Abstract
Purpose of Review
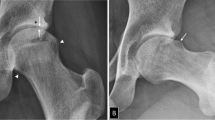
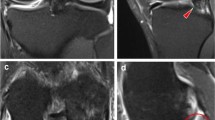
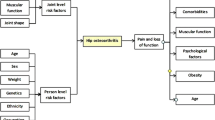
This review describes the contributions of abnormal bone circulation to the pathophysiology of osteoarthritis. Combining dynamic imaging with MRI and PET with previous observations reveals that venous stasis and a venous outlet syndrome is most likely the key circulatory pathology associated with the initiation or progression of osteoarthritis.
Recent Findings
MRI and PET have revealed that venous outflow obstruction results in physicochemical changes in subchondral bone to which osteoblasts are responsive. The osteoblasts express an altered pattern of cytokines, many of which can serve as structural or signaling molecules contributing to both bone remodeling and cartilage degeneration.
Summary
The patterns of circulatory changes are associated with alterations in the physicochemical environment of subchondral bone, including hypoxia. Osteoblast cytokines can transit the subchondral bone plate and calcified cartilage and communicate with chondrocytes.






Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Fell HB, Jubb RW. The effect of synovial tissue on the breakdown of articular cartilage in organ culture. Arthritis Rheum. 1977;20(7):1359–71.
Boniface RJ, Cain PR, Evans CH. Articular responses to purified cartilage proteoglycans. Arthritis Rheum. 1988;31(2):258–66.
Aaron RK, Sledge CB, Shortkroff S. Experimental arthritis induced by polysaccharide macromolecules. Arthritis Rheum. 1987;30(6):679–88.
Brooks M, Revell W. Blood supply of bone: scientific aspects. London: Springer; 1998.
Welch RD, Johnston 2nd CE, Waldron MJ, et al. Bone changes associated with intraosseous hypertension in the caprine tibia. J Bone Joint Surg Am. 1993;75(1):53–60.
Arnoldi CC. Vascular aspects of degenerative joint disorders. A synthesis. Acta Orthop Scand Suppl. 1994;261:1–82.
Arnoldi CC, Djurhuus JC, Heerfordt J, et al. Intraosseous phlebography, intraosseous pressure measurements and 99mTC-polyphosphate scintigraphy in patients with various painful conditions in the hip and knee. Acta Orthop Scand. 1980;51(1):19–28.
Arnoldi CC, Lemperg K, Linderholm H. Intraosseous hypertension and pain in the knee. J Bone Joint Surg Br. 1975;57(3):360–3.
Arnoldi CC, Linderholm H, Mussbichler H. Venous engorgement and intraosseous hypertension in osteoarthritis of the hip. J Bone Joint Surg Br. 1972;54(3):409–21.
Lemperg RK, Arnoldi CC. The significance of intraosseous pressure in normal and diseased states with special reference to the intraosseous engorgement-pain syndrome. Clin Orthop Relat Res. 1978;136:143–56.
• Aaron RK. Circulatory pathology in osteoarthritis. In: Aaron RK, editor. Skeletal circulation in clinical practice. Singapore: World Scientific; 2016. p. 233–51. Overview of the vascular pathophysiology in OA.
Ma HT, Griffith JF, Yeung DK, et al. Modified brix model analysis of bone perfusion in subjects of varying bone mineral density. J Magn Reson Imaging. 2010;31(5):1169–75.
Dyke JP, Aaron RK. Noninvasive methods of measuring bone blood perfusion. Ann N Y Acad Sci. 2010;1192:95–102.
Piert M, Zittel TT, Becker GA, et al. Assessment of porcine bone metabolism by dynamic [18] F fluoride ion PET: correlation with bone histomorphometry. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2001;42(7):1091–100.
Messa C, Goodman WG, Hoh CK, et al. Bone metabolic activity measured with positron emission tomography and [18F] fluoride ion in renal osteodystrophy: correlation with bone histomorphometry. J Clin Edocrinol Metab. 1993;77(4):949–55.
