Abstract
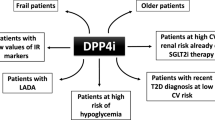
Sulfonylureas (SUs) are commonly used as add-on to metformin in treatment of type 2 diabetes in patients who are insufficiently controlled by metformin alone. They have good efficacy and have been shown to prevent microvascular complications. However, treatment with SUs is also associated with a high frequency of hypoglycemia, increased body weight, and a high risk of secondary failure. During recent years, dipeptidyl peptidase-4 (DPP-4) inhibitors have emerged as alternatives to SUs. They show similar efficacy as SUs but with lower risk of hypoglycemia, and reduction or no change in body weight, and if confirmed in humans, they may preserve islet function and thereby minimize the risk for secondary failure. Their limitation at present is the lack of long-term (>5 years) experience on durability and safety. Overall, therefore, the conclusion emerges that SUs are less desirable than DPP-4 inhibitors in management of hyperglycemia in type 2 diabetes.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
U.K. Prospective Diabetes Study (UKPDS) Group: Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 1998, 352: 837–853.
Gaede P, Vedel P, Larsen N, et al.: Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med 2003, 348:383–393.
Kahn SE: The relative contributions of insulin resistance and beta-cell dysfunction to the pathophysiology of type 2 diabetes. Diabetologia 2003, 46:3–19.
Dunning BE, Foley JE, Ahrén B: Alpha cell function in health and disease: influence of glucagon-like peptide-1. Diabetologia 2005, 48:1700–1713.
Butler AE, Janson J, Bonner-Weir S, Ritzel R, Rizza RA, Butler PC: Beta-cell deficit and increased beta-cell apoptosis in humans with type 2 diabetes. Diabetes 2003, 52:102–110
Ahrén B: Beta- and alpha-cell dysfunction in subjects developing impaired glucose tolerance: outcome of a 12-year prospective study in postmenopausal Caucasian women. Diabetes 2009, 58:726–731.
Adler AI, Shaw EJ, Stokes T, Ruiz F: Newer agents for blood glucose control in type 2 diabetes: summary of NICE guidance. Brit Med J 2009, 338:b1668
IDF Clinical guidelines Task Force: Global guideline for type 2 diabetes: recommendations for standard, comprehensive, and minimal care. Diabet Med. 2006,23:579–593.
Rodbard HW, Jellinger PS, Davidson JA, et al.: Statement by an American Association of Clinical Endocrinologists/American College of Endocrinology consensus panel on type 2 diabetes mellitus: an algorithm for glycemic control. Endocr Pract 2009, 15:540–559.
Nathan DM, Buse JB, Davidson MB, et al.: Medical management of hyperglycaemia in type 2 diabetes mellitus: a consensus algorithm for the initiation and adjustment of therapy. A consensus statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetologia 2009, 52:17–30.
Ahrén B: Emerging dipeptidyl peptidase-4 inhibitors for the treatment of diabetes. Expert Opin Emerg Drugs 2008, 13: 593–507.
Levine R: Sulfonylureas: background and development of the field. Diabetes Care 1984, 7: suppl 1:3–7.
Lupi R, Del Prato S: Beta-cell apoptosis in type 2 diabetes: quantitative and functional consequences. Diabetes Metab 2008, 34: suppl 2:S56–64.
Meinert CL, Knatterud GL, Prout TE, Klimt CR: A study of the effects of hypoglycemic agents on vascular complications in patients with adult-onset diabetes. II. Mortality results. Diabetes 1970, 19: suppl.: 789–830.
Gribble FM, Reimann F: Sulphonylurea action revisited: the post-cloning area. Diabetologia 2003, 46:875–891.
Bokvist K, Olsen HL, Hoy M, et al.: Characterisation of sulphonylurea and ATP-regulated K + channels in rat pancreatic A-cells. Pfl Arch 1999, 438:428–436.
Østergård T, Degn KB, Gall MA, et al.: The insulin secretagogues glibenclamide and repaglinide do not influence growth hormone secretion in humans but stimulate glucagon secretion during profound insulin deficiency. J Clin Endocrinol Metab 2004, 89:297–302.
