Abstract
Background
Accurate concussion monitoring requires access to preinjury baseline data. This is particularly important in adolescent athletes who have a high risk of concussion and are prone to prolonged recovery. As Rugby Union is governed by similar laws for men and women, it is also an ideal population to rigorously examine the impact of biological sex on concussion symptoms.
Aims
To evaluate self-reported concussion symptoms at baseline in adolescent rugby union players, and examine if subtype-specific symptoms are affected by concussion history and biological sex.
Methods
Adolescent rugby union players aged 16–18 years were recruited during the 2022–2023 playing season. Participants completed a series of questionnaires covering post-concussion symptoms, concussion clinical profiles, anxiety, depression and fear avoidance behaviours. Independent variables of interest in analysis were biological sex and concussion history.
Results
149 participants (75% male) were included. 42% (63/149) reported at least one previous concussion (average time since concussion: 18.7 months, range 1–72). Adolescents with a concussion history reported significantly higher scores than those with no history, across two clinical profiles (ocular and sleep), concussion symptom severity, and depression, all based on medium effect sizes (SMD 0.3–0.5). Females had significantly higher scores across cognitive/fatigue, ocular and sleep clinical profiles, concussion symptoms, anxiety and depression, each with large effect sizes (SMD > 0.5).
Conclusions
Concussion history and sex are associated with higher baseline scores on specific concussion clinical profile, concussion symptom severity, and anxiety symptoms. These findings highlight the importance of considering baseline differences when interpreting post-injury clinical profile symptoms in adolescent rugby players. (Trial registration: ACTRN12622000931774).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sport-related concussion is a complex pathophysiological process affecting the brain, induced by traumatic biomechanical forces [1]. Adolescents may be at particularly high risk from concussion in contact and collision sports such as rugby union, ice hockey and American Football [1]. Prospective research indicates that adolescent rugby union has an incidence of 6.1 concussions per 1000 game hours, which is equivalent to 3 concussions per team per season [2]. Clinical recovery after concussion in adolescents is reported to occur within 30 days [1], but in general, recovery post-concussion is more unpredictable in paediatric patients compared to adults [3,4,5,6,7,8].
Current guidelines [1] suggest that youth athletes participating in high-risk sports should undergo a preseason baseline assessment. This can improve concussion recognition, and inform individualized treatment planning and decision-making on returning a player to training and competitive games. Normative baseline values are increasingly available for adult and collegiate athletes, but are lacking for adolescents, particularly in female sports [9]. It is currently unclear if baseline symptoms in adolescents are affected by concussion history [10,11,12,13,14]. Two large observational studies undertaken in high-school aged ice hockey [11] and American football players [13], consistently reported that athletes with history of multiple prior concussions, had a higher symptom burden at baseline (as quantified by the Post-Concussion Symptom Scale). In contrast, a recent study involving adolescent rugby union players found no significant differences in baseline symptom scores, between those with and without a history of concussion [14].
Baseline symptom reporting is also related to sex. A study of adolescent athletes, found that females were more likely to report baseline symptoms compared to males [12]. A multi-site study across concussion clinics in the United States also found adolescent females to be twice as likely to have a prolonged recovery compared to males, this may relate to biological differences in neck strength, hormonal factors, and psycho-social differences in symptom perception / likelihood of symptom reporting [15]. In a recent systematic review, younger age, female sex, and history of previous concussion were found to be key pre-injury risk factors for prolonged recovery after concussion [16]. Preseason baseline comparisons in Rugby Union players also show significant sex differences, with females reporting a higher number of symptoms and greater symptom severity [17, 18].
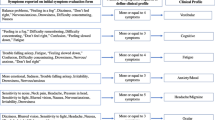
Most studies in this field have focused on baseline data on symptom burden and/or neurocognitive function. As concussions can affect multiple systems, baseline data should be multidimensional and include key clinical profiles such as: cognitive, fatigue, anxiety-mood, headache-migraine, ocular, and vestibular constructs [1, 19, 20]. There needs to be a importance of quantifying baseline levels of anxiety and depression [21], with studies suggesting that premorbid psychopathology directly interacts with post-concussion recovery [22].
