Abstract
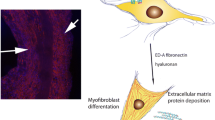
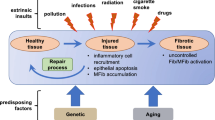
The concept of mesenchymal fibroblasts has evolved over the past two decades from a relatively inert structural cell type to a dynamic, pluripotent cell lineage controlling normal connective tissue formation, homeostasis, and repair and as principle players in pathogenic scarring and fibrosis. In wound healing and tissue repair, fibroblasts provide proinflammatory signals and synthesize interstitial collagens, fibronectins, and other matrix components to repair the damaged tissue. Fibroblasts can differentiate into the myofibroblast, a specialized contractile cell type responsible for wound closure, tissue contraction, and scarring. This article reviews our current understanding of the origins of mesenchymal cells and their role in excessive scarring and fibrogenesis and in the systemic fibrotic disease scleroderma.
Similar content being viewed by others
References and Recommended Reading
Tomasek JJ, Gabbiani G, Hinz B, et al.: Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat Rev Mol Cell Biol 2002, 3:349–363.
Korn JH: What’s wrong with the scleroderma fibroblast? Clin Exp Rheumatol 2004, 22:S64–S65.
Strehlow D, Korn JH: Biology of the scleroderma fibroblast. Curr Opin Rheumatol 1998, 10:572–578.
Jelaska A, Korn JH: Role of apoptosis and transforming growth factor beta1 in fibroblast selection and activation in systemic sclerosis. Arthritis Rheum 2000, 43:2230–2239.
Harper RA, Grove G: Human skin fibroblasts derived from papillary and reticular dermis: differences in growth potential in vitro. Science 1979, 204:526–527.
Bayreuther K, Rodemann HP, Hommel R, et al.: Human skin fibroblasts in vitro differentiate along a terminal cell lineage. Proc Natl Acad Sci U S A 1988, 85:5112–5116.
Chang HY, Sneddon JB, Alizadeh AA, et al.: Gene expression signature of fibroblast serum response predicts human cancer progression: similarities between tumors and wounds. PLoS Biol 2004, 2:E7.
Parsonage G, Filer AD, Haworth O, et al.: A stromal address code defined by fibroblasts. Trends Immunol 2005, 26:150–156.
Koumas L, Smith TJ, Feldon S, et al.: Thy-1 expression in human fibroblast subsets defines myofibroblastic or lipofibroblastic phenotypes. Am J Pathol 2003, 163:1291–1300.
Sanders YY, Kumbla P, Hagood JS: Enhanced myofibroblastic differentiation and survival in thy-1(-) lung fibroblasts. Am J Respir Cell Mol Biol 2006, Epub ahead of print.
Gabbiani G: The myofibroblast in wound healing and fibrocontractive diseases. J Pathol 2003, 200:500–503.
Serini G, Bochaton-Piallat ML, Ropraz P, et al.: The fibronectin domain ED-A is crucial for myofibroblastic phenotype induction by transforming growth factor-beta1. J Cell Biol 1998, 142:873–881.
Rajkumar Vs, Howell K, Csiszar K, et al.: Shared expression of phenotypic markers in systemic sclerosis indicates a convergence of pericytes and fibroblasts to a myofibroblast lineage in fibrosis. Arthritis Res Ther 2005, 7:R1113–1123.
Shephard P, Martin G, Smola-Hess S, et al.: Myofibroblast differentiation is induced in keratinocyte-fibroblast cocultures and is antagonistically regulated by endogenous transforming growth factor-beta and interleukin-1. Am J Pathol 2004, 164:2055–2066.
Denton CP, Black CM, Abraham DJ: Mechanisms and consequences of fibrosis in systemic sclerosis. Nat Clin Pract Rheumatol 2006, 2:134–144.
Kulozik M, Hogg A, Lankat-Buttgereit B, Krieg T: Colocalization of transforming growth factor beta 2 with alpha 1(I) procollagen mRNA in tissue sections of patients with systemic sclerosis. J Clin Invest 1990, 86:917–922.
Leask A, Abraham DJ: All in the CCN family: essential matricellular signaling modulators emerge from the bunker. J Cell Sci 2006, 119:4803–4810.
Rajkumar VS, Sundberg C, Abraham DJ, et al.: Activation of microvascular pericytes in autoimmune Raynaud’s phenomenon and systemic sclerosis. Arthritis Rheum 1999, 42:930–941.
