphenols to yield a minimum chlorophyll content. This clearly states that using pure water for the extraction of polyphenols does not co-extract chlorophylls. However, since some polyphenols are not soluble in water, it is preferred to find a low concentration of ethanol that extracts a low concentration of chlorophylls, while having an acceptable recovery of phenolic compounds.
Response surface plots showing total phenolic compound (TPC) affected by the interaction of solid:liquid ratio vs. ethanol concentration (a), chlorophyll yield (CY) affected by the interaction of solid:liquid ratio vs. ethanol concentration (b), and total flavonoid content (TFC) affected by the interaction of solid:liquid ratio vs. ethanol concentration (c), time vs. ethanol concentration (d), and solid:liquid ratio vs. time (e)
SBLs are rich in polyphenols owning a high antioxidant activity (Ebrahimi et al., 2022). The results of the present paper show that SBLs have high TPC ranging from 2.05 ± 0.10 to 12.12 ± 0.09 mmol GAE/L, and high TFC ranging from 0.45 ± 0.05 to 3.09 ± 0.02 mmol QE/L, depending on the extraction parameters (Table 1). However, lower TPC values have been reported in the literature for SBLs, which could be due to variations in the extraction processes, plant age and variety, storage conditions and duration, and cultivation methods (Dukić et al., 2022; Ebrahimi, Bayram, et al. 2024; Maravić et al., 2022).
The TPC and TFC values of the extracts were significantly affected by solid:liquid ratio and the concentration of the extracting solvent (e.g., ethanol). Wen et al. (2019) reported that the combination of organic solvents and water increases the yield of extraction of polyphenols significantly. Ethanol is a green extraction solvent for obtaining bioactive compounds, which presents many advantages as it can be renewably produced from biotechnological processes and is non-toxic, with low flammability and environmental footprint (Cassiana Frohlich et al., 2022). The addition of water to pure ethanol provides a better distribution of polyphenols and enhances the yield of this process (Ninčević Grassino et al., 2020). Differences in the structure of phenolic compounds also determine their solubility in solvents with different polarity. Therefore, the type of solvent as well as the extraction method may have a significant impact on the yield of recovery of polyphenols from plant materials (Złotek et al., 2016).
Furthermore, solid:liquid ratio is another crucial factor in the extraction process of polyphenols. Papoutsis et al. (2018) reported that solid:liquid ratio significantly affects the recovery of phenolic compounds. Shahidi, (2022) reported that the TPC of samples increases with the increase in solid:liquid ratio. According to Fig. 2 (a, c, e), the maximum solid:liquid ratio resulted in a higher TPC and TFC in the extract. However, the optimization criteria focused on minimizing the amount of sample required to adhere to sustainability principles and enhance the eco-efficiency of the extraction process. Moreover, the high quantities of SBLs, due to the low density and high volume of the leaves, posed technical challenges such as increased viscosity of the extraction mixture and potential clogging of the equipment. These issues can impede ultrasound cavitation and hinder the dispersion of extracting solvent in the sample (Linares & Rojas, 2022). Another reason for minimizing the solid:liquid ratio was to avoid the co-extraction of other bioactive compounds. Kaur et al. (2021) reported that solid:liquid ratio may increase the extraction of other biomolecules (e.g., proteins and polysaccharides), which could dissolve in the solvent and influence the dissolution of polyphenols. The results of the present paper showed that a lower solid:liquid ratio could result in a lower chlorophyll content in the extracts. Therefore, minimizing this parameter was crucial to ensure efficient extraction while overcoming technical challenges and limiting unwanted co-extractions.
According to the research aim, the criteria for the optimization of extraction parameters was to minimize the CY and maximize TPC, and TFC, while keeping the solid:liquid ratio minimum. Based on the desirability function approach, the optimum UAE extraction condition was 25% ethanol as solvent, 3.97% (w/v) solid:liquid ratio, 8 min extraction time. The amplitude was excluded from the predictive models, and for the extraction under optimal conditions, it was fixed at 25%. The desirability level of prediction was 0.918 on a scale of 0 to 1. To validate the optimization, the extraction was repeated under the optimal conditions, and the actual experimental results were compared to the predicted values for TPC, TFC, and CY (Table 3). The comparison showed no significant differences (p < 0.05) between the predicted and actual values, confirming the successful optimization of the extraction parameters.