The aim of this study was to evaluate the role of concussion history and biological sex on overall concussion symptom severity, concussion clinical profile symptoms, as well as anxiety, depression and fear avoidance symptoms at baseline among adolescent rugby union players. As Rugby Union is governed by similar laws for men and women, it is an ideal population to rigorously examine the impact of biological sex on concussion symptoms. It is hypothesized that players with a history of concussion and females would report higher symptoms across all these measures. It is anticipated that there would be an interactive effect for concussion history and biological sex (i.e., female) on these measures.
Methods
Study design
This was a cross-sectional study involving adolescents affiliated to Rugby Union playing schools or clubs in the Northern Ireland. The data presented are part of a larger longitudinal study that is tracking recovery after concussion in adolescent rugby players [23].
Participants
Male and female adolescent rugby union players were recruited from schools and clubs registered in Northern Ireland; some schools and clubs had been previously involved in the Rugby Injury Surveillance in Ulster Schools (RISUS) [2, 24, 25]. To be eligible for inclusion participants must have been aged between 16 and 18 years and injury free at the time of recruitment; they must also have been registered to play rugby union with a school or club for the 2022/23 season.
Procedures
Ethical approval was granted by the Ulster University Institutional Review Board (REC/14/0060). After providing informed written parental consent and participant assent, participants provided demographic information, injury history and completed the measures (listed below) in the following order: Post-Concussion Symptom Scale (PCSS), Clinical-Profile Screen (CP Screen), Paediatric Fear-Avoidance Behaviours after Traumatic Brain Injury Questionnaire (PFAB-TBI), Generalised Anxiety Disorder Assessment (GAD-7), and the Patient Health Questionnaire (PHQ-9). Baseline testing was completed prior to the start of the 2022–2023 playing season. Researchers were present whilst participants were completing the questionnaires.
Sports-related concussion definition followed the criteria set out by the Consensus Statement on Concussion in Sport; that is, a direct blow to the head, face, neck or elsewhere on the body with an impulsive force transmitted to the head resulting in one or more of the symptoms in one or more of the following clinical domains (which may or may not have involved loss of consciousness): somatic, cognitive, emotional/behavioural and sleep disturbance [26].
Measures
Demographics/medical history
This involved completing a demographic questionnaire (age, weight, and height), injury history (previous concussion [yes/no]), and time loss from sport (days) associated with a previous concussion.
Concussion symptoms
The Post-Concussion Symptom Scale is a self-reported measure of common concussion symptoms (e.g., headache, dizziness, nausea). The PCSS consists of 22 items rated on a 7-point Likert-type scale ranging from 0 (none) to 6 (severe). Total PCSS symptom severity scores range from 0–132, with higher scores indicating a greater severity of post-concussive symptoms. The scale has high internal consistency [27] in healthy and concussed adolescents, as well as construct and discriminate validity (concussed v non concussed athletes) has been established [27, 28].
Concussion clinical profiles
The CP Screen is a self-reported measure of symptoms representing five clinical profiles: cognitive/fatigue, vestibular, ocular, migraine, anxiety/mood; and two modifiers: sleep, neck (34). The CP Screen consists of 29 items rated on a 4-point Likert-type scale ranging from 0 (none) to 3 (severe). Item raw scores are totalled for each profile, with higher scores indicating greater severity of symptoms for that profile. The scale has high internal consistency, as well as construct and discriminate validity [29].
Fear/avoidance
The PFAB-TBI is a self-reported measure of fear and avoidance behaviours [30]. The PFAB-TBI includes 16-items related to fear and avoidance behaviour during the past month rated on a 4-point Likert-type scale ranging from 0 (strongly disagree) to 3 (strongly agree). Higher scores indicate greater fear avoidance behaviour (possible range 0–48) [31].
Anxiety
The GAD-7 questionnaire is a self-report screen of anxiety symptoms (e.g., nervousness, fear, worry). The GAD-7 includes 7-items examining presence and frequency of anxiety symptoms during the past two weeks rated on a 4-point Likert-type scale ranging from 0 (not at all) to 3 (nearly every day). Total scores range from 0 to 21, with higher scores indicating more severe anxiety symptoms. The GAD-7 has good reliability, as well as criterion, construct, factorial, and procedural validity [32].
