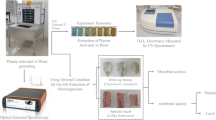
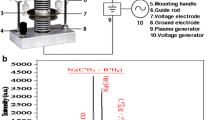
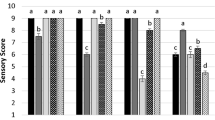
Abstract
The aim of this study was to determine the optimal conditions for gliding arc plasma using a response surface methodology and consider a low total mesophilic aerobic bacteria (TMAB) count, peroxide value (PV), color differences (ΔE), and appropriate sensory properties of stored Pacific white shrimp (PWS; Litopenaeus vannamei). A Box–Behnken design was used with three independent variables: argon gas (50–100%), treatment time (2–10 min), and sample distance (3–7 cm). Argon gas and treatment time significantly influenced TMAB, PV, ΔE, and sensory properties of the PWS, while sample distance had less effect. A high concentration (100%) of argon gas in the plasma caused a decrease in TMAB (1.2 CFU/g) compared to low concentrations (50%) of argon gas (5.41 CFU/ml). Also, high concentrations of argon gas cause a decrease in PV and an enhancement in ΔE and in the sensory properties of the shrimp. The results show that the use of 94.15% argon gas for 7.10 min with a sample distance of 6.54 cm provides optional gliding arc plasma conditions of 76.3% desirability. Using these numbers while maintaining desirable properties in stored PWS caused an identical reduction of Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa, and Proteus mirabilis under in vitro and in vivo conditions. In optimal conditions, concentrations of H2O2, nitrate, and nitrite in plasma-activated water (PAW) measured were 0.92 mM, 19.42 μM, and 9.62 μM respectively. Scanning electron microscopy (SEM) analysis demonstrated that optimized plasma can destroy the cells of Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa, and Proteus mirabilis bacteria.







Similar content being viewed by others
Data Availability
Not applicable.
References
Al-Rawaf, A. F., Fuliful, F. K., Khalaf, M. K., & Oudah, H. (2018). Studying the non-thermal plasma jet characteristics and application on bacterial decontamination. Journal of Theoretical and Applied Physics, 12(1), 45–51. https://doi.org/10.1007/s40094-018-0279-y
Annamalai, J., Sivam, V., Unnikrishnan, P., Sivasankara, S. K., Pansingh, R. K., Abdul, K. S., & Nagarajarao, R. C. (2020). Effect of electron beam irradiation on the biochemical, microbiological and sensory quality of Litopenaeus vannamei during chilled storage. Journal of Food Science and Technology, 57(6), 2150–2158. https://doi.org/10.1002/fft2.80
Bagheri, H., Abbaszadeh, S., & Salari, A. (2020). Optimization of decontamination conditions for Aspergillus flavus inoculated to military rations snack and physicochemical properties with atmospheric cold plasma. Journal of Food Safety, 40(6), e12850. https://doi.org/10.1111/jfs.12850
Beyrer, M., Smeu, I., Martinet, D., Howling, A., Pina-Pérez, M. C., & Ellert, C. (2020). Cold atmospheric plasma inactivation of microbial spores compared on reference surfaces and powder particles. Food and Bioprocess Technology, 13(5), 827–837. https://doi.org/10.1007/s11947-020-02438-5
Castro, D. R. G., Mar, J. M., da Silva, L. S., da Silva, K. A., Sanches, E. A., de Araújo Bezerra, J., & Campelo, P. H. (2020). Improvement of the bioavailability of Amazonian juices rich in bioactive compounds using glow plasma technique. Food and Bioprocess Technology, 13(4), 670–679. https://doi.org/10.1007/s11947-020-02427-8
da Silva Campelo, M. C., & de OLIVEIRA SOARES REBOUÇAS, L., de Oliveira Vitoriano, J., ALVES, C., ALVES da SILVA, J. B., & de Oliveira Lima, P. (2019). Use of cold atmospheric plasma to preserve the quality of white shrimp (Litopenaeus vannamei). Journal of Food Protection, 82(7), 1217–1223. https://doi.org/10.4315/0362-028X.JFP-18-369
Darvish, F., Sarkari, N. M., Khani, M., Eslami, E., Shokri, B., Mohseni, M., & Dee, C. F. (2020). Direct plasma treatment approach based on non-thermal gliding arc for surface modification of biaxially-oriented polypropylene with post-exposure hydrophilicity improvement and minus aging effects. Applied Surface Science, 509, 144815. https://doi.org/10.1016/j.apsusc.2019.144815