Characteristics of the optimized extract
Table 4 shows the phenolic compounds, chlorophyll a and b, and carotenoid content of the optimized extract of SBLs. The phenolic profile of the optimized extract was evaluated using an HPLC method, and the selected phenolic compounds were chosen according to the most abundant phenolic compounds in SBLs based on the literature (Battistella Lasta et al., 2019; Ebrahimi, Hoxha, et al., 2024a, 2024b; Maravić et al., 2022; Vissers et al., 2017). Phenolic acids constitute a significant portion of the phenolic profile in plants from the beta vulgaris L. species (Vissers et al., 2017). The highest concentration among the characterized phenolic compounds was trans-ferulic acid, which is consistent with the results reported in the literature (Battistella Lasta et al., 2019; Maravić et al., 2022). Ferulic acid, derived from the metabolism of phenylalanine and tyrosine, stands out as a highly abundant phenolic compound occurring in plants (Ebrahimi et al., 2020), with the trans isomer being particularly prevalent in many plant extracts. Ferulic acid is also known to have high ferrous chelation activity due to the prevention of reactive hydroxyl radical (HO●) formation via Fenton or similar reactions (Truong et al., 2020). Thus, the high concentration of ferulic acid in the optimized extract of SBLs may have a positive effect on the efficiency of the extract in delaying lipid oxidation.
Furthermore, the results indicate that while the concentration of chlorophyll a and b in the optimized extract is negligible, it has a high polyphenolic content (i.e., TPC, and TFC) and antioxidant activity (i.e., 3.16 ± 0.25 mmol TE/L FRAP) compared to the previous literature on the extraction of polyphenols from SBLs (Dukić et al., 2022; Ebrahimi et al., 2022; El-Gengaihi et al., 2016; Maravić et al., 2022). This shows that the optimization process for minimizing the chlorophyll content and maximizing the phenolic content was successful and yielded an extract with robust antioxidant activity.
Metal chelating activity of extracts
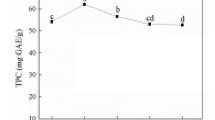
Figure 3 demonstrates the metal chelating activity of optimized extract at different concentrations. With increasing the concentration of extract, the chelation of cupric and ferrous ions both increased significantly (p < 0.05) due to the presence of more phenolic compounds. This shows that a TPC of 300 µmol GAE/L is the best concentration of optimized extract that could be used to inhibit lipid oxidation in the oil systems. The high content of ferulic acid in the optimized extract justifies its high metal chelating activity. The metal chelating activity of ferulic acid and caffeic acid has been proven by AAlikhani et al. (2022). Indeed, polyphenols can protect unsaturated lipids by binding to metal ions, such as iron and copper, which can normally trigger lipid oxidation (Bayram & Decker, 2023b; Villeneuve et al., 2023). Free ions of iron and copper can catalyze the activity of free radicals, induce the generation of reactive oxygen species (ROS), and function as prooxidants, accelerating the formation of primary and secondary products of oxidation (Kolev, 2022). Therefore, measuring the metal chelating activity of phenolic extracts is prominent to evaluate their potential effect in delaying lipid oxidation in real food systems.
The cupric (a) and ferrous (b) ion chelating activity of the optimized extracts in different concentrations. Data is presented as the mean ± SD (n = 3). Different letters show that there is a significant difference (p < 0.05) among different concentrations of optimized extract, according to one-way ANOVA and Tukey’s honestly significant difference (HSD) tests
Effect of optimized extract on delaying lipid oxidation
Lipid oxidation is the deterioration of unsaturated lipids, which causes off-flavors and aromas ("rancidity"), a decrease in nutritional value, and an increase in food waste. Moreover, manufacturers are incorporating higher amounts of unsaturated lipids into their products for health reasons, thereby making them more sensitive to oxidative degradation. Therefore, finding effective solutions to slow down lipid oxidation is crucial (Villeneuve et al., 2023). To this end, one of the most widely used strategies to protect unsaturated lipids is incorporating antioxidants during product formulation. In this work, the formation of volatile secondary oxidation products in five different oils was measured before and after the addition of BHT and the optimized extract of SBLs as a synthetic and natural antioxidant, respectively. The volatile oxidation products, generated due to high temperature and airflow in the Rancimat method, cause a breakpoint in the plotted curve of conductivity. The time in which this breakpoint occurs is known as the induction period, and a shorter induction period indicates faster lipid oxidation (Quiroga et al., 2013).