• Dyke JP. MRI and PET. In: Aaron RK, editor. Skeletal circulation in clinical practice. Singapore: World Scientific; 2016. p. 117–38. Description of the techniques and uses of dynamic imaging in OA bone.
Aaron RK, Dyke JP, Ciombor DM, et al. Perfusion abnormalities in subchondral bone associated with marrow edema, osteoarthritis, and avascular necrosis. Ann N Y Acad Sci. 2007;1117:124–37.
Lee JH, Dyke JP, Ballon D, et al. Subchondral fluid dynamics in a model of osteoarthritis: use of dynamic contrast-enhanced magnetic resonance imaging. Osteoarthr Cartil. 2009;17(10):1350–5.
Brix G, Semmler W, Port R, et al. Pharmacokinetic parameters in CNS Gd-DTPA enhanced MR imaging. J Comput Assist Tomogr. 1991;15(4):621–8.
Tofts PS, Brix G, Buckley DL, et al. Estimating kinetic parameters from dynamic contrast-enhanced T(1)-weighted MRI of a diffusable tracer: standardized quantities and symbols. J Magn Reson Imaging. 1999;10(3):223–32.
Tsai PH, Lee HS, Siow TY, et al. Abnormal perfusion in patellofemoral subchondral bone marrow in the rat anterior cruciate ligament transection model of post-traumatic osteoarthritis: a dynamic contrast-enhanced magnetic resonance imaging study. Osteoarthr Cartil. 2016;24(1):129–33.
Wang YX, Griffith JF, Deng M, et al. Compromised perfusion in femoral head in normal rats: distinctive perfusion MRI evidence of contrast washout delay. Br J Radiol. 2012;85(1016):e436–41.
• Dyke JP, Synan M, Ezell P, et al. Characterization of bone perfusion by dynamic contrast-enhanced magnetic resonance imaging and positron emission tomography in the Dunkin-Hartley guinea pig model of advanced osteoarthritis. J Orthop Res. 2015;33(3):366–72. Demonstration of the uses of dynamic and functional imaging to describe bone and circulatory physiology
van Dijke CF, Peterfy CG, Brasch RC, et al. MR imaging of the arthritic rabbit knee joint using albumin-(Gd-DTPA) 30 with correlation to histopathology. Magn Reson Imaging. 1999;17(2):237–45.
Frost ML, Siddique M, Blake GM, et al. Differential effects of teriparatide on regional bone formation using 18F-fluoride positron emission tomography. J Bone Miner Res. 2011;26(5):1002–11.
Piert M, Zittel TT, Machulla HJ, et al. Blood flow measurements with [(15)O] H2O and [18F] fluoride ion PET in porcine vertebrae. J Bone Miner Res. 1998;13(8):1328–36.
Temmerman OP, Raijmakers PG, Heyligers IC, et al. Bone metabolism after total hip revision surgery with impacted grafting: evaluation using H2 15O and [18F] fluoride PET; a pilot study. Molecular imaging and biology : MIB : the official publication of the Academy of Molecular Imaging. 2008;10(5):288–93.
Lee JH, Dyke JP, Ballon D, et al. Assessment of bone perfusion with contrast-enhanced magnetic resonance imaging. Orthop Clin North Am. 2009;40(2):249–57.
Kiaer T, Dahl B, Lausten G. Partial pressures of oxygen and carbon dioxide in bone and their correlation with bone-blood flow: effect of decreased arterial supply and venous congestion on intraosseous oxygen and carbon dioxide in an animal model. J Orthop Res. 1992;10(6):807–12.
Kiaer T, Gronlund J, Jensen B, et al. Effects of variation in systemic blood pressure on intraosseous pressure, PO2, and PCO2. J Orthop Res. 1990;8(4):618–22.
Kiaer T, Dahl B, Lausten GS. The relationship between inert gas wash-out and radioactive tracer microspheres in measurement of bone blood flow: effect of decreased arterial supply and venous congestion on bone blood flow in an animal model. J Orthop Res. 1993;11(1):28–35.
Kiaer T, Pedersen NW, Kristensen KD, et al. Intra-osseous pressure and oxygen tension in avascular necrosis and osteoarthritis of the hip. J Bone Joint Surg Br. 1990;72(6):1023–30.
