Abstract
There is an urgent need to develop sustainable and environmentally benign integrated pest management (IPM) strategies for arable crops. The enhancement and manipulation of naturally occurring populations of the natural enemies of crop pests through habitat management for ‘conservation biological control’, as well as habitat management to manipulate populations of the pests themselves, have the potential to become major components of successful IPM strategies. We review the studies that have contributed to our current understanding of how the crop margin, local landscape, and regional landscape can influence pollen beetle Brassicogethes aeneus (syn. Meligethes aeneus) (Coleoptera: Nitidulidae) abundance and damage to oilseed rape crops (Brassica napus), and the efficacy of their natural enemies. We also discuss how habitat management across these multiple scales may improve pollen beetle control, reducing the need for insecticide use and contributing towards sustainable production of this important crop which is grown on increasing areas for both food and fuel.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Throughout Europe, the pollen beetle Brassicogethes aeneus F. (syn. Meligethes aeneus) (Coleoptera: Nitidulidae) is a major pest of oilseed rape Brassica napus L. (Brassicaceae) (OSR) (Williams 2010). The crop is an increasingly important source of vegetable oil and biofuel feedstock, with EU production rising from 19.0 M tonnes in 2008 to 21.7 M tonnes in 2015 (Eurostat 2016). Seed yield losses caused by pollen beetles can be significant, particularly in spring-sown crops (Hansen 2004) although widespread losses to winter-sown crops have been reported (Free and Williams 1978a; Zlof 2008). The adult beetles emerge in early spring from their overwintering habitats in wood- and grassland (Rusch et al. 2012a), and feed on pollen from a range of spring flowers, before seeking brassicaceous plants for feeding and oviposition (Free and Williams 1978b). Feeding damage of adults, and in some cases larvae, to flower buds can lead to abscission and subsequent podless stalks (Nilsson 1988). Both visual and olfactory cues are used for host plant location (Williams and Cook 2010), and the beetles colonise OSR crops at or around the green bud stage (Cook et al. 2006; Williams 2010). After eggs are laid in the buds, the developing larvae feed for around 2 weeks before pupation within the soil. New generation adults emerge in summer and again feed on pollen from plants of various families before overwintering (Williams and Free 1978; Ouvrard et al. 2016).
Over the last 40 years, heavy use of pyrethroid insecticides, often applied prophylactically, has led to the increasing occurrence of insecticide resistant pollen beetle populations (Thieme et al. 2010; Zimmer et al. 2014), and hence there is an urgent need to reduce insecticide use and develop more sustainable integrated pest management (IPM) practices for this pest. IPM is defined as the ‘careful consideration of all available plant protection methods and subsequent integration of appropriate measures that discourage the development of populations of harmful organisms and keep the use of plant protection products and other forms of intervention to levels that are economically and ecologically justified and reduce or minimise risks to human health and the environment. IPM emphasises the growth of a healthy crop with the least possible disruption to agroecosystems and encourages natural pest control mechanisms’ [European Union Framework Directive on the Sustainable Use of Pesticides (Directive 2009/128/EC)]. Several tactics for IPM strategies of pollen beetles in OSR are under development. These include efforts to reduce prophylactic insecticide applications through improved pollen beetle forecasting (Johnen and von Richthofen 2013; Ferguson et al. 2016), and monitoring (Cook et al. 2013; Skellern et al. 2017), and determination of more accurate control thresholds (AHDB-HGCA 2012; Ellis and Berry 2012; Ramsden et al. 2017). Other strategies aim to reduce pest numbers to below economic threshold levels; these include the use of plant- or insect-derived semiochemicals or ‘signal chemicals’, enabling behavioural manipulation of pests or their natural enemies (Cook et al. 2007a; Mauchline et al. 2008, 2013; Pavela 2011; reviewed by Mauchline et al. in press), botanical insecticides or other natural products such as silicate dusts as alternatives to synthetic insecticides (Pavela 2011; Daniel et al. 2013; Dorn et al. 2014), modification of crop management practices (Valantin-Morison et al. 2007; Veromann et al. 2009, 2013; reviewed by Skellern and Cook in press), breeding for OSR resistance to the pest (reviewed by Hervé and Cortesero 2016; Hervé 2017), and inundative biological control via the application or release of natural enemies such as entomopathogenic fungi (e.g. Hokkanen 1993; Husberg and Hokkanen 2001; reviewed by Hokkanen and Menzler-Hokkanen 2017) or nematodes (Menzler-Hokkanen and Hokkanen 2005).
Research into the use of generalist predators and specialist parasitoids of the immature stages of pollen beetles (see Table 1) in IPM strategies has focussed on conservation biological control (CBC), rather than the release of imported exotic or mass-reared natural enemies for biocontrol (Williams 2010). CBC involves the manipulation of the environment (i.e. the habitat) of natural enemies to enhance their survival and/or physiological performance resulting in enhanced effectiveness (Barbosa 1998). It is ‘based on the premise that countering habitat loss and environmental disturbance associated with intensive crop production will conserve natural enemies, thus contributing to pest suppression’ (Begg et al. 2017). CBC practices are therefore central to delivering pest management ecosystem services within IPM strategies.