Figure 4 shows the induction period (a) and antioxidant activity index (b) of different oils fortified with the 300 µmol GAE/L TPC of optimized extract or 900 µmol/L of BHT. The results show that the induction period in all types of tested oils increased upon the addition of the optimized extract of SBLs with a phenolic concentration of 300 µmol GAE/L (p < 0.05). An increase in the oxidative stability of corn oil using phenolic extracts from various plants has been similarly reported in the literature (Lante et al., 2011; D. S. Mihaylova et al., 2014; Şahin et al., 2017). According to the induction periods and AAI of the oils, almost in all the cases, the optimized extract of SBLs had better functionality in increasing the oxidative stability of the oils, compared to BHT. This can confirm that the obtained extract could be a reliable substitute for synthetic antioxidants.
Induction period (a) and antioxidant activity index (b) of different oils fortified with the 300 µmol GAE/L TPC of optimized extract or 900 µmol/L of BHT. Data is presented as the mean ± SD (n = 3). Different small letters show that there is a significant difference (p < 0.05) in the induction periods within the same type of oil, according to one-way ANOVA and Tukey’s HSD tests. Different capital letters for AAI show that there is a significant difference (p< 0.05) between the antioxidant activities of optimized extract and BHT in the same type of oil, according to a t test
The more efficient inhibition of lipid oxidation with the obtained extract could be related to its phenolic profile, making it able to increase the oxidative shelf life of oil-containing foods (Ebrahimi & Lante, 2021). This increase is mostly associated with the ability of phenolic compounds to donate their hydrogen to free lipid radicals, as well as their ability to chelate prooxidant metal ions as shown in Fig. 3. Similarly, the major phenolic compounds present in the optimized extract were trans-ferulic acid, 3,4-dihydroxybenzoic acid, caffeic acid, and gallic acid (Table 4), whose free radical scavenging and metal chelating activities due to the presence of hydroxyl groups have been proven (Borges et al., 2003; Moran et al., 1997; Truong et al., 2020). The highest oxidative stability and AAI was obtained for peanut oil, which could be associated with its high monounsaturated fatty acid content (e.g., oleic acid) compared to high polyunsaturated fatty acid content (e.g., linoleic acid, linolenic acid) of other oils tested (Urszula Fabiszewska & Białecka-Florjańczyk, 2014). It is crucial to note that this test was performed at accelerated shelf-life conditions at 120 ℃, meaning that the increase in induction periods would correspond to a more noticeable shelf life at room temperature storage.
Conclusion
This paper highlights that many challenges are still associated with using extracts recovered from food by-products. In this respect, the co-extraction of chlorophylls with polyphenols limits the practical utilization of phenolic extracts in food products because these chromophores can change the color of food and may increase the risk of photo-oxidation. For this reason, the green and cost-effective extraction condition proposed in the present paper can help design an extract rich in phenolic compounds with the lowest chlorophyll concentration. Although extraction time and amplitude in the optimization were found to be insignificant for some responses, their inclusion in the initial experimental design was crucial for evaluating their potential impact and ensuring that no relevant factors were overlooked. However, this approach may have led to a more complex optimization process than necessary. Future studies may benefit from incorporating preliminary screening tests to streamline the optimization process, potentially simplifying the experimental design and focusing on the most impactful variables.
Key insights from this research include the successful optimization of the extraction process, highlighting the remarkable metal chelating activity exhibited by the optimized extract and demonstrating its potential as a viable alternative to synthetic antioxidants, such as BHT. Furthermore, the observed efficacy of this phenolic extract in delaying lipid oxidation in various vegetable oils underscores its potential as a natural antioxidant sourced from an agricultural food by-product. The findings of this study could be a booster for further exploration and utilization of such natural resources in sustainable food preservation strategies. Additionally, future research should evaluate the impact of the natural phenolic extract on the sensory properties of food products.