Dasan, B. G., Onal-Ulusoy, B., Pawlat, J., Diatczyk, J., Sen, Y., & Mutlu, M. (2017). A new and simple approach for decontamination of food contact surfaces with gliding arc discharge atmospheric non-thermal plasma. Food and Bioprocess Technology, 10(4), 650–661. https://doi.org/10.1007/s11947-016-1847-2
de Souza Silva, D. A., da Silva Campelo, M. C., & de OLIVEIRA SOARES REBOUÇAS, L., de Oliveira Vitoriano, J., ALVES, C., Alves da Silva, J. B., & de Oliveira Lima, P. (2019). Use of cold atmospheric plasma to preserve the quality of white shrimp (Litopenaeus vannamei). Journal of Food Protection, 82(7), 1217–1223. https://doi.org/10.4315/0362-028X.JFP-18-369
Dong, X. Y., & Yang, Y. L. (2019). A novel approach to enhance blueberry quality during storage using cold plasma at atmospheric air pressure. Food and Bioprocess Technology, 12(8), 1409–1421. https://doi.org/10.1007/s11947-019-02305-y
Du, S., Zhang, Z., Xiao, L., Lou, Y., Pan, Y., & Zhao, Y. (2016). Acidic electrolyzed water as a novel transmitting medium for high hydrostatic pressure reduction of bacterial loads on shelled fresh shrimp. Frontiers in Microbiology, 7, 305. https://doi.org/10.3389/fmicb.2016.00305
El-Zein, A., Talaat, M., El-Aragi, G., & El-Amawy, A. (2016). Electrical characteristics of nonthermal gliding arc discharge reactor in argon and nitrogen gases. IEEE Transactions on Plasma Science, 44(7), 1155–1159. https://doi.org/10.1109/TPS.2016.2578301
Elliot, M., Chen, J., Chen, D.-Z., Hu, X.-M., Ekaterina, N., & Deng, S.-G. (2021). Effects of a cold plasma-assisted shrimp processing chain on biochemical and sensory quality alterations in Pacific white shrimps (Penaeus vannamei). Food Science and Technology International. https://doi.org/10.1177/10820132211050847
Ghimire, B., Szili, E. J., Patenall, B. L., Lamichhane, P., Gaur, N., Robson, A. J., & Choi, E. H. (2021). Enhancement of hydrogen peroxide production from an atmospheric pressure argon plasma jet and implications to the antibacterial activity of plasma activated water. Plasma Sources Science and Technology, 30(3), 035009. https://doi.org/10.1088/1361-6595/abe0c9
Gök, V., Aktop, S., Özkan, M., & Tomar, O. (2019). The effects of atmospheric cold plasma on inactivation of Listeria monocytogenes and Staphylococcus aureus and some quality characteristics of pastırma—a dry-cured beef product. Innovative Food Science & Emerging Technologies, 56, 102188. https://doi.org/10.1016/j.ifset.2019.102188
Gonçalves, A. A., & de Oliveira, A. R. M. (2016). Melanosis in crustaceans: A review. LWT-Food Science and Technology, 65, 791–799. https://doi.org/10.1016/j.lwt.2015.09.011
Jambrak, A. R., Vukušić, T., Stulić, V., Mrvčić, J., Milošević, S., Šimunek, M., & Herceg, Z. (2015). The effect of high power ultrasound and cold gas-phase plasma treatments on selected yeast in pure culture. Food and Bioprocess Technology, 8(4), 791–800. https://doi.org/10.1007/s11947-014-1442-3
Joshi, S. G., Cooper, M., Yost, A., Paff, M., Ercan, U. K., Fridman, G., & Brooks, A. D. (2011). Nonthermal dielectric-barrier discharge plasma-induced inactivation involves oxidative DNA damage and membrane lipid peroxidation in Escherichia coli. Antimicrobial Agents and Chemotherapy, 55(3), 1053–1062. https://doi.org/10.1128/AAC.01002-10
Kamgang-Youbi, G., Herry, J. M., Meylheuc, T., Brisset, J. L., Bellon-Fontaine, M. N., Doubla, A., & Naitali, M. (2009). Microbial inactivation using plasma-activated water obtained by gliding electric discharges. Letters in Applied Microbiology, 48(1), 13–18. https://doi.org/10.1111/j.1472-765X.2008.02476.x
Khodadadi, I., Ardebili, S. M. S., Eyvazzadeh, O., Zargari, K., & Moradi, M. (2014). Comparing the sensitivity of spot test method and a novel computer vision system for iron detecting in fortificated flours. Food Analytical Methods, 7(6), 1213–1218. https://doi.org/10.1007/s12161-013-9735-0