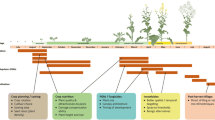
Management of the crop itself can contribute to CBC of OSR crop pests, including pollen beetles (reviewed by Rusch et al. 2010; Skellern and Cook in press). However, the crop margin (crop scale), local landscape [the field surroundings up to a range of a few thousand metres; at the scale of the farm unit or several adjacent farm units suitable to the dispersal abilities of the pest (e.g. Juhel et al. 2017)], and regional landscape (large land tracts of indefinite extent, but over scales of tens or hundreds of kilometres, that are sufficiently homogeneous in physical geography or ecology to be viewed as an entity distinct from other adjacent regions) are also known to affect OSR pest abundance and damage, as well as natural enemy efficacy (e.g. Thies and Tscharntke 1999; Buchi 2002; Cook et al. 2004; Rusch et al. 2010, 2011, 2013b). Therefore, better informed management of habitats over these multiple scales has great potential for improving pollen beetle control while diminishing the need for insecticide use. We review the studies that have contributed to our current understanding of how habitat management in non-cropped areas (‘off-crop’) may improve pollen beetle control. We first consider management of the crop margin, in terms of the development of trap cropping strategies, where plant stands are grown to attract pest insects to protect target crops from attack (Hokkanen 1991), and in terms of the potential of flower-rich field margins to enhance natural pest control. We then consider larger-scale landscape effects on pollen beetles and their natural enemies, and their implications for habitat management strategies. These are summarised in Table 2.
Habitat management of the crop margin
Trap cropping to reduce pollen beetle infestation of oilseed rape
Trap crops are ‘plant stands that are, per se or via manipulation, deployed to attract, divert, intercept, and/or retain targeted insects or the pathogens they vector in order to reduce damage to the main crop’ (Shelton and Badenes-Perez 2006). Pollen beetles show clear host plant preferences that can be exploited in trap cropping strategies (Cook et al. 2006), which aim to concentrate the pest on attractive trap crop plants while detracting it from the target OSR at its damage-susceptible stage (Hokkanen 1991); i.e. at the green-yellow bud stage (Williams 2010). Trap cropping has potential for management of pollen beetles in OSR, especially as the ‘pull’ element in push–pull strategies which combine a trap crop or other attractant or arrestant stimulus outside the crop and a deterrent or repellent near or within the crop, to divert pests, reducing their populations on the target crop (Cook et al. 2007b; Eigenbrode et al. 2016).
Several species of the Brassicaceae appear more attractive to pollen beetles than spring-sown OSR, including turnip rape Brassica rapa (Hokkanen 1989, Hokkanen et al. 1986; Buchi 1990; Cook et al. 2004, 2006, 2007c), yellow mustard Brassica juncea (Kaasik et al. 2014b), black mustard Brassica nigra (Veromann et al. 2012; Kaasik et al. 2014a), and white mustard Sinapis alba (Kaasik et al. 2014b). However, few studies have tested the possibility that trap crops can reduce populations in OSR crops in practice. Hokkanen et al. (1986) and Hokkanen (1989) tested turnip rape trap crops comprising 1–15% of the main crop, placed in the middle or surrounding spring OSR fields. Areas of 10–15% reduced pollen beetle populations in the OSR crop and reduced the need for insecticide applications by 50–85%. Simulations using a spatially explicit individual-based model indicate that a perimeter trap crop would be the most appropriate arrangement (Potting et al. 2005). Cook et al. (2004) showed that spring OSR plots with a perimeter trap crop comprising c. 10% of the crop area had significantly reduced pollen beetle populations compared with control plots; populations were maintained below threshold levels and bud damage was significantly reduced. The turnip rape developed more quickly than the OSR and flowered c. 2 weeks before OSR; early flowering was also mentioned as essential to function by Hokkanen et al. (1986) and growth stage shown to be a critical mechanism of the success of turnip rape as a trap crop in later studies (Cook et al. 2007c). Buchi (1990) attributed the attraction of pollen beetles to turnip rape to the yellow-green colour of the leaves which may be more attractive than the blue-green colour of OSR. Studies on the colour responses of pollen beetles support this theory (Döring et al. 2012). Host plant odour has also been studied and the increased amounts of phenylacetaldehyde and indole in turnip rape also play a role in its success (Cook et al. 2007c).