Depression
The PHQ-9 is a self-report screen for depressive symptoms (e.g., hopelessness, sleep, fatigue, disinterest). The PHQ-9 consists of 9-items that assess the frequency and severity of depressive symptoms during the past two weeks rated on a scale from 0 (not at all) to 3 (nearly every day). Total scores range from 0–27 with higher scores indicating more frequent or severe symptoms. The PHQ-9 has been validated for primary care use [33].
Statistical analysis
All data were analysed using the Statistical Package of Social Sciences (SPSS) (Version 29; SPSS Inc). Outcomes were summarised using medians (range) for scale variables and frequencies and percentages for nominal variables. We used histograms to examine the frequency distributions of scale variables. All outcome variables were not normally distributed, therefore we used a non-parametric omnibus test (Kruskal-Wallis) followed by pairwise comparisons (Dunn-Bonferroni post hoc tests) across each level of the independent variables: male no history, female no history, male concussion history, female concussion history. As the distribution of data for each group had a similar shape (right skew), we focused on the median scores (rather than the mean ranks). For our primary analysis was used total scores from each questionnaire. We ran separate analyses on each CP Screen factor and modifier scores. The threshold for statistical significance was P < 0.01 for all tests, with Bonferroni adjustments used to reduce the risk of Type 1 error due multiple comparisons. We presented between group differences for concussion history and sex, using standardised mean differences (SMD) and 95% confidence intervals (99% CIs).
Sample size
The cross-sectional data presented in this study form part of a larger longitudinal investigation tracking recovery after concussion in adolescent rugby players. Therefore, the original sample size was determined based on our longitudinal data, assuming a 20% loss to follow-up throughout the entire study and a 10% prevalence of concussion during a single playing season [2, 24]. Complete details can be found in the published protocol [23]. For the current cross sectional analysis, we needed a minimum sample size of 65 participants per group to test our primary hypothesis (that players with a history of concussion would report higher symptoms). This was based on a medium effect size (SMD 0.5) when comparing independent groups (concussion history vs no history), and alpha and beta levels of 1% and 20% respectively.
Results
A total of 149 participants, from 10 rugby union teams or clubs, participated in the study (n = 113 male [75.8%], n = 36 female [24.2%]) aged 16 to 18 years (Mean age 17.2yrs ± 0.7). Males were taller and heavier (mean height 180.6 cm ± 6.4, mean weight 83.0 kg ± 15.5) than females (mean height 164.4 cm ± 6.5, mean weight 64.7 kg ± 6.7). Of the 149 participants, 63 (42.3%) reported a history of at least 1 concussion. The average time since last concussion was 18.7 months (range 1–72 months). A total of 47.8% of males (54/113) reported a history of at least 1 concussion, compared to 25.0% of females (9/36).
Table 1 shows the median (range) scores for each self-reported questionnaire, split by history of concussion and biological sex. Kruskal-Wallis H result supported a statistically significant difference in scores between the independent groups for PCSS (p < 0.01), CP Screen total score (p < 0.001), CP Screen ocular (p < 0.01) and sleep subscales (p < 0.001), PHQ (p < 0.001) and GAD-7 (p < 0.01). The highest median scores for PCSS (26/132), CP Screen (ocular and sleep subscales) and PHQ (6/27), were reported in females with a history of concussion. The boxplots below show the scores for PCSS (Fig. 1), Ocular (Fig. 2), CP Screen (Sleep) (Fig. 3) and PHQ (Fig. 4), split by concussion history and biological sex. The findings from Dunn Bonferroni post-hoc tests are also shown on each figure; the median scores in females with a history of concussion were significantly higher (adjusted p < 0.01) than males with no history for each of these outcomes. For clinical profiles for Sleep, females with a history of concussion also had significantly higher median symptoms (p < 0.01) compared to the males with a history of concussion (Fig. 3). There were no differences for other pairwise comparisons.