Rajkumar VS, Shiwen X, Bostrom M, et al.: Platelet-derived growth factor-beta receptor activation is essential for fibroblast and pericyte recruitment during cutaneous wound healing. Am J Pathol 2006, 169:2254–2265.
Distler O, Pap T, Kowal-Bielecka O, et al.: Overexpression of monocyte chemoattractant protein 1 in systemic sclerosis: role of platelet-derived growth factor and effects on monocyte chemotaxis and collagen synthesis. Arthritis Rheum 2001, 44:2665–2678.
Yamamoto T, Hartmann K, Eckes B, Krieg T: Role of stem cell factor and monocyte chemoattractant protein-1 in the interaction between fibroblasts and mast cells in fibrosis. J Dermatol Sci 2001, 26:106–111.
Ong VH, Evans LA, Shiwen X, et al.: Monocyte chemoattractant protein 3 as a mediator of fibrosis: Overexpression in systemic sclerosis and the type 1 tight-skin mouse. Arthritis Rheum 2003, 48:1979–1991.
Kawaguchi Y, Hara M, Wright TM: Endogenous IL-1alpha from systemic sclerosis fibroblasts induces IL-6 and PDGF-A. J Clin Invest 1999, 103:1253–1260.
Higgins GC, Wu Y, Postlethwaite AE: Intracellular IL-1 receptor antagonist is elevated in human dermal fibroblasts that overexpress intracellular precursor IL-1 alpha. J Immunol 1999, 163:3969–3975.
Kodera T, McGaha TL, Phelps R, et al.: Disrupting the IL-4 gene rescues mice homozygous for the tight-skin mutation from embryonic death and diminishes TGF-beta production by fibroblasts. Proc Natl Acad Sci U S A 2002, 99:3800–3805.
Banning U, Krutmann J, Korholz D: The role of IL-4 and IL-12 in the regulation of collagen synthesis by fibroblasts. Immunol Invest 2006, 35:199–207.
Hasegawa M, Fujimoto M, Kikuchi K, Takehara K: Elevated serum levels of interleukin 4 (IL-4), IL-10, and IL-13 in patients with systemic sclerosis. J Rheumatol 1997, 24:328–332.
Gallucci RM, Lee EG, Tomasek JJ: IL-6 modulates alpha-smooth muscle actin expression in dermal fibroblasts from IL-6-deficient mice. J Invest Dermatol 2006, 126:561–568.
Fichtner-Feigl S, Strober W, Kawakami K, et al.: IL-13 signaling through the IL-13alpha2 receptor is involved in induction of TGF-beta1 production and fibrosis. Nat Med 2006, 12:99–106.
Kurasawa K, Hirose K, Sano H, et al.: Increased interleukin-17 production in patients with systemic sclerosis. Arthritis Rheum 2000, 43:2455–2463.
Mayes MD: Endothelin and endothelin receptor antagonists in systemic rheumatic disease. Arthritis Rheum 2003, 48:1190–1199.
Abraham D, Ponticos M, Nagase H: Connective tissue remodeling: cross-talk between endothelins and matrixmetalloproteinases. Curr Vasc Pharmacol 2005, 3:369–379.
Shi-wen X, Chen Y, Denton CP, et al.: Endothelin-1 promotes myofibroblast induction through the ETA receptor via a rac/phosphoinositide 3-kinase/Akt-dependent pathway and is essential for the enhanced contractile phenotype of fibrotic fibroblasts. Mol Biol Cell 2004, 15:2707–2719.
Minguell JJ, Erices A, Conget P: Mesenchymal stem cells. Exp Biol Med (Maywood) 2001, 226:507–520.
Chang HY, Chi JT, Dudoit S, et al.: Diversity, topographic differentiation, and positional memory in human fibroblasts. Proc Natl Acad Sci U S A 2002, 99:12877–12882.
Roufosse CA, Direkze NC, Otto WR, Wright NA: Circulating mesenchymal stem cells. Int J Biochem Cell Biol 2004, 36:585–597.
Caplan AI, Dennis JE: Mesenchymal stem cells as trophic mediators. J Cell Biochem 2006, 98:1076–1084.
Prockop DJ: Marrow stromal cells as stem cells for nonhematopoietic tissues. Science 1997, 276:71–74.
Fathke C, Wilson L, Hutter J, et al.: Contribution of bone marrow-derived cells to skin: collagen deposition and wound repair. Stem Cells 2004, 22:812–822.