Kim, H. S., Cho, Y. I., Hwang, I. H., Lee, D. H., Cho, D. J., Rabinovich, A., & Fridman, A. (2013). Use of plasma gliding arc discharges on the inactivation of E. coli in water. Separation and Purification Technology, 120, 423–428. https://doi.org/10.1016/j.seppur.2013.09.041
Lee, S. Y., Lee, W. K., Lee, J. W., Chung, M.-S., Oh, S.-W., Shin, J.-K., & Min, S. C. (2021). Microbial decontamination of rice germ using a large-scale plasma jet-pulsed light-ultraviolet-C integrated treatment system. Food and Bioprocess Technology, 14(3), 542–553. https://doi.org/10.1007/s11947-021-02590-6
Liao, X., Su, Y., Liu, D., Chen, S., Hu, Y., Ye, X., & Ding, T. (2018). Application of atmospheric cold plasma-activated water (PAW) ice for preservation of shrimps (Metapenaeus ensis). Food Control, 94, 307–314. https://doi.org/10.1016/j.foodcont.2018.07.026
Miguel, T. B. A. R., Porto, E. C. M., de Paiva Pinheiro, S. K., de Castro Miguel, E., Fernandes, F. A. N., & Rodrigues, S. (2021). Protective effect of natural and processed coconut water by non-thermal technologies against oxidative stress in brine shrimp (Artemia salina). Food and Bioprocess Technology, 14(4), 702–716. https://doi.org/10.1007/s11947-021-02600-7
Misra, N., Moiseev, T., Patil, S., Pankaj, S., Bourke, P., Mosnier, J., & Cullen, P. (2014). Cold plasma in modified atmospheres for post-harvest treatment of strawberries. Food and Bioprocess Technology, 7(10), 3045–3054. https://doi.org/10.1007/s11947-014-1356-0
Mitra, A., Li, Y.-F., Klämpfl, T. G., Shimizu, T., Jeon, J., Morfill, G. E., & Zimmermann, J. L. (2014). Inactivation of surface-borne microorganisms and increased germination of seed specimen by cold atmospheric plasma. Food and Bioprocess Technology, 7(3), 645–653. https://doi.org/10.1007/s11947-013-1126-4
Mošovská, S., Medvecká, V., Klas, M., Kyzek, S., Valík, Ľ, Mikulajová, A., & Zahoranová, A. (2022). Decontamination of Escherichia coli on the surface of soybean seeds using plasma activated water. LWT, 154, 112720. https://doi.org/10.1016/j.lwt.2021.112720
Mousavi, M., Heshmati, A., Garmakhany, A. D., Vahidinia, A., & Taheri, M. (2019). Optimization of the viability of Lactobacillus acidophilus and physico-chemical, textural and sensorial characteristics of flaxseed-enriched stirred probiotic yogurt by using response surface methodology. LWT, 102, 80–88. https://doi.org/10.1016/j.lwt.2018.12.023
Niedźwiedź, I., Juzwa, W., Skrzypiec, K., Skrzypek, T., Waśko, A., Kwiatkowski, M., & Polak-Berecka, M. (2020). Morphological and physiological changes in Lentilactobacillus hilgardii cells after cold plasma treatment. Scientific Reports, 10(1), 1–14. https://doi.org/10.1038/s41598-020-76053-x
Nirmal, N. P., & Benjakul, S. (2010). Effect of catechin and ferulic acid on melanosis and quality of Pacific white shrimp subjected to prior freeze–thawing during refrigerated storage. Food Control, 21(9), 1263–1271. https://doi.org/10.1016/j.foodcont.2010.02.015
Norton, T., & Sun, D.-W. (2008). Recent advances in the use of high pressure as an effective processing technique in the food industry. Food and Bioprocess Technology, 1(1), 2–34. https://doi.org/10.1007/s11947-007-0007-0
Okpala, C. O. R., Bono, G., Geraci, M. L., Sardo, G., Vitale, S., & Schaschke, C. J. (2016). Lipid oxidation kinetics of ozone-processed shrimp during iced storage using peroxide value measurements. Food Bioscience, 16, 5–10. https://doi.org/10.1016/j.fbio.2016.07.005
Okpala, C. O. R., Choo, W. S., & Dykes, G. A. (2014). Quality and shelf life assessment of Pacific white shrimp (Litopenaeus vannamei) freshly harvested and stored on ice. LWT-Food Science and Technology, 55(1), 110–116. https://doi.org/10.1016/j.lwt.2013.07.020
Olatunde, O. O., & Benjakul, S. (2018). Natural preservatives for extending the shelf-life of seafood: A revisit. Comprehensive Reviews in Food Science and Food Safety, 17(6), 1595–1612. https://doi.org/10.1111/1541-4337.12390