Figures 5 and 6 show the standardized effect magnitudes (SMD, 99% CIs) associated with concussion history and sex, across all self-reported outcomes. Figure 5 shows that scores were consistently higher in adolescents with a history of concussion compared to controls; the largest differences between groups (depicted by the filled circles) were seen in PCSS (SMD 0.50; 0.16 to 0.82), Ocular (SMD 0.39; 0.06 to 0.72) and PHQ (SMD 0.31; 0.02 to 0.64). Figure 6 shows that the average scores were higher in females across most outcomes, aside from the Neck subscale of CP Screen and PFAB. The largest differences between groups (depicted by filled circles) show that females had significantly higher scores (vs males) across 3 clinical profiles, Cognitive/Fatigue (SMD 0.60; 0.09 to 1.10), Ocular (SMD 0.67; 0.16 to 1.20) and Sleep (SMD 0.52; 0.02 to 1.00), and in depression (PHQ) (SMD 0.56; 0.18 to 0.94) and anxiety (GAD-7) (SMD 0.54; 0.15 to 0.92), all with medium effect sizes.
Discussion
Sport-related concussions are complex pathophysiological processes affecting the brain, with heterogenous clinical presentations. Accurate concussion monitoring requires access to preinjury baseline data, across multidimensional clinical profiles. There is no clear consensus on the moderating effect that demographic variables (eg. sex and concussion history) have on preseason/baseline data; as Rugby Union is governed by similar laws for men and women, it is an ideal population to rigorously examine this. The aim of this study was to examine self-reported symptoms in adolescent rugby players, across a range of clinical profiles at baseline. This study found concussion history and sex were associated with higher baseline scores for specific clinical profile symptoms. Adolescents with a concussion history reported higher ocular and sleep symptoms, total concussion symptom severity, and depression. Regarding sex, females reported higher cognitive/fatigue, ocular, and sleep clinical profile symptoms, as well as total concussion symptom severity, anxiety, and depression symptoms.
PCSS is one of the most used tools to assess and quantify the severity of concussion symptoms. Consistent with previous research undertaken in groups of adolescent footballers [11, 34], middle school [35] and high school athletes [10, 36], we found that adolescents with previous concussion demonstrated higher symptom scores (on PCSS), compared to those with no prior concussions. In a recent study involving adolescent rugby players, Cosgrave et al. recorded a similar prevalence of previous concussions at baseline (45% vs 42% in the current study), but found no significant difference in baseline symptoms. This divergence may relate to differences in outcome measures (SCAT vs PCSS) and demographics (they exclusively focused on males). We note that in the current study, the median baseline symptom score reported in males with a history of concussion was 7.5, which is largely comparable to previous data (where average scores ranged from 6 to 13) [10, 13, 35].
Separate analyses were run on each Clinical profile screen factor and modifier scores. One of the largest differences was evident in the ocular profile, with higher symptoms reported in athletes with a history of concussion. (SMD 0.39; 0.06 to 0.72). Others report light sensitivity [13] and visual problems [13] as the most commonly endorsed baseline symptoms, with one study finding differences in pupillary light reflex metrics when comparing adolescent rugby players with and without a history of concussion at baseline [37]. Baseline depression scores were higher in adolescents with a history of concussion, based on PHQ-9, but we did not report any differences in anxiety or fear avoidance scores. Earlier retrospective studies in adolescents (13–17 years) found a correlation between concussion history and self-reported depression [38], with previously concussed athletes 2.5 times more likely to report elevated scores (≥ 5) on the PHQ-9 at baseline. The relationship between variables relating to concussion history, baseline symptoms and depression may be complex. For example, it is unclear if the baseline depressive symptoms reported are a reaction to experiencing persistent post-concussion symptoms, or that depressive symptoms increase the likelihood of reporting persisting symptoms.
Statistically significant differences were found between males and females for ocular, cognitive/fatigue and sleep clinical profiles, as well as for depression and anxiety, based on moderate (SMD 0.3–0.5) to large (SMD > 0.5) effect sizes. There was an observed interaction (concussion history*sex) for PCSS, where scores were highest in females with a history of concussion (median scores of 26 points (range 1–57). This value exceeds baseline scores reported in a sample of female adolescents with a history of multiple concussions (18.1 ± 22.1) [39], and is comparable to those reported by adolescents in the early stages (within 7 days) post-concussion [40]. A large cross sectional study in rugby players (average age 19 years) found that sex effects baseline symptom scores, with females reporting significantly higher total SCAT symptoms (p < 0.001) than men [18]. The findings from this study align with previous research showing females report higher baseline symptoms on PCSS [27], ImPACT [41] and for symptoms relating to headache, sleep, vision, and reduced energy [42]. Others have found that females have higher symptom severity on individual clinical profiles of cognitive/fatigue, ocular and anxiety/mood compared with males [43]. These patterns may be due to females being more willing to reporting concussion-related symptoms [44]. Adult studies have also documented differences in concussion prognosis between males and females, with females more likely to present with lingering symptoms [43, 45,46,47,48].