While early studies focussed on spring OSR crops, later research investigated transfer to winter-sown systems, using a forage rape × forage turnip hybrid ‘Pasja’ (Cook et al. 2013) or a forage turnip × Chinese cabbage hybrid ‘Perko’ (Čuljak et al. 2016). Both studies tested the strategy on a field scale, showing that trap crop plants were significantly more infested by pollen beetles than OSR plants in the borders of control fields (Cook et al. 2013) or in field centres (Čuljak et al. 2016). While Čuljak et al. (2016) showed that trap crop strips, representing 15–25% of the total crop area, were able to maintain within-crop pollen beetle infestation at below threshold levels for 97.5% of the crop area, Cook et al. (2013) found the effect of the trap crop inconsistent between sites and years. However, in replicates/years where the trap crop was ineffective, flowering of the trap crop and OSR occurred only c. 1 week apart, and brassicas flowering 2–3 weeks earlier than the main crop are needed for the strategy to be reliable (Cook et al. 2013). Spraying the trap crop had no significant impact on populations in the main crop (Cook et al. 2013). For a conventionally managed crop, a cost:benefit analysis indicated that trap cropping is not cost effective, with a return of c. £400/ha compared with c. £450 if the crop needs to be sprayed and c. £480/ha if the crop receives no insecticide. Currently the strategy is only likely to be economically useful to organic growers who can gain a significant premium on seed grown in this way (Cook et al. 2013). Food-grade cultivars of turnip rape seeds can be harvested for oil as with OSR; however, it is lower-yielding, and tends to ripen faster, and thus requires harvesting earlier than OSR drilled at the same time if seed losses are to be avoided. If early flowering cultivars of OSR could be developed with the same attractive properties as turnip rape, then the strategy may be more cost effective (Cook et al. 2013).
The possibility of using trap crops to trap out pollen beetles as they move from winter to spring-sown OSR crops or other flowering resources before hibernation has been suggested (Veromann et al. 2012). Fodder Radish, Raphanus sativus, was more attractive than OSR in the flowering stages and was acceptable for oviposition but larval development on the plant has shown to be poor, leading to the suggestion that it may also have potential as a ‘dead-end’ trap crop (Shelton and Nault 2004) for pollen beetles (Veromann et al. 2012, 2014).
The effects of trap crops on general predators have not, to our knowledge, been reported and effects on parasitoids and parasitism of pollen beetle larvae in OSR have been little studied. There is some evidence of earlier parasitoid attraction to OSR crops when trap crops are present than in their absence (Defra 2010) and Vinatier et al. (2012) produced models suggesting that trap crops improved biocontrol of pollen beetles in OSR. As well as being planted as borders/strips to the crop, the trap plants could form part of field margin mixtures as part of conservation biocontrol measures for the parasitoids of pollen beetles and this is reviewed below.
The potential of flower-rich field margins to enhance natural pest control in oilseed rape
Field margins and other semi-natural habitats represent environments of relative stability compared with cropped areas, and can provide natural enemies with a source of alternative hosts and prey, overwintering habitat, refuges from biotic and abiotic conditions including tillage as well as floral resources (e.g. Sotherton 1984; Pickett and Bugg 1998; Thomas et al. 1991; Denys and Tscharntke 2002; reviewed by Gillespie et al. 2016). Provision of floral resources can promote the development and activity of populations of predators (e.g. Robinson et al. 2008; Tschumi et al. 2016; Hatt et al. 2017) and parasitoids (e.g. Baggen and Gurr 1998; Rebek et al. 2006). Access to nutrition provided by pollen and nectar has been shown to benefit natural enemies, improving their efficacy through increasing longevity and fecundity (reviewed by Lu et al. 2014; Gillespie et al. 2016).
There are at least nine species of hymenopteran parasitoids which attack pollen beetle (Nilsson 2003; Ulber et al. 2010), with six being of importance for biocontrol purposes (Table 1) exerting parasitism levels in the range of 25–50% (reviewed by Ulber et al. 2010). For these parasitoids, as their hosts specialise on plants of the Brassicaceae for reproduction, brassicaceous plants represent important breeding sites (Nilsson 2003; Ulber et al. 2010). Field margin mixtures designed with conservation biocontrol of OSR pests in mind are likely to perform well if they contain appropriate brassica species which could support pollen beetle parasitoids by acting as natural ‘banker plants’ (see Frank 2010), i.e. by providing natural infestations of host larvae for reproduction (Skellern et al. submitted; Defra 2013). As the univoltine parasitoids of pollen beetles overwinter in the soil in which the OSR crop was grown, emerging the following spring into the following crop in the rotation (often wheat), individuals developing on these plants and pupating in field margin soil would be protected from the insecticide- and tillage-related mortality experienced within-field (Nilsson 2010). Inclusion of these plants in field margins, as well as performing a trap cropping function, may also help to sustain parasitoid populations throughout the arable rotation, particularly when OSR is absent. This may be particularly relevant in landscapes where the spatial separation of large blocks of OSR fields forces the parasitoids, which have weaker dispersal abilities than their hosts (Thies and Tscharntke 2010) to migrate large distances from their emergence sites. Brassicas under consideration for field margin inclusion, however, should be carefully chosen to favour production of the parasitoids, and not that of their injurious hosts (see Gurr et al. 2004).