Data Availability
The data will be available upon request.
References
AAlikhani, M., Khalili, M., & Jahanshahi, M. (2022). The natural iron chelators’ ferulic acid and caffeic acid rescue mice’s brains from side effects of iron overload. Frontiers in Neurology, 13(3). https://doi.org/10.3389/fneur.2022.951725
Akyüz, A., & Ersus, S. (2021). Optimization of enzyme assisted extraction of protein from the sugar beet (Beta vulgaris L.) leaves for alternative plant protein concentrate production. Food Chemistry, 335, 127673. https://doi.org/10.1016/j.foodchem.2020.127673
Battistella Lasta, H. F., Lentz, L., Gonçalves Rodrigues, L. G., Mezzomo, N., Vitali, L., & Salvador Ferreira, S. R. (2019). Pressurized liquid extraction applied for the recovery of phenolic compounds from beetroot waste. Biocatalysis and Agricultural Biotechnology, 21, 101353. https://doi.org/10.1016/j.bcab.2019.101353
Bayram, I., & Decker, E. A. (2023a). Underlying mechanisms of synergistic antioxidant interactions during lipid oxidation. Trends in Food Science & Technology, 133(February), 219–230. https://doi.org/10.1016/j.tifs.2023.02.003
Bayram, I., & Decker, E. A. (2023b). Analysis of the mechanism of antioxidant synergism between α‐tocopherol and myricetin in bulk oil. Journal of the American Oil Chemists’ Society, 1–16. https://doi.org/10.1002/aocs.12792
Bayram, I., Laze, A., & Decker, E. A. (2023). Synergistic Mechanisms of Interactions between Myricetin or Taxifolin with α-Tocopherol in Oil-in-Water Emulsions. Journal of Agricultural and Food Chemistry, 71(24), 9490–9500. https://doi.org/10.1021/acs.jafc.3c01226
Bengardino, M. B., Fernandez, M. V., Nutter, J., Jagus, R. J., & Agüero, M. V. (2019). Recovery of bioactive compounds from beet leaves through simultaneous extraction: Modelling and process optimization. Food and Bioproducts Processing, 118, 227–236. https://doi.org/10.1016/j.fbp.2019.09.013
Borges, F., Lima, J. L. F. C., Pinto, I., Reis, S., & Siquet, C. (2003). Application of a Potentiometric System with Data-Analysis Computer Programs to the Quantification of Metal-Chelating Activity of Two Natural Antioxidants: Caffeic Acid and Ferulic Acid. Helvetica Chimica Acta, 86(9), 3081–3087. https://doi.org/10.1002/hlca.200390250
Cassiana Frohlich, P., Andressa Santos, K., Din Mahmud Hasan, S., & Antônio da Silva, E. (2022). Evaluation of the ethanolic ultrasound-assisted extraction from clove (Syzygium aromaticum) leaves and chemical characterization of the extracts. Food Chemistry, 373(Part A), 131351. https://doi.org/10.1016/j.foodchem.2021.131351
Cisneros-Yupanqui, M., Chalova, V. I., Kalaydzhiev, H. R., Mihaylova, D., Krastanov, A. I., & Lante, A. (2021). Preliminary characterisation of wastes generated from the rapeseed and sunflower protein isolation process and their valorisation in delaying oil oxidation. Food and Bioprocess Technology, 14(10), 1962–1971. https://doi.org/10.1007/s11947-021-02695-y
Codex Alimentarius. (2023). Codex General Standard for Food Additives" (GSFA, Codex STAN 192–1995). Codex General Standard for Food Additives" (GSFA, Codex STAN 192–1995). https://www.fao.org/fao-who-codexalimentarius/codex-texts/dbs/gsfa/en/. Accessed 29 January 2024