The baseline differences recorded for ocular, cognitive/fatigue and sleep clinical profiles, between males and females, provide further evidence that that biological sex is an important covariate to consider for future search in this field. The largest interaction (concussion history*sex) was for the sleep clinical profile, whereby, females with a history of concussion reported significantly more baseline symptoms that males with no history of concussion and males with a history of concussion. This study did not find any interaction effects (concussion history*sex) for PHQ-9 and previous research is inconsistent regarding the influence of sex on depressive outcomes associated with concussion. One study found that among collegiate athletes, women have an 84% increased risk of reporting clinically significant depressive symptoms after sustaining a concussion compared to men [49]. Conversely, a study separate did not reveal any differences in the development of depressive symptoms between male and female athletes following a concussion [50].
Normative baseline data can provide accurate comparisons if individual baseline data are not available [51]. Although baseline data are accessible for adult and collegiate athletic populations, they are lacking for adolescents [9]. In accordance with previous research, this study found that few participants had baseline scores of 0 across the self-reported outcomes assessed [9, 11, 51]. Regardless of concussion history, participants reported sleep issues (PCSS and CP-Screen), anxiety issues (GAD-7 and CP-Screen), fatigue (PCSS, PHQ-9 and CP-Screen), nervousness (GAD-7), drowsiness (PCSS, PHQ-9 and CP-Screen), and difficulty concentrating (PCSS and CP-Screen). This aligns with previous reports [9, 11,12,13] suggesting that complete symptom resolution post-concussion may not be realistic for all adolescent athletes.
Limitations
This study had unbalanced comparison groups, particularly for sex, where more males were recruited than females into the study. This affects the generalizability of the findings to the broader population of females. In general, females are under-represented across all sports medicine research, and few have been included in adolescent concussion studies to date [1].
Females with a history of concussion were particularly underrepresented in the current study, and interaction affects (concussion history*sex) were observed for some outcomes, these findings should be interpreted cautiously until it is replicated using a larger, and more balanced representation of females and males.
Measures used in this study were all self-reported, which introduces recall, social desirability, and reference bias into the study. It can only be assumed that participants were honest and accurate in their reporting of symptoms but cannot confirm this assumption. Another limitation of the current study is a lack of temporal standardization across participants. Participants were recruited from a range of schools and clubs at times convenient to their training schedules, which impacted the ability to standardize the time of day that participants completed the study measures. Self-reported symptoms may be affected by diurnal variation, with some symptoms worsening over the course of a day, due to increasing levels of exertion, physical or mental activity [39, 52]. The majority of our data collection occurred in the early afternoon when the effects of diurnal variation are likely to be lower [52]. None the less, the variability in timing could have influenced the responses provided by participants.
Conclusion
Concussion history and sex each affect baseline symptom reporting in adolescent rugby players. Athletes with a history of concussion reported significantly higher concussion total symptom severity, ocular clinical profile symptoms as well as depression and anxiety symptoms compared to those with no history. Regarding sex, females reported higher ocular, cognitive/fatigue, and sleep clinical profiles symptoms. Females reported higher levels of depression and anxiety symptoms compared to males. The findings also support an interaction effect for sleep clinical profiles, with females with a history of concussion being more likely to report baseline symptoms than males with a history of concussion. Overall, these findings highlight the importance of considering baseline differences when interpreting post-injury clinical profile symptoms in adolescent rugby players.
Availability of data and materials
The data used and/or analysed during the current study are available from the corresponding author upon reasonable request.