Studies assessing the influence of field margins on the biological control of pollen beetles in adjacent OSR crops have shown positive effects. Thies and Tscharntke (1999) observed that old field margin strips alongside winter OSR increased larval mortality through parasitism, and that larger, old fallows showed a more pronounced effect. Buchi (2002) showed that pollen beetle larval parasitism rates were significantly higher in OSR fields next to flower-rich margins than in those next to extensively managed meadows. Hatt et al. (in press) showed that the density of pollen beetle parasitoids caught in yellow traps within wildflower strips were related to floral traits not host abundance, but the relationship with adults, not the larval hosts was tested and parasitism was not assessed. In both studies where parasitism was assessed, better provision (quality and/or quantity) of floral resources from the field margins may have enhanced parasitoid activity in the crop. Indeed, Rusch et al. (2013a) showed that the parasitoid Tersilochus heterocerus (Hymenoptera: Ichneumonidae) had already consumed high amounts of sugar by the time they appeared in OSR fields, indicating their acquisition of non-OSR floral resources, and Jönsson and Anderson (2007) showed that starved Phradis morionellus (Hymenoptera: Ichneumonidae) parasitoids were less responsive than fed individuals to host-infested OSR racemes, perhaps because the starved individuals were in food-seeking rather than host-seeking mode.
Several studies have examined pollen beetle host plant preferences, performance, and parasitism rates on a range of species of the Brassicaceae, providing information that is relevant to a consideration of the utility of these as field margin banker plants. Brassicaceous plants vary in their acceptability for pollen beetle oviposition and feeding. While the beetles are a particular pest of OSR and turnip rape (Bromand 1990), other species accepted for oviposition and feeding include the mustards B. juncea and B. nigra and Abyssinian cabbage B. carinata (Ekbom and Borg 1996). White mustard, S. alba, and crambe, Crambe abyssinica, are accepted for feeding but have low acceptability for oviposition (Ekbom and Borg 1996). Winter cress Barbarea vulgaris and land cress B. verna have low acceptability for both oviposition and feeding (Borjesdotter 2000), and pollen beetle has been shown not to feed on candytuft, Iberis amara (Bartlet et al. 2004). Rocket, Eruca sativa, is relatively unattractive to the beetle (Ekbom 1998) and shows low acceptability for oviposition (Kaasik et al. 2014a).
Of the brassicaceous plants exploited by pollen beetles, Brassica nigra has been proposed as a potential ‘banker plant’ (Kaasik et al. 2014a) as it was particularly well utilised by T. heterocerus. Other studies, however, suggest that members of the brassicaceae which are lower quality host plants for the pest may also hold potential. Pollen beetles exhibit reduced egg production (Hopkins and Ekbom 1999) and egg size (Ekbom and Popov 2004) on less acceptable host plants (S. alba) vs. those of high quality (B. napus). Larvae reared on S. alba exhibit slow development, display increased mortality, and develop into smaller adults than those reared on high-quality host plants. In turn, the resultant smaller females also lay fewer eggs (Ekbom 1997). Furthermore, larvae developing on S. alba have been shown to have a significantly higher likelihood of being parasitised by both Diospilus capito (Hymenoptera: Braconidae) (Billqvist and Ekbom 2001a) and P. morionellus (Billqvist and Ekbom 2001b) than those developing on B. napus; the increased mobility of larvae on these plants may lead them to greater parasitoid exposure. Neither survival nor size of D. capito individuals appears to differ between those developing on S. alba and B. napus (Billqvist and Ekbom 2001a), suggesting that S. alba favours the production and survival of pollen beetle parasitoids over that of the pest. Indeed, Scheid et al. (2011) found that undisturbed sown wildflower fields with high S. alba densities hosted more pollen beetle parasitoids and suffered less from herbivory than fields with low S. alba densities. The authors concluded that undisturbed flower fields containing S. alba provide a suitable habitat for the build-up and maintenance of pollen beetle parasitoid populations, aside from OSR crops—in other words, S. alba appears to be a promising banker plant for inclusion in field margin mixtures.
Raphanus sativus is another poor-quality pollen beetle host plant with potential for use as a banker plant. Even though pollen beetle larval mortality on the plant is high (Veromann et al. 2012, 2014), emergence trap experiments showed that R. sativus facilitated high pollen beetle parasitoid production (Defra 2013; Skellern et al. submitted). However, there was also strong evidence that the plant could assist proliferation of seed weevil (Ceutorhynchus obstrictus) (Coleoptera: Curculionidae), as larval parasitism rates on R. sativus were extremely low. Forage rape cv. ‘Hobson’ exhibited similarly high pollen beetle parasitoid production characteristics to R. sativus, but there was no evidence that this brassica would exacerbate problems with other pests, indicating that it may be a favourable option as a banker plant in winter-sown mixtures (Defra 2013; Skellern et al. submitted).