Culler, M. D., Bayram, I., & Decker, E. A. (2022). Enzymatic Modification of Lecithin for Improved Antioxidant Activity in Combination with Tocopherol in Emulsions and Bulk Oil. Journal of Agricultural and Food Chemistry, 70(41), 13404–13412. https://doi.org/10.1021/acs.jafc.2c05182
Derrien, M., Aghabararnejad, M., Gosselin, A., Desjardins, Y., Angers, P., & Boumghar, Y. (2018). Optimization of supercritical carbon dioxide extraction of lutein and chlorophyll from spinach by-products using response surface methodology. Lwt, 93(March), 79–87. https://doi.org/10.1016/j.lwt.2018.03.016
Derrien, M., Badr, A., Gosselin, A., Desjardins, Y., & Angers, P. (2017). Optimization of a green process for the extraction of lutein and chlorophyll from spinach by-products using response surface methodology (RSM). Lwt, 79, 170–177. https://doi.org/10.1016/j.lwt.2017.01.010
Dukić, J., Hunić, M., Nutrizio, M., & RežekJambrak, A. (2022). Influence of High-Power Ultrasound on Yield of Proteins and Specialized Plant Metabolites from Sugar Beet Leaves (Beta vulgaris subsp. vulgaris var. altissima). Applied Sciences, 12(18), 8949. https://doi.org/10.3390/app12188949
Ebrahimi, P., & Lante, A. (2021). Polyphenols: A comprehensive review of their nutritional properties. The Open Biotechnology Journal, 15(1), 164–172. https://doi.org/10.2174/1874070702115010164
Ebrahimi, P., & Lante, A. (2022). Environmentally friendly techniques for the recovery of polyphenols from food by-products and their impact on polyphenol oxidase: A critical review. Applied Sciences, 12(4), 1923. https://doi.org/10.3390/app12041923
Ebrahimi, P., Bayram, I., Lante, A., & Decker, E. A. (2024). Antioxidant and prooxidant activity of acid-hydrolyzed phenolic extracts of sugar beet leaves in oil-in-water emulsions. Journal ofthe American Oil Chemists’ Society, 1–11. https://doi.org/10.1002/aocs.12891
Ebrahimi, P., Hoxha, L., Mihaylova, D., Nicoletto, M., & Lante, A. (2024b). UV-A Treatment of Phenolic Extracts Impacts Colour, Bioactive Compounds, and Antioxidant Activity. Journal of the Science of Food and Agriculture. https://doi.org/10.1002/jsfa.13780
Ebrahimi, P., Mihaylova, D., Marangon, C. M., Grigoletto, L., & Lante, A. (2022). Impact of sample pretreatment and extraction methods on the bioactive compounds of sugar beet (Beta vulgaris L ) leaves. Molecules, 27(22), 8110. https://doi.org/10.3390/molecules27228110
Ebrahimi, P., Shahidi, S.-A., & Bijad, M. (2020). A rapid voltammetric strategy for determination of ferulic acid using electrochemical nanostructure tool in food samples. Journal of Food Measurement and Characterization, 14(6), 3389–3396. https://doi.org/10.1007/s11694-020-00585-z
Ebrahimi, P., Shokramraji, Z., Tavakkoli, S., Mihaylova, D., & Lante, A. (2023). Chlorophylls as Natural Bioactive Compounds Existing in Food By-Products: A Critical Review. Plants, 12(7), 1533. https://doi.org/10.3390/plants12071533
El-Gengaihi, S. E., Hamed, M. A., Aboubaker, D. H., & Mossa, A. T. H. (2016). Flavonoids from sugar beet leaves as hepatoprotective agent. International Journal of Pharmacy and Pharmaceutical Sciences, 8(4), 281–286.