References
Patricios JS, Schneider KJ, Dvorak J et al (2023) Consensus statement on concussion in sport: the 6th international conference on concussion in sport – Amsterdam, October 2022. Br J Sports Med 57:695–711. https://doi.org/10.1136/bjsports-2023-106898
Archbold HA, Rankin AT, Webb M et al (2017) RISUS study: rugby injury surveillance in ulster schools. Br J Sports Med 51:600–606. https://doi.org/10.1136/bjsports-2015-095491
Davis GA, Anderson V, Babl FE et al (2017) What is the difference in concussion management in children as compared with adults? A systematic review. Br J Sports Med 51:949–957. https://doi.org/10.1136/bjsports-2016-097415
Leung FT, Mendis MD, Franettovich Smith MM et al (2022) Sensorimotor system changes in adolescent rugby players post-concussion: A prospective investigation from the subacute period through to return-to-sport. Musculoskelet Sci Pract 57:102492. https://doi.org/10.1016/j.msksp.2021.102492
Manzanero S, Elkington LJ, Praet SF et al (2017) Post-concussion recovery in children and adolescents: A narrative review. J Concussion 1:2059700217726874. https://doi.org/10.1177/2059700217726874
McCrea M, Guskiewicz K, Randolph CBW et al (2013) Incidence, clinical course, and predictors of prolonged recovery time following sport-related concussion in high school and college athletes. J Int Neuropsychol Soc 19(1):22–33. https://doi.org/10.1017/S1355617712000872
Miller JH, Gill C, Kuhn EN et al (2016) Predictors of delayed recovery following pediatric sports-related concussion: a case-control study. J Neurosurg Pediatr 17:491–496. https://doi.org/10.3171/2015.8.PEDS14332
Thomas D, Coxe K, Li H et al (2018) Length of recovery from sports-related concussions in pediatric patients treated at concussion clinics. Clin J Sport Med 28:56–63. https://doi.org/10.1097/JSM.0000000000000413
Hunt AW, Paniccia M, Reed N et al (2016) Concussion-like symptoms in child and youth athletes at baseline: What Is “Typical”? J Athl Train 51:749–757. https://doi.org/10.4085/1062-6050-51.11.12
Mannix R, Iverson GL, Maxwell B et al (2014) Multiple prior concussions are associated with symptoms in high school athletes. Ann Clin Transl Neurol 1:433–438. https://doi.org/10.1002/acn3.70
Brooks BL, McKay CD, Mrazik M et al (2013) Subjective, but not objective, lingering effects of multiple past concussions in adolescents. J Neurotrauma 30:1469–1475. https://doi.org/10.1089/neu.2012.2720
Iverson GL, Silverberg ND, Mannix R et al (2017) Factors associated with concussion-like symptom reporting in high school athletes. JAMA Pediatr 169(12):1132–1140. https://doi.org/10.1001/jamapediatrics.2015.2374
Brooks BL, Mannix R, Maxwell B et al (2016) Multiple past concussions in high school football players: are there differences in cognitive functioning and symptom reporting? Am J Sports Med 44(12):3243–3251. https://doi.org/10.1177/0363546516655095
Cosgrave C, Fuller C, Kung S et al (2023) A comparison of clinical assessment with common diagnostic tools for monitoring concussion recovery in adolescent rugby union players. Phys Ther Sport 61:165–171. https://doi.org/10.1016/j.ptsp.2023.04.003
Baker JG, Leddy JJ, Darling SR et al (2016) Gender differences in recovery from sports-related concussion in adolescents. Clin Pediatr 55:771–775. https://doi.org/10.1177/0009922815606417
Wait TJ, Eck AG, Loose T et al (2023) Median time to return to sports after concussion is within 21 days in 80% of published studies. J Arthrosc Relat Surg 39:887–901. https://doi.org/10.1016/j.arthro.2022.11.029
Salmon DM, Chua J, Sullivan SJ et al (2021) Baseline concussion assessment performance of community-based senior rugby players: a cross-sectional study. Brain Inj 35:1433–1442. https://doi.org/10.1080/02699052.2021.1972452
Katherine JH, Cameron KL, Roach MH et al (2023) Baseline concussion assessment performance by sex in military service academy rugby players: findings from the CARE Consortium. BMJ Military Health Feb 17:e002358. https://doi.org/10.1136/military-2023-002358
