Abstract
Osteoporosis is defined as a reduction in bone mass and impairment of bone quality that lead to bone fragility and fracture risk. Bone quality includes a hierarchy of properties from macroscopic to nanoscale level. Several techniques have been developed in an attempt to measure these non-density properties. Densitometry, high-resolution images (radiography, CT scan), and MRI can measure the geometry and microarchitecture of bone. Tissue mineralization and composition can be assessed by use of microradiography, Fourier-transform infrared spectroscopy, or Raman microspectroscopy. Finite-element analysis is an image-based method that enables calculation of bone strength. More recently, microindentation has enabled direct estimation of bone material strength, measured in the cortical bone of the tibia. Most of these techniques are of limited use to clinics, although finite-element analysis and microindentation have high potential for clinical use and can enable more comprehensive and accurate evaluation of bone fragility and fracture susceptibility.


Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy. Osteoporosis prevention, diagnosis, and therapy. JAMA. 2001 Feb 14;285(6):785–95.
NIA workshop on aging and bone quality. Bethesda, September 3-4, 1992, Proceedings. Calcif Tissue Int. 1993;53 Suppl 1:S1–180.
Cummings SR, Bates D, Black DM. Clinical use of bone densitometry: Scientific review. JAMA. 2002;288(15):1889–97.
Maricic M, Gluck O. Densitometry in glucocorticoid-induced osteoporosis. J Clin Densitom. 2004;7(4):359–63.
Watts NB, Geusens P, Barton IP, Felsenberg D. Relationship between changes in BMD and nonvertebral fracture incidence associated with risedronate: Reduction in risk of nonvertebral fracture is not related to change in BMD. J Bone Miner Res. 2005;20(12):2097–104.
Ettinger B, Black DM, Mitlak BH, et al. Reduction of vertebral fracture risk in postmenopausal women with osteoporosis treated with raloxifene: Results from a 3-year randomized clinical trial. multiple outcomes of raloxifene evaluation (MORE) investigators. JAMA. 1999;282(7):637–45.
Delmas PD, Seeman E. Changes in bone mineral density explain little of the reduction in vertebral or nonvertebral fracture risk with anti-resorptive therapy. Bone. 2004;34(4):599–604.
Chapurlat RD, Palermo L, Ramsay P, Cummings SR. Risk of fracture among women who lose bone density during treatment with alendronate. The fracture intervention trial. Osteoporos Int. 2005;16(7):842–8.
Cummings SR, Bates D, Black DM. Clinical use of bone densitometry: Scientific review. JAMA. 2002;288(15):1889–97.
Black DM, Cummings SR, Karpf DB, et al. Randomized trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture intervention trial research group. Lancet. 1996;348(9041):1535–41.
Austin M, Yang YC, Vittinghoff E, et al. FREEDOM Trial. Relationship between bone mineral density changes with denosumab treatment and risk reduction for vertebral and nonvertebral fractures. J Bone Miner Res. 2012;27(3):687–93.
Felsenberg D, Boonen S. The bone quality framework: Determinants of bone strength and their interrelationships, and implications for osteoporosis management. Clin Ther. 2005;27(1):1–11.
Bouxsein ML, Seeman E. Quantifying the material and structural determinants of bone strength. Best Pract Res Clin Rheumatol. 2009;23(6):741–53.
Ahlborg HG, Johnell O, Turner C, et al. Bone loss and bone size after menopause. N Engl J Med. 2003;349(4):327–34.
Kleerekoper M, Villanueva AR, Stanciu J, et al. The role of three-dimensional trabecular microstructure in the pathogenesis of vertebral compression fractures. Calcif Tissue Int. 1985;37(6):594–7.
Slyfield CR, Tkachenko EV, Fischer SE, et al. Mechanical failure begins preferentially near resorption cavities in human vertebral cancellous bone under compression. Bone. 2012;50(6):1281–7.
Silva MJ, Gibson LJ. Modeling the mechanical behavior of vertebral trabecular bone: Effects of age-related changes in microstructure. Bone. 1997;21(2):191–9.
Weinstein RS, Hutson MS. Decreased trabecular width and increased trabecular spacing contribute to bone loss with aging. Bone. 1987;8(3):137–42.
Macdonald HM, Nishiyama KK, Kang J, et al. Age-related patterns of trabecular and cortical bone loss differ between sexes and skeletal sites: A population-based HR-pQCT study. J Bone Miner Res. 2011;26(1):50–62.