Ferrando, B. O., Baenas, N., Rincón, F., & Periago, M. J. (2023). Green Extraction of Carotenoids from Tomato By-products Using Sodium Dodecyl Sulphate. Food and Bioprocess Technology, (0123456789). https://doi.org/10.1007/s11947-023-03292-x
González-Silva, N., Nolasco-González, Y., Aguilar-Hernández, G., Sáyago-Ayerdi, S. G., Villagrán, Z., Acosta, J. L., et al. (2022). Ultrasound-Assisted Extraction of Phenolic Compounds from Psidium cattleianum Leaves: Optimization Using the Response Surface Methodology. Molecules, 27(11), 3557. https://doi.org/10.3390/molecules27113557
Kaur, B., Panesar, P. S., & Anal, A. K. (2022). Standardization of ultrasound assisted extraction for the recovery of phenolic compounds from mango peels. Journal of Food Science and Technology, 59(7), 2813–2820. https://doi.org/10.1007/s13197-021-05304-0
Kolev, N. D. (2022). Natural antioxidants – an alternative for reduction of nitrites in cooked meat products. Food Science and Applied Biotechnology, 5(1), 64–76. https://doi.org/10.30721/fsab2022.v5.i1.167
Kumar, M. trends in extraction of plant bioactives using green technologies: A. review, Dahuja, A., Tiwari, S., Punia, S., Tak, Y., Amarowicz, R., et al. (2021). Recent trends in extraction of plant bioactives using green technologies: A review. Food Chemistry, 353(October 2020), 129431. https://doi.org/10.1016/j.foodchem.2021.129431
Lante, A., Nardi, T., Zocca, F., Giacomini, A., & Corich, V. (2011). Evaluation of Red Chicory Extract as a Natural Antioxidant by Pure Lipid Oxidation and Yeast Oxidative Stress Response as Model Systems. Journal of Agriculture and Food Chemistry, 59, 5318–5324. https://doi.org/10.1021/jf2003317
Lichtenthaler, H. K., & Buschmann, C. (2001). Chlorophylls and Carotenoids: Measurement and Characterization by Spectroscopy. Current Protocols in Food Analytical Chemistry, 1(1), F4.3.1-F4.3.8. https://doi.org/10.1002/0471142913.faf0403s01
Linares, G., & Rojas, M. L. (2022). Ultrasound-Assisted Extraction of Natural Pigments From Food Processing By-Products: A Review. Frontiers in Nutrition, 9(May), 1–17. https://doi.org/10.3389/fnut.2022.891462
Liu, H., Cao, J., & Jiang, W. (2015). Evaluation and comparison of vitamin C, phenolic compounds, antioxidant properties and metal chelating activity of pulp and peel from selected peach cultivars. Lwt, 63(2), 1042–1048. https://doi.org/10.1016/j.lwt.2015.04.052
Maravić, N., Teslić, N., Nikolić, D., Dimić, I., Šereš, Z., & Pavlić, B. (2022). From agricultural waste to antioxidant-rich extracts: Green techniques in extraction of polyphenols from sugar beet leaves. Sustainable Chemistry and Pharmacy, 28(May), 1–12. https://doi.org/10.1016/j.scp.2022.100728
Marcheafave, G. G., Tormena, C. D., Pauli, E. D., Rakocevic, M., Bruns, R. E., & Scarminio, I. S. (2019). Experimental mixture design solvent effects on pigment extraction and antioxidant activity from Coffea arabica L. leaves. Microchemical Journal, 146(January), 713–721. https://doi.org/10.1016/j.microc.2019.01.073
Mehmood, T., Ahmed, A., Ahmad, A., Ahmad, M. S., & Sandhu, M. A. (2018). Optimization of mixed surfactants-based β-carotene nanoemulsions using response surface methodology: An ultrasonic homogenization approach. Food Chemistry, 253(October 2017), 179–184. https://doi.org/10.1016/j.foodchem.2018.01.136
Miao, M., Chen, X., Wu, Z., Liu, J., Xu, C., Zhang, Z., & Wang, J. (2022). Extraction, Composition, and Antioxidant Activity of Flavonoids from Xanthoceras sorbifolium Bunge Leaves. Journal of AOAC INTERNATIONAL, 106(May 2022), 769–777. https://doi.org/10.1093/jaoacint/qsac148
Mihaylova, D. S., Lante, A., Tinello, F., & Krastanov, A. I. (2014). Study on the antioxidant and antimicrobial activities of Allium ursinum L. pressurised-liquid extract. Natural Product Research, 28(22), 2000–2005. https://doi.org/10.1080/14786419.2014.923422