Hashimoto N, Jin H, Liu T, et al.: Bone marrow-derived progenitor cells in pulmonary fibrosis. J Clin Invest 2004, 113:243–252.
Quan TE, Cowper S, Wu SP, et al.: Circulating fibrocytes: collagen-secreting cells of the peripheral blood. Int J Biochem Cell Biol 2004, 36:598–606.
Zhao Y, Glesne D, Huberman E: A human peripheral blood monocyte-derived subset acts as pluripotent stem cells. Proc Natl Acad Sci U S A 2003, 100:2426–2431.
Postlethwaite AE, Shigemitsu H, Kanangat S: Cellular origins of fibroblasts: possible implications for organ fibrosis in systemic sclerosis. Curr Opin Rheumatol 2004, 16:733–738.
Kalluri R, Neilson EG: Epithelial-mesenchymal transition and its implications for fibrosis. J Clin Invest 2003, 112:1776–1784.
Neilson EG: Mechanisms of disease: Fibroblasts—a new look at an old problem. Nat Clin Pract Nephrol 2006, 2:101–108.
Willis BC, Liebler JM, Luby-Phelps K, et al.: Induction of epithelial-mesenchymal transition in alveolar epithelial cells by transforming growth factor-beta1: potential role in idiopathic pulmonary fibrosis. Am J Pathol 2005, 166:1321–1332.
Strutz F, Zeisberg M, Ziyadeh FN, et al.: Role of basic fibroblast growth factor-2 in epithelial-mesenchymal transformation. Kidney Int 2002, 61:1714–1728.
Sims DE: Diversity within pericytes. Clin Exp Pharmacol Physiol 2000, 27:842–846.
Gerhardt H, Betsholtz C: Endothelial-pericyte interactions in angiogenesis. Cell Tissue Res 2003, 314:15–23.
Sundberg C, Ivarsson M, Gerdin B, Rubin K: Pericytes as collagen-producing cells in excessive dermal scarring. Lab Invest 1996, 74:452–466.
Eckes B, Zweers MC, Zhang ZG, et al.: Mechanical tension and integrin alpha 2 beta 1 regulate fibroblast functions. J Investig Dermatol Symp Proc 2006, 11:66–72.
Rinn JL, Bondre C, Gladstone HB, et al.: Anatomic demarcation by positional variation in fibroblast gene expression programs. PLoS Genet 2006, 2:e119.
Leask A, Abraham DJ: TGF-beta signaling and the fibrotic response. FASEB J 2004, 18:816–827.
White B: Immunopathogenesis of systemic sclerosis. Rheum Dis Clin North Am 1996, 22:695–708.
Heldin CH, Westermark B: Mechanism of action and in vivo role of platelet-derived growth factor. Physiol Rev 1999, 79:1283–1316.
Shi-wen X, Pennington D, Holmes A, et al.: Autocrine overexpression of CTGF maintains fibrosis: RDA analysis of fibrosis genes in systemic sclerosis. Exp Cell Res 2000, 259:213–224.
Leask A, Abraham DJ, Finlay DR, et al.: Dysregulation of transforming growth factor beta signaling in scleroderma: overexpression of endoglin in cutaneous scleroderma fibroblasts. Arthritis Rheum 2002, 46:1857–1865.
Whitfield ML, Finlay DR, Murray JI, et al.: Systemic and cell type-specific gene expression patterns in scleroderma skin. Proc Natl Acad Sci U S A 2003, 100:12319–12324.
Chen Y, Shi-wen X, van Beek J, et al.: Matrix contraction by dermal fibroblasts requires transforming growth factor-beta/activin-linked kinase 5, heparan sulfate-containing proteoglycans, and MEK/ERK: insights into pathological scarring in chronic fibrotic disease. Am J Pathol 2005, 167:1699–1711.
Gardner H, Shearstone JR, Bandaru R, et al.: Gene profiling of scleroderma skin reveals robust signatures of disease that are imperfectly reflected in the transcript profiles of explanted fibroblasts. Arthritis Rheum 2006, 54:1961–1973.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abraham, D.J., Eckes, B., Rajkumar, V. et al. New developments in fibroblast and myofibroblast biology: Implications for fibrosis and scleroderma. Curr Rheumatol Rep 9, 136–143 (2007). https://doi.org/10.1007/s11926-007-0008-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11926-007-0008-z