Kiaer T, Gronlund J, Sorensen KH. Subchondral pO2, pCO2, pressure, pH, and lactate in human osteoarthritis of the hip. Clin Orthop Relat Res. 1988;229:149–55.
Kiaer T. Bone perfusion and oxygenation. Animal experiments and clinical observations. Acta Orthop Scand Suppl. 1994;257:1–41.
Imhof H, Breitenseher M, Kainberger F, et al. Degenerative joint disease: cartilage or vascular disease? Skelet Radiol. 1997;26(7):398–403.
Imhof H, Sulzbacher I, Grampp S, et al. Subchondral bone and cartilage disease: a rediscovered functional unit. Investig Radiol. 2000;35(10):581–8.
Watt I. Osteoarthritis revisited—again! Skelet Radiol. 2009;38(5):419–23.
Pedersen NW, Kiaer T, Kristensen KD, et al. Intraosseous pressure, oxygenation, and histology in arthrosis and osteonecrosis of the hip. Acta Orthop Scand. 1989;60(4):415–7.
Warren SM, Steinbrech DS, Mehrara BJ, et al. Hypoxia regulates osteoblast gene expression. J Surg Res. 2001;99(1):147–55.
Dodd JS, Raleigh JA, Gross TS. Osteocyte hypoxia: a novel mechanotransduction pathway. Am J Phys. 1999;277(3 Pt 1):C598–602.
Steinbrech DS, Mehrara BJ, Saadeh PB, et al. Hypoxia increases insulinlike growth factor gene expression in rat osteoblasts. Ann Plast Surg. 2000;44(5):529–34. discussion 534-525
Hillsley MV, Frangos JA. Bone tissue engineering: the role of interstitial fluid flow. Biotechnol Bioeng. 1994;43(7):573–81.
Wong SY, Evans RA, Needs C, et al. The pathogenesis of osteoarthritis of the hip. Evidence for primary osteocyte death. Clin Orthop Relat Res. 1987;214:305–12.
Tanaka SM, Sun HB, Roeder RK, et al. Osteoblast responses one hour after load-induced fluid flow in a three-dimensional porous matrix. Calcif Tissue Int. 2005;76(4):261–71.
McAllister TN, Frangos JA. Steady and transient fluid shear stress stimulate NO release in osteoblasts through distinct biochemical pathways. J Bone Miner Res. 1999;14(6):930–6.
Wadhwa S, Choudhary S, Voznesensky M, et al. Fluid flow induces COX-2 expression in MC3T3-E1 osteoblasts via a PKA signaling pathway. Biochem Biophys Res Commun. 2002;297(1):46–51.
Burr DB. The importance of subchondral bone in osteoarthrosis. Curr Opin Rheumatol. 1998;10(3):256–62.
Lavigne P, Benderdour M, Lajeunesse D, et al. Subchondral and trabecular bone metabolism regulation in canine experimental knee osteoarthritis. Osteoarthr Cartil. 2005;13(4):310–7.
Lahm A, Kreuz PC, Oberst M, et al. Subchondral and trabecular bone remodeling in canine experimental osteoarthritis. Arch Orthop Trauma Surg. 2006;126(9):582–7.
Westacott CI, Webb GR, Warnock MG, et al. Alteration of cartilage metabolism by cells from osteoarthritic bone. Arthritis Rheum. 1997;40(7):1282–91.
Hilal G, Martel-Pelletier J, Pelletier JP, et al. Osteoblast-like cells from human subchondral osteoarthritic bone demonstrate an altered phenotype in vitro: possible role in subchondral bone sclerosis. Arthritis Rheum. 1998;41(5):891–9.
Martel-Pelletier J, Faure MP, McCollum R, et al. Plasmin, plasminogen activators and inhibitor in human osteoarthritic cartilage. J Rheumatol. 1991;18(12):1863–71.
Pelletier JP, Mineau F, Faure MP, et al. Imbalance between the mechanisms of activation and inhibition of metalloproteases in the early lesions of experimental osteoarthritis. Arthritis Rheum. 1990;33(10):1466–76.