Olatunde, O. O., Benjakul, S., & Vongkamjan, K. (2019). Dielectric barrier discharge cold atmospheric plasma: Bacterial inactivation mechanism. Journal of Food Safety, 39(6), e12705. https://doi.org/10.1111/jfs.12705
Onal-Ulusoy, B. (2021). Effects of cold atmospheric gliding arc discharge plasma, non-thermal ultrasound, and low-temperature oven treatments on quality parameters of Turkish blossom honey. Food and Bioprocess Technology, 14(9), 1763–1771. https://doi.org/10.1007/s11947-021-02688-x
Patange, A., Lu, P., Boehm, D., Cullen, P., & Bourke, P. (2019). Efficacy of cold plasma functionalised water for improving microbiological safety of fresh produce and wash water recycling. Food Microbiology, 84, 103226. https://doi.org/10.1016/j.fm.2019.05.010
Pazoki, M., Rahnama, F., Abbaszadeh, R., & Mirabdollah, E. (2018). Assessment of anti-bacterial activity of non-thermal plasma in sterilization of infectious wastes. Advances in Environmental Technology, 4(4), 197–202. https://doi.org/10.22104/aet.2019.3251.1160
Pérez-Andrés, J. M., Cropotova, J., Harrison, S. M., Brunton, N. P., Cullen, P. J., Rustad, T., & Tiwari, B. K. (2020). Effect of cold plasma on meat cholesterol and lipid oxidation. Foods, 9(12), 1786. https://doi.org/10.3390/foods9121786
Pilevar, Z., Hosseini, H., Abdollahzadeh, E., Shojaee-Aliabadi, S., Tajedin, E., Yousefi, M., & Khosroshahi, N. K. (2020). Effect of Zataria multiflora Boiss. Essential oil, time, and temperature on the expression of Listeria monocytogenes virulence genes in broth and minced rainbow trout. Food Control, 109, 106863. https://doi.org/10.1016/j.foodcont.2019.106863
Puligundla, P., Kim, J.-W., & Mok, C. (2017). Effects of nonthermal plasma treatment on decontamination and sprouting of radish (Raphanus sativus L.) seeds. Food and bioprocess technology, 10(6), 1093–1102. https://doi.org/10.1007/s11947-017-1886-3
Qian, J., Ma, L., Yan, W., Zhuang, H., Huang, M., Zhang, J., & Wang, J. (2022). Inactivation kinetics and cell envelope damages of foodborne pathogens Listeria monocytogenes and Salmonella Enteritidis treated with cold plasma. Food Microbiology, 101, 103891. https://doi.org/10.1016/j.fm.2021.103891
Rabiey, S., Hosseini, H., & Rezaei, M. (2014). Use Carum copticum essential oil for controlling the Listeria monocytogenes growth in fish model system. Brazilian Journal of Microbiology, 45, 89–96. https://doi.org/10.1590/S1517-83822014000100013
Schmidt-Bleker, A., Winter, J., Bösel, A., Reuter, S., & Weltmann, K.-D. (2015). On the plasma chemistry of a cold atmospheric argon plasma jet with shielding gas device. Plasma Sources Science and Technology, 25(1), 015005. https://doi.org/10.1088/0963-0252/25/1/015005
Segura-Ponce, L. A., Reyes, J. E., Troncoso-Contreras, G., & Valenzuela-Tapia, G. (2018). Effect of low-pressure cold plasma (LPCP) on the wettability and the inactivation of Escherichia coli and Listeria innocua on fresh-cut apple (Granny Smith) skin. Food and Bioprocess Technology, 11(5), 1075–1086. https://doi.org/10.1007/s11947-018-2079-4
Senphan, T., & Benjakul, S. (2012). Compositions and yield of lipids extracted from hepatopancreas of Pacific white shrimp (Litopenaeus vannamei) as affected by prior autolysis. Food Chemistry, 134(2), 829–835. https://doi.org/10.1016/j.foodchem.2012.02.188
Sergeichev, K. F., Lukina, N. A., Sarimov, R. M., Smirnov, I. G., Simakin, A. V., Dorokhov, A. S., & Gudkov, S. V. (2021). Physicochemical properties of pure water treated by pure argon plasma jet generated by microwave discharge in opened atmosphere. Frontiers in Physics, 8, 614684. https://doi.org/10.3389/fphy.2020.614684
Shiekh, K. A., & Benjakul, S. (2020). Effect of high voltage cold atmospheric plasma processing on the quality and shelf-life of Pacific white shrimp treated with Chamuang leaf extract. Innovative Food Science & Emerging Technologies, 64, 102435. https://doi.org/10.1016/j.ifset.2020.102435