The commercial field margin mixtures used in European agri-environment schemes so far have tended to be designed for conservation purposes of specific groups such as farmland birds (e.g. Vickery et al. 2009) or pollinators (e.g. Carvell et al. 2006), and few contain species in the Brassicaceae. However, these often have value for supporting predator species in agroecosystems (reviewed by e.g. Gurr et al. 2000; Haaland et al. 2011). Although predators are probably less important than parasitoids in biocontrol of pollen beetle (Nilsson and Andreasson 1987; Hokkanen et al. 1988), several species in groups including carabid beetles, rove beetles, and spiders may predate on pollen beetle larvae, either in the flowers or as they drop to the ground to pupate (Büchs and Alford 2003; Warner et al. 2008; Table 1). Data on predation rates and effects on populations of pollen beetles are scarce but predation has been estimated to cause around 20% of total pollen beetle mortality (Buchi 2002; reviewed by Büchs 2003). These groups are known to be supported by field margins due to provision of food and refuge sites, and in particular overwintering sites. Carabid beetles (e.g. Rouabah et al. 2015; reviewed by Haaland et al. 2011), ladybirds, in particular Coccinella septempunctata (e.g. Meek et al. 2002; Burgio et al. 2006; Tschumi et al. 2016) and Staphylinid beetles (e.g. Burgio et al. 2006) as well as some spiders (e.g. Meek et al. 2002) often show a greater diversity in flowering field margins than other margins or habitats, although vegetation structure species composition, age, and management are also important (Rouabah et al. 2015; reviewed by Haaland et al. 2011). Field margins have been shown to have positive effects on the abundance of these predators in adjacent crops including OSR; Frank and Nentwig (1995) and Frank (1997) showed species richness of carabid beetles and spiders in OSR was positively influenced by adjacent flowering ‘weed’ strips but the number of species and individuals was reduced with distance in-crop from the margin (see also Büchs 2003). A better understanding of the movement of predators between field margins and the crop (and vice versa) is required in order to optimise the potential of field margins to underpin conservation biocontrol by generalist predators in OSR (see Begg et al. 2017). The negative effects of these interventions on other pests must also be understood and minimised; wildflower strips sown next to OSR have been shown to lead to slug damage (Frank 1998).
In recent years, the focus has now turned towards development of ‘multi-functional’ margins that provide a wider range of ecosystem services to crops throughout entire arable rotations (Baverstock et al. 2014; Hatt et al. in press). Field margin mixtures containing at least a cereal, legume, and a member of the Brassicaceae have been shown to support populations of the natural enemies of crop pests of the main arable rotation, and careful selection of plant composition and timing of sowing can improve margin value by increasing floral resource availability early and late into the season (Baverstock et al. 2015; Defra 2016). In particular, margins containing flowering brassicas can support large numbers of parasitoids (Mansion-Vaquié et al. 2017), many of them brassica specialist (PURE 2013), and for generalists, there is some evidence that these margins may also support carabid species typical of communities associated with OSR (PURE 2013). Spatial positioning of margins also needs to be considered. There is potential to improve biocontrol by spatially separating margins with variation in flowering phenology; there is some evidence that spatial separation of margins providing early and late season floral resources may encourage aphid parasitoid movement into wheat crops (Defra 2016), but the potential of this method to encourage movement of the natural enemies of brassica specialist pests into OSR has not yet been investigated. Clearly more work is needed to optimise margin mixtures for biocontrol of pollen beetle, show yield effects in the crop, and elucidate minimum margin areas needed for maximal effect.
Local and regional landscape influences on pollen beetles and their natural enemies
Pollen beetle density and herbivory
Investigations into the impact of local landscape complexity (frequently measured as proportions of semi-natural habitats or non-crop area) on pollen beetle density and herbivory have had contrasting conclusions. Some studies found reduced densities or plant damage associated with more complex landscapes (Thies et al. 2003; Thies and Tscharntke 1999; Gladbach et al. 2011; Beduschi et al. 2015), while others have shown a positive relationship (Zaller et al. 2008b; Rusch et al. 2012b, 2013b). Differences in methodologies makes comparisons difficult, and for the studies showing negative relationships, assessments were sometimes made on potted spring OSR plants (Thies et al. 2003) or ‘phytometer’ Sinapis arvensis plants (Gladbach et al. 2011) used towards the end of winter OSR flowering, rather than during initial crop colonisation from overwintering sites. Beetles dispersing from OSR crops at the end of flowering are likely to be less specific (to the Brassicaceae) in their requirements than those primarily interested in finding breeding sites early in the season, and may simply move on to other (non-brassicaceous) flowering plants to feed (Free and Williams 1978b; Williams and Free 1978; Ouvrard et al. 2016). The greater availability of these plants in more complex landscapes may have led to a dilution effect on beetle numbers found on the Brassicaceae sampled at that time of year.