Sanchez C, Deberg MA, Piccardi N, et al. Subchondral bone osteoblasts induce phenotypic changes in human osteoarthritic chondrocytes. Osteoarthr Cartil. 2005;13(11):988–97.
Sanchez C, Deberg MA, Piccardi N, et al. Osteoblasts from the sclerotic subchondral bone downregulate aggrecan but upregulate metalloproteinases expression by chondrocytes. This effect is mimicked by interleukin-6, -1beta and oncostatin M pre-treated non-sclerotic osteoblasts. Osteoarthr Cartil. 2005;13(11):979–87.
Prasadam I, van Gennip S, Friis T, et al. ERK-1/2 and p38 in the regulation of hypertrophic changes of normal articular cartilage chondrocytes induced by osteoarthritic subchondral osteoblasts. Arthritis Rheum. 2010;62(5):1349–60.
• Goldring SR, Goldring MB. Changes in the osteochondral unit during osteoarthritis: structure, function and cartilage-bone crosstalk. Nat Rev Rheumatol. 2016;12(11):632–44. This study summarizes current information on bone-cartilage crosstalk
Suri S, Walsh DA. Osteochondral alterations in osteoarthritis. Bone. 2012;51(2):204–11.
Lyons TJ, McClure SF, Stoddart RW, et al. The normal human chondro-osseous junctional region: evidence for contact of uncalcified cartilage with subchondral bone and marrow spaces. BMC Musculoskelet Disord. 2006;7:52.
Simkin P, Peterson J. The cartilage/bone interface is permeable to saline under phsyiologic articular pressures. Osteoarthr Cartil. 2001;9(Supp 2):S4.
Arkill KP, Winlove CP. Solute transport in the deep and calcified zones of articular cartilage. Osteoarthritis and cartilage / OARS, Osteoarthritis Research Society. 2008;16(6):708–14.
Pan J, Wang B, Li W, et al. Elevated cross-talk between subchondral bone and cartilage in osteoarthritic joints. Bone. 2012;51(2):212–7.
Tami AE, Schaffler MB, Knothe Tate ML. Probing the tissue to subcellular level structure underlying bone’s molecular sieving function. Biorheology. 2003;40(6):577–90.
Knothe Tate ML. “Whither flows the fluid in bone?” an osteocyte’s perspective. J Biomech. 2003;36(10):1409–24.
Wang L, Fritton SP, Weinbaum S, et al. On bone adaptation due to venous stasis. J Biomech. 2003;36(10):1439–51.
Karsdal MA, Leeming DJ, Dam EB, et al. Should subchondral bone turnover be targeted when treating osteoarthritis? Osteoarthr Cartil. 2008;16(6):638–46.
Mapp PI, Avery PS, McWilliams DF, et al. Angiogenesis in two animal models of osteoarthritis. Osteoarthritis and cartilage / OARS, Osteoarthritis Research Society. 2008;16(1):61–9.
Walsh DA, Bonnet CS, Turner EL, et al. Angiogenesis in the synovium and at the osteochondral junction in osteoarthritis. Osteoarthritis and cartilage / OARS, Osteoarthritis Research Society. 2007;15(7):743–51.
Ashraf S, Walsh DA. Angiogenesis in osteoarthritis. Curr Opin Rheumatol. 2008;20(5):573–80.
Murata M, Yudoh K, Masuko K. The potential role of vascular endothelial growth factor (VEGF) in cartilage: how the angiogenic factor could be involved in the pathogenesis of osteoarthritis? Osteoarthritis and cartilage / OARS, Osteoarthritis Research Society. 2008;16(3):279–86.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Osteoarthritis
Rights and permissions
About this article
Cite this article
Aaron, R.K., Racine, J. & Dyke, J.P. Contribution of Circulatory Disturbances in Subchondral Bone to the Pathophysiology of Osteoarthritis. Curr Rheumatol Rep 19, 49 (2017). https://doi.org/10.1007/s11926-017-0660-x
Published:
DOI: https://doi.org/10.1007/s11926-017-0660-x