Shiekh, K. A., Benjakul, S., Qi, H., Zhang, B., & Deng, S. (2021). Combined hurdle effects of pulsed electric field and vacuum impregnation of Chamuang leaf extract on quality and shelf-life of Pacific white shrimp subjected to high voltage cold atmospheric plasma. Food Packaging and Shelf Life, 28, 100660. https://doi.org/10.1016/j.fpsl.2021.100660
Shimizu, T., Steffes, B., Pompl, R., Jamitzky, F., Bunk, W., Ramrath, K., & Urayama, T. (2006). Microwave plasma torch for bacterial sterilization. 6th Int. Conf. Reactive Plasmas, Matsushima, Japan,
Trondsen, T., Braaten, T., Lund, E., & Eggen, A. (2004). Consumption of seafood—the influence of overweight and health beliefs. Food Quality and Preference, 15(4), 361–374. https://doi.org/10.1016/S0950-3293(03)00083-1
Ulbin-Figlewicz, N., Jarmoluk, A., & Marycz, K. (2015). Antimicrobial activity of low-pressure plasma treatment against selected foodborne bacteria and meat microbiota. Annals of Microbiology, 65(3), 1537–1546. https://doi.org/10.1007/s13213-014-0992-y
Wang, J., Zhuang, H., & Zhang, J. (2016). Inactivation of spoilage bacteria in package by dielectric barrier discharge atmospheric cold plasma—treatment time effects. Food and Bioprocess Technology, 9(10), 1648–1652. https://doi.org/10.1007/s11947-016-1746-6
Xu, L., Yepez, X., Applegate, B., Keener, K. M., Tao, B., & Garner, A. L. (2020). Penetration and microbial inactivation by high voltage atmospheric cold plasma in semi-solid material. Food and Bioprocess Technology, 13(10), 1688–1702. https://doi.org/10.1007/s11947-020-02506-w
Yang, L., Chen, J., & Gao, J. (2009). Low temperature argon plasma sterilization effect on Pseudomonas aeruginosa and its mechanisms. Journal of Electrostatics, 67(4), 646–651. https://doi.org/10.1016/j.elstat.2009.01.060
Zhao, Y.-M., de Alba, M., Sun, D.-W., & Tiwari, B. (2019). Principles and recent applications of novel non-thermal processing technologies for the fish industry—A review. Critical Reviews in Food Science and Nutrition, 59(5), 728–742. https://doi.org/10.1080/10408398.2018.1495613
Ziuzina, D., Patil, S., Cullen, P., Keener, K., & Bourke, P. (2013). Atmospheric cold plasma inactivation of E scherichia coli in liquid media inside a sealed package. Journal of Applied Microbiology, 114(3), 778–787. https://doi.org/10.1111/jam.12087
Ziuzina, D., Patil, S., Cullen, P. J., Keener, K., & Bourke, P. (2014). Atmospheric cold plasma inactivation of Escherichia coli, Salmonella enterica serovar Typhimurium and Listeria monocytogenes inoculated on fresh produce. Food Microbiology, 42, 109–116. https://doi.org/10.1016/j.fm.2014.02.007
Zouelm, F., Abhari, K., Hosseini, H., & Khani, M. (2019). The effects of cold plasma application on quality and chemical spoilage of pacific white shrimp (Litopenaeus vannamei) during refrigerated storage. Journal of Aquatic Food Product Technology, 28(6), 624–636. https://doi.org/10.1080/10498850.2019.1627452
Acknowledgements
This research was supported by a grant from the National Nutrition and Food Technology Research Institute of Iran. Furthermore, this article was a part of a PhD dissertation at Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Funding
This work was supported by the Shahid Beheshti University of Medical Sciences [grant number 29026].
Author information
Authors and Affiliations
Contributions
MM, SMH, and HH designed and performed the experiments and wrote the manuscript. ASA, AH, and KA provided critical feedback and helped shape the research, analysis, and manuscript. MK and AA contributed to sample treatment by gliding arc device plasma. MT and FS performed the data analysis. All authors reviewed the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mousavi, M., Hosseini, S.M., Hosseini, H. et al. Gliding Arc Plasma Discharge Conditions on Microbial, Physicochemical, and Sensory Properties of Shrimp (Litopenaeus vannamei): In Vivo and In Vitro Studies. Food Bioprocess Technol 15, 2327–2343 (2022). https://doi.org/10.1007/s11947-022-02886-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-022-02886-1