Mihaylova, D., Desseva, I., Stoyanova, M., Petkova, N., Terzyiska, M., & Lante, A. (2021). Impact of In Vitro Gastrointestinal Digestion on the Bioaccessibility of Phytochemical Compounds from Eight Fruit Juices. Molecules, 26(4), 1187. https://doi.org/10.3390/molecules26041187
Moran, J. F., Klucas, R. V., Grayer, R. J., Abian, J., & Becana, M. (1997). Complexes of Iron with Phenolic Compounds from Soybean Nodules and Other Legume Tissues: Prooxidant and Antioxidant Properties. Free Radical Biology and Medicine, 22(5), 861–870. https://doi.org/10.1016/S0891-5849(96)00426-1
Ninčević Grassino, A., Ostojić, J., Miletić, V., Djaković, S., Bosiljkov, T., Zorić, Z., et al. (2020). Application of high hydrostatic pressure and ultrasound-assisted extractions as a novel approach for pectin and polyphenols recovery from tomato peel waste. Innovative Food Science and Emerging Technologies, 64(June). https://doi.org/10.1016/j.ifset.2020.102424
Nutter, J., Fernandez, M. V., Jagus, R. J., & Agüero, M. V. (2020). Development of an aqueous ultrasound-assisted extraction process of bioactive compounds from beet leaves: A proposal for reducing losses and increasing biomass utilization. Journal of the Science of Food and Agriculture, 101(5), 1989–1997. https://doi.org/10.1002/jsfa.10815
Palos-Hernández, A., Gutiérrez Fernández, M. Y., EscuadraBurrieza, J., Pérez-Iglesias, J. L., & González-Paramás, A. M. (2022). Obtaining green extracts rich in phenolic compounds from underexploited food by-products using natural deep eutectic solvents. Opportunities and challenges. Sustainable Chemistry and Pharmacy, 29, 100773. https://doi.org/10.1016/j.scp.2022.100773
Papoutsis, K., Pristijono, P., Golding, J. B., Stathopoulos, C. E., Bowyer, C. M., Scarlett, J. C., & Vuong, V. Q. (2018). Optimizing a sustainable ultrasound-assisted extraction method for the recovery of polyphenols from lemon by-products: comparison with hot water and organic solvent extractions. European Food Research and Technology, 244, 1353–1365. https://doi.org/10.1007/s00217-018-3049-9
Quiroga, P. R., Grosso, N. R., Lante, A., Lomolino, G., Zygadlo, J. A., & Nepote, V. (2013). Chemical composition, antioxidant activity and anti-lipase activity of Origanum vulgare and Lippia turbinata essential oils. International Journal of Food Science & Technology, 48(3), 642–649. https://doi.org/10.1111/ijfs.12011
Rebollo-Hernanz, M., Cañas, S., Taladrid, D., Segovia, Á., Bartolomé, B., Aguilera, Y., & Martín-Cabrejas, M. A. (2021). Extraction of phenolic compounds from cocoa shell: Modeling using response surface methodology and artificial neural networks. Separation and Purification Technology, 270, 118779. https://doi.org/10.1016/j.seppur.2021.118779
Şahin, S., Bilgin, M., Sayım, E., & Güvenilir, B. (2017). Effects of natural antioxidants in the improvement of corn oil quality: olive leaf vs. lemon balm. International Journal of Food Science and Technology, 52(2), 374–380. https://doi.org/10.1111/ijfs.13291
Shahidi, S.-A. (2022). Effect of solvent type on ultrasound-assisted extraction of antioxidant compounds from Ficaria kochii: Optimization by response surface methodology. Food and Chemical Toxicology, 163, 112981. https://doi.org/10.1016/j.fct.2022.112981
Sharma, M., Usmani, Z., Gupta, V. K., & Bhat, R. (2021). Valorization of fruits and vegetable wastes and by-products to produce natural pigments. Critical Reviews in Biotechnology, 41(4), 535–563. https://doi.org/10.1080/07388551.2021.1873240
Sharma, S., Chakkaravarthi, S., & Bhattacharya, B. (2023). Enhancement of oxidative stability of soybean oil via nano-emulsification of eggplant peel extract: Process development and application. Food Chemistry, 402(September 2022), 134249. https://doi.org/10.1016/j.foodchem.2022.134249