Williams RM, Johnson RS, Snyder Valier AR et al (2021) Evaluating multiple domains of health in high school athletes with sport-related concussion. J Sport Rehabil 30:545–551. https://doi.org/10.1123/jsr.2019-0517
Pertab JL, Merkley TL, Cramond AJ et al (2018) Concussion and the autonomic nervous system: an introduction to the field and the results of a systematic review. NeuroRehabilitation 42(4):397–427. https://doi.org/10.3233/NRE-172298
Barlow KM (2016) Postconcussion syndrome: a review. J Child Neurol 31(1):57–67. https://doi.org/10.1177/0883073814543305
Polinder S, Cnossen MC, Real RGL et al (2018). Multidimensional approach to post-concussion symptoms in mild traumatic brain injury. https://doi.org/10.3389/fneur.2018.01113
McKee CS, Matthews M, Rankin A et al (2023) Multisystem recovery after sport-related concussion in adolescent rugby players: a prospective study protocol. BMJ Open 13:e073677. https://doi.org/10.1136/bmjopen-2023-073677
Archbold P, Rankin A, Webb M et al (2021) Injury patterns in U15 rugby players in Ulster Schools: A Rugby Injury Surveillance (RISUS) study. Transl Sports Med. https://doi.org/10.1002/tsm2.248
Matthews M, Johnston W, Bleakley C et al (2021) Concussion history and balance performance in adolescent rugby union players. Am J Sports Med 49:1348–1354. https://doi.org/10.1177/0363546521998709
McCrory P, Meeuwisse W, Dvořák J et al (2017) Consensus statement on concussion in sport-the 5(th) international conference on concussion in sport held in Berlin, October 2016. Br J Sports Med 51:838–847. https://doi.org/10.1136/bjsports-2017-097699
Lovell MR, Iverson GL, Collins MW et al (2006) Measurement of symptoms following sports-related concussion: reliability and normative data for the post-concussion scale. Appl Neuropsychol 13:166–174. https://doi.org/10.1207/s15324826an1303_4
Riegler KE, Guty ET, Arnett PA (2019) Validity of the ImPACT Post-Concussion Symptom Scale (PCSS) affective symptom cluster as a screener for depression in collegiate athletes. Archives of clinical neuropsychology : the official journal of the National Academy of Neuropsychologists 34:563–574. https://doi.org/10.1093/arclin/acy081
Kontos A, Trbovich A, Sandel N et al (2019) Sport-related concussion clinical profiles: clinical characteristics, targeted treatments, and preliminary evidence. Curr Sports Med Rep 18:82–92. https://doi.org/10.1249/JSR.0000000000000573
Snell D, Siegert R, Debert C et al (2020) Evaluation of the fear avoidance behavior after traumatic brain injury questionnaire. J Neurotrauma. https://doi.org/10.1089/neu.2019.6729
Cairncross M, Brooks BL, Virani S et al (2021) Fear avoidance behavior in youth with poor recovery from concussion: measurement properties and correlates of a new scale. Child neuropsychology : a journal on normal and abnormal development in childhood and adolescence 27:911–921. https://doi.org/10.1080/09297049.2021.1908533
Spitzer RL, Kroenke K, Williams JBW et al (2006) A brief measure for assessing Generalized Anxiety Disorder: The GAD-7. Arch Intern Med 166:1092–1097. https://doi.org/10.1001/archinte.166.10.1092
Cameron IM, Crawford JR, Lawton K et al (2008) Psychometric comparison of PHQ-9 and HADS for measuring depression severity in primary care. The British journal of general practice : the journal of the Royal College of General Practitioners 58:32–36. https://doi.org/10.3399/bjgp08X263794
Brooks MA, Peterson K, Biese K et al (2016) Concussion increases odds of sustaining a lower extremity musculoskeletal injury after return to play among collegiate athletes. Am J Sports Med 44:742–747. https://doi.org/10.1177/0363546515622387
Iverson GL, Berkner PD, Zafonte R et al (2022) Preseason symptom reporting and cognition in middle school athletes with past concussions. Int J Sports Med 43(6):553–560. https://doi.org/10.1055/a-1538-0075
Schatz P (2009) Long-term test-retest reliability of baseline cognitive assessments using ImPACT. Am J Sports Med 38:47–53. https://doi.org/10.1177/0363546509343805