Bell KL, Loveridge N, Jordan GR, et al. A novel mechanism for induction of increased cortical porosity in cases of intracapsular hip fracture. Bone. 2000;27(2):297–304.
Ebacher V, Tang C, McKay H, et al. Strain redistribution and cracking behavior of human bone during bending. Bone. 2007;40(5):1265–75.
Koester KJ, Ager 3rd JW, Ritchie RO. The true toughness of human cortical bone measured with realistically short cracks. Nat Mater. 2008;7(8):672–7.
•• Zebaze RM, Ghasem-Zadeh A, Bohte A, et al. Intracortical remodelling and porosity in the distal radius and post-mortem femurs of women: A cross-sectional study. Lancet. 2010;375(9727):1729–36. Cortical bone is a key component of bone strength. The transition between cortical and trabecular compartments is difficult to establish due to the trabecularization of the inner side of the cortex.
Follet H, Boivin G, Rumelhart C, Meunier PJ. The degree of mineralization is a determinant of bone strength: A study on human calcanei. Bone. 2004;34(5):783–9.
Viguet-Carrin S, Farlay D, Bala Y, et al. An in vitro model to test the contribution of advanced glycation end products to bone biomechanical properties. Bone. 2008;42(1):139–49.
•• Saito M, Marumo K. Collagen cross-links as a determinant of bone quality: A possible explanation for bone fragility in aging, osteoporosis, and diabetes mellitus. Osteoporos Int. 2010;21(2):195–214. Collagen maturity is relevant for the mechanical properties of bone. Increased cross-linking is associated with older, less mechanically efficient bone.
Boivin G, Bala Y, Doublier A, et al. The role of mineralization and organic matrix in the microhardness of bone tissue from controls and osteoporotic patients. Bone. 2008;43(3):532–8.
Currey JD, Brear K, Zioupos P. The effects of ageing and changes in mineral content in degrading the toughness of human femora. J Biomech. 1996;29(2):257–60.
Burr DB, Forwood MR, Fyhrie DP, et al. Bone microdamage and skeletal fragility in osteoporotic and stress fractures. J Bone Miner Res. 1997;12(1):6–15.
Burr DB. Targeted and nontargeted remodeling. Bone. 2002;30(1):2–4.
• Allen MR, Burr DB. Bisphosphonate effects on bone turnover, microdamage, and mechanical properties: What we think we know and what we know that we don’t know. Bone. 2011;49(1):56–65. Excellent review on the potential deleterious effects of this group of drugs on bone mechanical properties.
Currey JD. The effects of drying and re-wetting on some mechanical properties of cortical bone. J Biomech. 1988;21(5):439–41.
Seeman E, Delmas PD. Bone quality–the material and structural basis of bone strength and fragility. N Engl J Med. 2006;354(21):2250–61.
Marcott C, Reeder RC, Paschalis EP, et al. Infrared microspectroscopic imaging of biomineralized tissues using a mercury-cadmium-telluride focal-plane array detector. Cell Mol Biol (Noisy-le-grand). 1998;44(1):109–15.
Gourion-Arsiquaud S, West PA, Boskey AL. Fourier transform-infrared microspectroscopy and microscopic imaging. Methods Mol Biol. 2008;455:293–303.
Tjhia CK, Odvina CV, Rao DS, et al. Mechanical property and tissue mineral density differences among severely suppressed bone turnover (SSBT) patients, osteoporotic patients, and normal subjects. Bone. 2011;49(6):1279–89.
• Chappard D, Basle MF, Legrand E, Audran M. New laboratory tools in the assessment of bone quality. Osteoporos Int. 2011;22(8):2225–40. Comprehensive review of the available methods for the assessment of the BMD-independent elements of bone.
Burket J, Gourion-Arsiquaud S, Havill LM, et al. Microstructure and nanomechanical properties in osteons relate to tissue and animal age. J Biomech. 2011;44(2):277–84.
• Danielson ME, Beck TJ, Karlamangla AS, et al. A comparison of DXA and CT based methods for estimating the strength of the femoral neck in post-menopausal women. Osteoporos Int. 2013;24(4):1379–88. Study of the two methods commonly used for the calculations in finite element analyses.
Thevenot J, Hirvasniemi J, Finnila M, et al. Trabecular homogeneity index derived from plain radiograph to evaluate bone quality. J Bone Miner Res. 2013
Diez-Perez A, Olmos JM, Nogues X, et al. Risk factors for prediction of inadequate response to antiresorptives. J Bone Miner Res. 2012;27(4):817–24.