Skendi, A., Irakli, M., Chatzopoulou, P., Bouloumpasi, E., & Biliaderis, C. G. (2022). Phenolic extracts from solid wastes of the aromatic plant essential oil industry: Potential uses in food applications. Food Chemistry Advances, 1(April), 100065. https://doi.org/10.1016/j.focha.2022.100065
Truong, D. H., Nhung, N. T. A., & Dao, D. Q. (2020). Iron ions chelation-based antioxidant potential vs. pro-oxidant risk of ferulic acid: A DFT study in aqueous phase. Computational and Theoretical Chemistry, 1185(April), 112905. https://doi.org/10.1016/j.comptc.2020.112905
Tzima, K., Brunton, N. P., & Rai, D. K. (2020). Evaluation of the impact of chlorophyll removal techniques on polyphenols in rosemary and thyme by-products. Journal of Food Biochemistry, 44(3), 1–22. https://doi.org/10.1111/jfbc.13148
UrszulaFabiszewska, A., & Białecka-Florjańczyk, E. (2014). Factors Influencing Synthesis of Extracellular Lipases By Yarrowia Lipolytica in Medium Containing Vegetable Oils. Journal of microbiology, biotechnology and food sciences, 4(3), 231–237. https://doi.org/10.15414/jmbfs.2014-15.4.3.231-237
Villeneuve, P., Bourlieu-Lacanal, C., Durand, E., Lecomte, J., McClements, D. J., & Decker, E. A. (2023). Lipid oxidation in emulsions and bulk oils: A review of the importance of micelles. Critical Reviews in Food Science and Nutrition, 63(20), 4687–4727. https://doi.org/10.1080/10408398.2021.2006138
Vissers, A., Kiskini, A., Hilgers, R., Marinea, M., Wierenga, P. A., Gruppen, H., & Vincken, J.-P. (2017). Enzymatic Browning in Sugar Beet Leaves ( Beta vulgaris L.): Influence of Caffeic Acid Derivatives, Oxidative Coupling, and Coupled Oxidation. Journal of Agricultural and Food Chemistry, 65(24), 4911–4920. https://doi.org/10.1021/acs.jafc.7b01897
Wen, L., Zhang, Z., Rai, D., Sun, D. W., & Tiwari, B. K. (2019). Ultrasound-assisted extraction (UAE) of bioactive compounds from coffee silverskin: Impact on phenolic content, antioxidant activity, and morphological characteristics. Journal of Food Process Engineering, 42(6), 1–11. https://doi.org/10.1111/jfpe.13191
Zhang, S., Xie, H., Huang, J., Chen, Q., Li, X., Chen, X., et al. (2024). Ultrasound-assisted extraction of polyphenols from pine needles (Pinus elliottii): Comprehensive insights from RSM optimization, antioxidant activity, UHPLC-Q-Exactive Orbitrap MS/MS analysis and kinetic model. Ultrasonics Sonochemistry, 102(December 2023). https://doi.org/10.1016/j.ultsonch.2023.106742
Złotek, U., Mikulska, S., Nagajek, M., & Świeca, M. (2016). The effect of different solvents and number of extraction steps on the polyphenol content and antioxidant capacity of basil leaves (Ocimum basilicum L.) extracts. Saudi Journal of Biological Sciences, 23(5), 628–633. https://doi.org/10.1016/j.sjbs.2015.08.002
Acknowledgements
The authors acknowledge financial support (PhD scholarship) from Fondazione Cassa di Risparmio di Padova e Rovigo (CARIPARO).
Funding
Open access funding provided by Università degli Studi di Padova within the CRUI-CARE Agreement. No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
Peyman Ebrahimi: investigation, conceptualization formal analysis, data curation, visualization, writing original draft. Ipek Bayram: methodology, validation, writing—review and editing. Dasha Mihaylova: methodology, writing—review and editing. Anna Lante: conceptualization, resources, writing—review and editing, supervision. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ebrahimi, P., Bayram, I., Mihaylova, D. et al. A Strategy to Minimize the Chlorophyll Content in the Phenolic Extract of Sugar Beet Leaves: Can this Extract Work as a Natural Antioxidant in Vegetable Oils?. Food Bioprocess Technol (2024). https://doi.org/10.1007/s11947-024-03601-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11947-024-03601-y