Rendell M: The role of sulphonylureas in the management of type 2 diabetes mellitus. Drugs 2004, 64:1339–1358.
ADVANCE Collaborative Group: Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008, 358:2560–2572.
Tzoulaki I, Molokhia M, Curcin V, et al.: Risk of cardiovascular disease and all cause mortality among patients with type 2 diabetes prescribed oral antidiabetes drugs: retrospective cohort study using UK general practice research base. Brit Med J 2009, 339:b4731
Zhou M, He HJ, Suzuki R, et al.: Localization of sulfonylurea receptor subunits, SUR2A and SUR2B, in rat heart. J Histochem Cytochem 2007, 55:795–804.
Nordin C: The case for hypoglycaemia as a proarrhythmic event: basic and clinical evidence. Diabetologia 2010, 53:1552–1561.
Mannucci E, Monami M, Lamanna C, Gori F, Marchionni N: Prevention of cardiovascular disease through glycemic control in type 2 diabetes: a meta-analysis of randomized clinical trials. Nutr Metab Cardiovasc Dis 2009, 19:604–612
Belcher G, Lambert C, Goh KL, Edwards G, Valbuena M: Cardiovascular effects of treatment of type 2 diabetes with pioglitazone, metformin and gliclazide. Int J Clin Pract 2004, 58: 833–837.
Amiel SA, Dixon T, Mann R, Jameson K: Hypoglycaemia in type 2 diabetes. Diabet Med 25:245–254, 2008
Gerstein HC, Miller ME, Byington RP, et al.: Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008, 358:2545–2559.
Campbell IW: Comparing the actions of older and newer therapies on body weight: to what extent should these effects guide the selection of antidiabetic therapy? Int J Clin Pract 2010, 64:791–801.
Harrower AD: Comparison of efficacy, secondary failure rate, and complications of sulfonylureas. J Diabetes Complications 1994, 8:201–203.
Del Guerra S, Marselli L, Lupi R, et al.: Effects of prolonged in vitro exposure to sulphonylureas on the function and survival of human islets. J Diabetes Complications 2005, 19:60–64.
Drucker DJ, Nauck MA: The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet 2006, 368:1696–1705.
Ahrén B: Islet G-protein-coupled receptors as potential targets for treatment of type 2 diabetes. Nat Rev Drug Discov 2009, 8: 369–385.
Perfetti R, Hui H: The role of GLP-1 in the life and death of pancreatic beta cells. Horm Metab Res 2004, 36: 804–810.
Gutniak M, Ørskov C, Holst JJ, et al.: Antidiabetic effect of glucagon-like peptide-1 (7–36) amide in normal subjects and patients with diabetes mellitus. N Engl J Med 1992, 326: 1316–1322.
Ahrén B, Simonsson E, Larsson H, et al.: Inhibition of dipeptidyl peptidase IV improves metabolic control over a 4 week study period in type 2 diabetes. Diabetes Care 2002, 25: 869–875.
Ahrén B: Vildagliptin: an inhibitor of dipeptidyl peptidase-4 with antidiabetic properties. Exp Opin Invest Drugs 2006, 15:431–442.
Dhillon S: Sitagliptin: a review of its use in the management of type 2 diabetes mellitus. Drugs 2010, 70:489–512
Deacon CF, Holst JJ: Saxagliptin: a new dipeptidyl peptidase-4 inhibitor for the treatment of type 2 diabetes. Adv Ther 2009; 26: 488–499.
Deacon CF, Holst JJ: Linagliptin, a xanthine-based dipeptidyl peptidase-4 inhbitor with an unusual profile for the treatment of type 2 diabetes. Exp Opin Invest Drugs 210, 19: 133–140.
Pratley RE: Alogliptin: a new, highly selective dipeptidyl peptidase-4 inhibitor for the treatment of type 2 diabetes. Exp Opin Pharmacother 2009, 20:503–512.