When resolution of habitat types is increased and wooded areas are considered separately, the results have been more consistent across studies. Increasing proportions of woodland in the landscape are generally associated with higher pollen beetle densities and damage (Valantin-Morison et al. 2007; Zaller et al. 2008b, 2009; Rusch et al. 2012b, 2013b). Grasslands have shown inconsistent results, with both negative (Thies and Tscharntke 1999) and positive (Rusch et al. 2013b) relationships found between grassland proportions and pollen beetle infestation or herbivory. These results are most probably due to the role of such habitats, particularly woodlands, as overwintering sites which later become the springtime source of emerging beetles (Juhel et al. 2017). While Rusch et al. (2012a) showed that pollen beetles can overwinter in both woodland and grassland habitats, emerging beetles were more associated with local habitat characteristics such as low soil moisture and a thick litter layer than with habitat type per se. These characteristics may frequently be associated with woodlands, but less often with grasslands where the factors influencing litter thickness, such as habitat age and grazing intensity are likely to be more variable.
Oilseed rape area and spatial arrangement
Many studies that have investigated local landscape-scale effects of OSR area on pollen beetle density or damage in the same season have shown no influence (Thies and Tscharntke 1999; Thies et al. 2003, 2008; Gladbach et al. 2011; Scheid et al. 2011; Rusch et al. 2013b; Skellern et al. 2017), suggesting that proximity to overwintering and early spring feeding sites may be a more important determinant of infestation and damage than OSR area. Others, however, have shown negative relationships, often attributed to dilution effects (Zaller et al. 2008a, b; Moser et al. 2009; Schneider et al. 2015) or a positive relationship (Valantin-Morison et al. 2007). Interestingly, Schneider et al. (2015) found that a dilution of pollen beetle abundance associated with spatially increasing OSR landscape proportions led to higher OSR yields. Discrepancies among studies could result from unknown differences among study regions (Rusch et al. 2013b) or sampling methodology differences, particularly in relation to scale, or to the temporal dynamics of the relationship between the beetles and the OSR crop. Indeed, Beduschi et al. (2015) found that the effect of surrounding OSR area on beetle abundance changed with time, from negative during flowering to positive after flowering, probably reflecting dilution and crowding effects, respectively.
Few studies have investigated possible local landscape-scale effects of OSR area in the year previous to sampling, on pollen beetle infestation or damage. Schneider et al. (2015) showed that between-year increases in OSR proportions led to beetle dilution effects, and Beduschi et al. (2015) found that increasing landscape proportions of OSR negatively influenced beetle abundance 1 or 2 years later, though this effect was considered to be mediated through parasitism. Thies et al. (2008) observed no influence of this variable on pollen beetle herbivory, while a field-scale model of the influence of landscape and weather variables on trap catches of pollen beetles immigrating into OSR crops showed a positive influence of the previous season’s OSR proportions on trap catch (Skellern et al. 2017). Factors including the extent to which regional-scale landscape structure necessitates long-distance migration (Rusch et al. 2013b), and differences in parasitism rates, may explain these differences. For example, in landscapes with ample breeding and overwintering sites where only limited migration is necessary, it might be expected that beetle abundance would reflect surrounding OSR area in the previous year, particularly where parasitism rates are low. By contrast, in landscapes where long-distance migration is necessary because they are more compartmentalised at the regional scale (i.e. some areas with large open fields and other distinct areas with more complex landscapes), relationships between beetle abundance and the previous season’s OSR area are unlikely to be found, particularly at relatively fine (e.g. 1000 m) sampling scales.