Amin S, Kopperdhal DL, Melton 3rd LJ, et al. Association of hip strength estimates by finite-element analysis with fractures in women and men. J Bone Miner Res. 2011;26(7):1593–600.
Graeff C, Marin F, Petto H, et al. High resolution quantitative computed tomography-based assessment of trabecular microstructure and strength estimates by finite-element analysis of the spine, but not DXA, reflects vertebral fracture status in men with glucocorticoid-induced osteoporosis. Bone. 2013;52(2):568–77.
Srinivasan B, Kopperdahl DL, Amin S, et al. Relationship of femoral neck areal bone mineral density to volumetric bone mineral density, bone size, and femoral strength in men and women. Osteoporos Int. 2012;23(1):155–62.
•• Roux JP, Wegrzyn J, Arlot ME, et al. Contribution of trabecular and cortical components to biomechanical behavior of human vertebrae: An ex vivo study. J Bone Miner Res. 2010;25(2):356–61. Experimental research where the relative contribution of cortical and trabecular bone is studied. Fracture propensity is mainly driven by the cortical bone.
Zioupos P, Hansen U, Currey JD. Microcracking damage and the fracture process in relation to strain rate in human cortical bone tensile failure. J Biomech. 2008;41(14):2932–9.
Hvid I, Christensen P, Soondergaard J, Christensen PB, Larsen CG. Compressive strength of tibial cancellous bone. instron and osteopenetrometer measurements in an autopsy material. Acta Orthop Scand. 1983;54(6):819–25.
Imbeni V, Kruzic JJ, Marshall GW, et al. The dentin-enamel junction and the fracture of human teeth. Nat Mater. 2005;4(3):229–32.
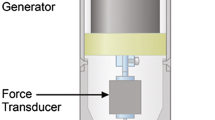
Hansma P, Turner P, Drake B, et al. The bone diagnostic instrument II: Indentation distance increase. Rev Sci Instrum. 2008;79(6):064303.
Hansma P, Yu H, Schultz D, et al. The tissue diagnostic instrument. Rev Sci Instrum. 2009;80(5):054303.
•• Diez-Perez A, Guerri R, Nogues X, et al. Microindentation for in vivo measurement of bone tissue mechanical properties in humans. J Bone Miner Res. 2010;25(8):1877–85. First publication on microindentation with the validation and basic methodology. The technique discriminates between fracture and non-fracture cases.
•• Guerri-Fernandez RC, Nogues X, Quesada Gomez JM, et al. Microindentation for in vivo measurement of bone tissue material properties in atypical femoral fracture patients and controls. J Bone Miner Res. 2013;28(1):162–8. Second publication in humans analyzing a paradigm of bone quality deterioration, atypical femoral fractures. In spite of preserved bone density, mechanical properties of bone tissue are deeply deteriorated.
Dall’Ara E, Schmidt R, Zysset P. Microindentation can discriminate between damaged and intact human bone tissue. Bone. 2012;50(4):925–9.
•• Gallant MA, Brown DM, Organ JM, et al. Reference-point indentation correlates with bone toughness assessed using whole-bone traditional mechanical testing. Bone. 2013;53(1):301–5. Animal study of microindentation correlating the obtained variables of the technique with the classical mechanical tests in the laboratory.
Fantner GE, Hassenkam T, Kindt JH, et al. Sacrificial bonds and hidden length dissipate energy as mineralized fibrils separate during bone fracture. Nat Mater. 2005;4(8):612–6.
Acknowledgments
Elisa Torres is recipient of a Jordi Gras research grant from the Parc de Salut Mar-IMIM. This work is sponsored in part by a grant from RETICEF, Instituto Carlos III, FEDER funds.
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
Adolfo Diez-Pérez has held stock and stock options in Active Life Scientific. Elisa Torres, Laia Vilaplana, and Roberto Güerri-Fernández declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with animal subjects. In addition, all procedures were followed in accordance with the ethical standards of the responsible committee on human experimentation and with the Helsinki Declaration of 1975, as revised in 2000 and 2008.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Osteoporosis and Metabolic Bone Disease
Rights and permissions
About this article
Cite this article
Torres-del-Pliego, E., Vilaplana, L., Güerri-Fernández, R. et al. Measuring Bone Quality. Curr Rheumatol Rep 15, 373 (2013). https://doi.org/10.1007/s11926-013-0373-8
Published:
DOI: https://doi.org/10.1007/s11926-013-0373-8