• Ahren B, Schweizer A, Dejager S, et al. Vildagliptin enhances islet responsiveness to both hyper- and hypoglycemia in patients with type 2 diabetes. J Clin Endocrinol Metab 2009, 94:1236–1243. This study shows that the glucagon counter-regulation to hypoglycemia is preserved during treatment with vildagliptin.
Mu J, Woods J, Zhou YP, et al.: Chronic inhibition of dipeptidyl peptidase-4 with a sitagliptin analog preserves pancreatic beta-cell mass and function in a rodent model of type 2 diabetes. Diabetes 2006, 55:1695–1704
Ahrén B, Foley JE, Ferrannini E, et al.: Changes in prandial glucagon levels after a 2-year treatment with vildagliptin or glimepiride in patients with type 2 diabetes inadequately controlled with metformin monotherapy. Diabetes Care 2010, 33:730–732.
Nauck MA, Maininger G, Sheng D, et al.: Efficacy and safety of the dipeptidyl peptidase-4 inhibitor, sitagliptin, compared with the sulfonylurea, glipizide, in patients with type 2 diabetes inadequately controlled on metformin alone: a randomized, double-blind, non-inferiority trial. Diabetes Obes Metab 2007, 9:194–205.
Seck T, Nauck M, Sheng D, et al.: Safety and efficacy of treatment with sitagliptin or glipizide in patients with type 2 diabetes inadequately controlled on metformin: a 2-year study. Int J Clin Pract 2010, 64:562–576.
Filozof C, Gautier JF: A comparison of efficacy and safety of vildagliptin and gliclazide in combination with metformin in patients with type 2 diabetes inadequately controlled with metformin alone: a 52-week, randomized study. Diabetic Med 2010, 27:318–326.
Göke B, Gallwitz B, Eriksson J, et al.: Saxagliptin is non-inferior to glipizide in patients with type 2 diabetes inadequately controlled on metformin alone: a 52 week randomized clinical trial. Int J Clin Pract 2010, 64:1619–1631.
Rizzo M, Rizvi AA, Spinas GA, Rini GB, Berneis K: Glucose lowering and anti-atherogenic effects of incretin-based therapies: GLP1 analogues and DPP-4 inhibitors. Exp Opin Investig Drug 2009, 18:1495–1503
• Ferrannini E, Fonseca V, Zinman B, et al.: Fifty-two-week efficacy and safety of vildagliptin vs. glimepiride in patients with type 2 diabetes mellitus inadequately controlled on metformin monotherapy. Diabetes Obes Metab 2009, 11:157–166. This study demonstrates that vildagliptin and glimepiride have the same reduction in HbA 1c but with marked difference in that glimepiride is associated with much more hypoglycemic events and body weight gain.
Matthews DR, Dejager S, Ahrén B, et al.: Vildagliptin add-on to metformin produces similar efficacy and reduced hypoglycaemic risk compared with glimepiride, with no weight gain: results from a 2-year study. Diabetes Obes Metab 2010, 12:780–789.
Arechavelata R, Seck T, Chen Y, et al.: Efficacy and safety of treatment with sitagliptin or glimepiride in patients with type 2 diabetes inadequately controlled on metformin monotherapy: a randomized, double-blind, non-inferiority trial. Diabetes Obes Metab 2011, 13:160–168.
Disclosure
Conflicts of interest: B. Ahrén: acknowledges the Swedish Research Council (grant nr 6834), Region Skåne, Faculty of Medicine, Lund University, and Novartis AG for financial support in his own studies reported here. He also discloses receipt of honoraria for lectures from AstraZeneca, GlaxoSmithKline, Merck, Novartis, Novo Nordisk, Roche, Sanofi Aventis, and Servier, and receipt of research grants from AstraZeneca, Merck, and Novartis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahrén, B. Are Sulfonylureas Less Desirable Than DPP-4 Inhibitors as Add-on to Metformin in the Treatment of Type 2 Diabetes?. Curr Diab Rep 11, 83–90 (2011). https://doi.org/10.1007/s11892-011-0179-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11892-011-0179-x