Parasitism
Local landscape complexity effects on pollen beetle parasitism appear generally more consistent across studies than those acting on the beetles themselves, and positive relationships have usually been reported (Thies et al. 2003; Thies and Tscharntke 1999; Scheid et al. 2011; Rusch et al. 2012b; Beduschi et al. 2015), with the exception of two studies, one conducted post-OSR flowering, which found no relationship (Gladbach et al. 2011), and one which found a negative relationship (Schneider et al. 2015). Increasing proportions of woodland, and particularly of grassland within the landscape, are consistently associated with higher parasitism rates in OSR (Thies and Tscharntke 1999; Zaller et al. 2009; Rusch et al. 2011, 2012b). Flowering plants within grasslands and woodlands are likely to provide parasitoids with supplementary nectar resources, and sugar-feeding is known to increase parasitoid fecundity and longevity (e.g. Wäckers et al. 2005; Gillespie et al. 2016), the latter being a factor which could be particularly important during dispersal from emergence sites into OSR crops (Vinatier et al. 2013). In addition to effects over distances of 1500–2000 m, Rusch et al. (2011) noted strong positive smaller-scale (250 m) influences of woodland and grassland area on parasitism rates. This, coupled with the fact that pollen beetle parasitoids sometimes unexpectedly emigrate from OSR patches to the surrounding environment (Williams et al. 2007), suggests that they may supplement their OSR-nectar diets with nectar from the surrounding environment to optimise fitness. Aside from these energetic considerations, grasslands and woodlands may also enhance parasitoid populations through provision of alternative hosts or host plants (Gillespie et al. 2016). Wild Brassicaceae may be important in sustaining parasitoid populations in grassland environments (Hokkanen et al. 1988; Billqvist and Ekbom 2001a, b; Scheid et al. 2011). In woodlands, however, members of the Brassicaceae do not frequently occur, but several Meligethes species, feeding on herbs such as Lamium spp. and members of the Rosaceae which are commonly present, are also hosts of pollen beetle parasitoids (Horstmann 1981). The importance of alternative hosts in terms of parasitoid production, however, has yet to be ascertained. The positive relationship between parasitism rates and woodland or grassland proportions within the landscape could also be driven by host density dependence, with increased beetle infestation levels leading to enhanced parasitism rates; several studies have reported a density-dependence effect on pollen beetle parasitism (Zaller et al. 2009; Gladbach et al. 2011; Scheid et al. 2011; Schneider et al. 2015).
The reported influence of the current season’s OSR proportions within the local landscape on parasitism rates has been mixed, with several studies finding no effect (Thies et al. 2003, 2008; Thies and Tscharntke 1999; Beduschi et al. 2015). However, Hokkanen et al. (1988) found that parasitism rates were lowest in areas where OSR cultivation occurred over the largest area. Similarly, Schneider et al. (2015) observed that parasitism rates decreased with spatially increasing OSR in the landscape and attributed this to dilution effects, and Thies et al. (2008) showed that inter-annual changes in OSR area resulted in concentration and dilution effects on parasitism (i.e. parasitism increased as OSR area decreased between years, and vice versa). Gladbach et al. (2011) found that parasitism rates on S. alba at the end of OSR flowering increased more strongly with larval density if the surrounding OSR crop area was high, possibly due to post-OSR flowering concentration effects.
The impact of OSR areas in the previous year has been more consistent. Beduschi et al. (2015) observed a strong positive effect of OSR in the previous year on parasitism rates in the next season, and this resulted in a carry-over effect to the following year, affecting the abundance of overwintered beetles. The proximity of OSR crops to those in OSR the previous year (Rusch et al. 2011) and the proportion of previous year’s OSR fields under reduced post-harvest tillage regimes (Rusch et al. 2011, 2012b) have shown positive effects on parasitism rates. Models developed by Vinatier et al. (2012, 2013) indicated that parasitism rates were negatively influenced by longer crop rotations (which reflected a lower proportion of OSR in the landscape) because decreased connectivity between the previous and current season’s OSR crops affected the parasitoid to a greater extent than the more dispersive pollen beetles. In general, the greater influence of the previous season’s vs. the sampled season’s OSR crop area on parasitism rates is not surprising since the parasitoids overwinter in the soil of former OSR fields and emerge from these sites in the following spring, and reduced tillage techniques used to establish crops following OSR minimise tillage-related mortality (Nilsson 2010).
Generalist predators
A handful of studies have investigated the influence of landscape factors on the fecundity, nutritional condition, activity–density, and species richness of generalist predators (spiders and carabids) within OSR crops. Haschek et al. (2012) found that Amara similata oocyte numbers were negatively related to distance from the nearest fallow, and that models best explaining the nutritional condition of male Poecilus cupreus and male A. similata contained parameters relating to percentage of surrounding crop area and distance from the nearest fallow, respectively. As grassy fallows and other non-crop areas represent important refuges for carabid beetles (Collins et al. 2002; Rouabah et al. 2015), the poorer condition of individuals needing to search further afield from these habitats may be related to scarcity of food resources. Indeed, pest abundance (a measure of prey availability) proved to be the most important factor explaining these measures of carabid fecundity and fitness, suggesting that knock-on effects of landscape factors on pest abundance are probably a more important influence on these characteristics in carabids than direct landscape effects.
In a study investigating landscape effects on Pardosa spp. wolf spiders, Drapela et al. (2011) showed that activity–density was highest when roadside grass strip length within the surrounding landscape was greatest, and the distance to the nearest fallow was shortest. There was also evidence to suggest that networks of grassy strips were more effective in facilitating colonisation of arable fields by Pardosa spp. than more patchily distributed fallow, as roadside strips showed higher explanatory power at large scales than fallow did at finer scales. Spider species richness has also been shown to positively respond to landscape factors, with increasing proportions of woody and non-crop areas (Drapela et al. 2008).