McKee C, Matthews M, Rankin A et al (2024) The role of concussion history and biological sex on pupillary light reflex metrics in adolescent rugby players: a cross-sectional study. Sports (Basel) 12(2):56. https://doi.org/10.3390/sports12020056
Chrisman SPD, Richardson LP (2014) Prevalence of diagnosed depression in adolescents with history of concussion. J Adolesc Health 54:582–586. https://doi.org/10.1016/j.jadohealth.2013.10.006
Landre N, Poppe CJ, Davis N et al (2006) Cognitive functioning and postconcussive symptoms in trauma patients with and without mild TBI. Arch Clin Neuropsychol 21(4):255–273. https://doi.org/10.1016/j.acn.2005.12.007
Eagle SR, Womble MN, Elbin RJ et al (2020) Concussion symptom cutoffs for identification and prognosis of sports-related concussion: role of time since injury. Am J Sports Med 48(10):2544–2551. https://doi.org/10.1177/0363546520937291
McKay CD, Brooks BL, Mrazik M et al (2014) Psychometric properties and reference values for the ImPACT neurocognitive test battery in a sample of elite youth ice hockey players. Arch Clin Neuropsychol 29(2):141–151. https://doi.org/10.1093/arclin/act116
Brown DA, Elsass JF, Miller AJ et al (2015) Differences in symptom reporting between males and females at baseline and after a sports-related concussion: a systematic review and meta-analysis. Sports Med 45(7):1027–1040. https://doi.org/10.1007/s40279-015-0335-6
Stephenson K, Womble MN, Frascoia C et al (2022) Sex differences on the concussion clinical profiles screening in adolescents with sport-related concussion. J Athl Train 58:65–70. https://doi.org/10.4085/1062-6050-0670.21
Pieroth EM, Wicklund A (2023) Concussion in female athletes. Oper Tech Sports Med 31:151026. https://doi.org/10.1016/j.otsm.2023.151026
Mollayeva T, El-Khechen-Richandi G, Colantonio A (2018) Sex & gender considerations in concussion research. Concussion 18;3(1):CNC51. https://doi.org/10.2217/cnc-2017-0015
Covassin T, Moran R, Elbin RJ (2016) Sex differences in reported concussion injury rates and time loss from participation: an update of the national collegiate athletic association injury surveillance program from 2004–2005 through 2008–2009. J Athl Train 51:189–194. https://doi.org/10.4085/1062-6050-51.3.05
Covassin T, Swanik CB, Sachs ML (2003) Sex differences and the incidence of concussions among collegiate athletes. J Athl Train 38:238–244
Balestrini C, Moir M, Abbott K et al (2019) Autonomic dysregulation in adolescent concussion is sex- and posture-dependent. Clin J Sport Med 31:257–265. https://doi.org/10.1097/JSM.0000000000000734
Wolanin A, Hong E, Marks D et al (2016). Prevalence of clinically elevated depressive symptoms in college athletes and differences by gender and sport. https://doi.org/10.1136/bjsports-2015-095756
Kontos AP, Elbin RJ, Newcomer Appaneal R et al (2013) A comparison of coping responses among high school and college athletes with concussion, orthopedic injuries, and healthy controls. Res Sports Med 21(4):367–379. https://doi.org/10.1080/15438627.2013.825801
Schmidt JD, Register-Mihalik JK, Kerr ZY et al (2012) Identifying Impairments after concussion: normative data versus individualized baselines. Med Sci Sports Exerc 44(9):1621–1628. https://doi.org/10.1249/mss.0b013e318258a9fb
Lewandowski L, Rieger B, Smyth J et al (2009) Measuring post-concussion symptoms in adolescents: feasibility of ecological momentary assessment. Arch Clin Neuropsychol 24:791–796. https://doi.org/10.1093/arclin/acp087
Funding
This research was conducted during a PhD funded by the Department for the Economy (DfE). The funding source was not influential in the design or writing of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
McKee, C., Matthews, M., Kontos, A.P. et al. The role of concussion history and biological sex on baseline concussion clinical profile symptoms in adolescent rugby players. Ir J Med Sci 193, 2061–2069 (2024). https://doi.org/10.1007/s11845-024-03677-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-024-03677-7