Land management implications and conclusions
The reviewed studies indicate that habitat management at the scales of the crop margin, the local landscape, and the regional landscape, particularly if conducted in a co-ordinated manner across these scales, has great potential for improving pollen beetle control and could reduce reliance on insecticides. However, the landscape factors affecting the abundance of pollen beetles on OSR and the efficacy of their natural enemies are complex, and the observations of different researchers in this area are often seemingly confounding. The apparent ambivalent effect of complex—especially woodland-rich—landscapes is particularly interesting as these characteristics promote high parasitism rates yet are also often associated with high beetle infestation and damage levels. However, this could be explained by regional-scale migration of pollen beetles just prior to overwintering (Rusch et al. 2013b; Mauchline et al. 2017); if beetles migrate from regions with a paucity of overwintering sites to more structurally complex regions where overwintering sites are more abundant, the effect of high parasitism rates in these areas may be reduced by inward migration. Thus, the major management implication of this regional-scale migration of beetles is that efforts to promote biological regulation should be concentrated within simple landscapes rather than in complex ones where parasitism rates are already high (see Tscharntke et al. 2005). Increased parasitism in structurally simple areas is likely then to have knock-on effects, lowering pollen beetle pressure in the more complex-landscaped areas.
The tendency of spatially and temporally (inter-annual) increasing OSR proportions within the landscape to result in dilution effects for pollen beetle abundance (Zaller et al. 2008a, b; Moser et al. 2009; Schneider et al. 2015) is important, particularly as pest dilution effects have now been shown to concurrently increase yields, despite the fact that dilution may also affect parasitism rates (Schneider et al. 2015). These findings have led to the proposal of a co-ordinated regional landscape-scale management scheme where OSR cover within a large area would be increased annually, maintaining the benefits of dilution effects for several years. A year with low crop cover (Schneider et al. 2015) or even a year when OSR cultivation ceases (Beduschi et al. 2015) would then be used to ‘reset’ the rotation. Again, however, such schemes are likely to be more successful in structurally simple regions, where dilution effects would not be counteracted by inward migration of beetles in search of overwintering sites, as in more complex landscapes. Also, practical implementation of such schemes, involving many individual land managers, may prove challenging.
Although landscape or regional-scale schemes hold great potential for improving pollen beetle control, fundamentally these must be underpinned by appropriate management at the within-field or crop margin scale, particularly within simple landscapes. The positive effects of pest dilution due to annually increasing OSR area may be offset by parasitoid dilution and lowered biological control (Schneider et al. 2015). Measures to bolster natural pest control are also therefore required, and could include encouraging farmers to use reduced tillage techniques or to leave fallow strips post-OSR in order to reduce tillage-related parasitoid mortality. In particular, however, the establishment of appropriate flower-rich field margin networks, containing appropriate brassicas acting as banker or trap crop plants, would continue to support parasitoid populations by providing nectar resources, encouraging parasitoid production, and aiding their dispersal through improving connectivity between current and previous year’s OSR crops.
References
AHDB-HGCA (2012) Controlling pollen beetle and combating insecticide resistance in oilseed rape. HGCA Information Sheet 13: Spring 2012
Baggen LR, Gurr GM (1998) The influence of food on Copidosoma koehleri (Hymenoptera: Encyrtidae), and the use of flowering plants as a habitat management tool to enhance biological control of potato moth, Phthorimaea operculella (Lepidoptera: Gelechiidae). Biol Control 11:9–17. https://doi.org/10.1006/bcon.1997.0566
Barbosa P (1998) Conservation biological control. Academic Press, San Diego
Bartlet E, Blight MM, Pickett JA, Smart LE, Turner G, Woodcock CM (2004) Orientation and feeding responses of the pollen beetle, Meligethes aeneus, to candytuft, Iberis amara. J Chem Ecol 30:913–925. https://doi.org/10.1023/B:Joec.0000028458.66738.E1
Baverstock J, Pell JK, Storkey J, Torrance MT, Cook SM (2014) Field margins for biocontrol and biodiversity across crop rotations: overview of the aims and approaches of Defra project IF01122. IOBC/wprs Bull 100:29–33
Baverstock J, Torrance MT, Skellern MP, Pell JK, Hartwell G, Cook SM (2015) The effect of sowing date (autumn vs. spring) on the flowering period of biennial margin mixes and their resource value for natural enemies of crop pests. In: Proceedings of the BASF farm network conference: results and recommendations for biodiversity and resource protection in European farms. Bad Dürkheim, Germany, 23–24/11/2015, pp 6–11
Beduschi T, Tscharntke T, Scherber C (2015) Using multi-level generalized path analysis to understand herbivore and parasitoid dynamics in changing landscapes. Landscape Ecol 30:1975–1986. https://doi.org/10.1007/s10980-015-0224-2
Begg GS, Cook SM, Dye R, Ferrante M, Franck P, Lavigne C, Lövei G, Mansion-Vaquie A, Pell JK, Petit S, Quesada N, Ricci B, Birch ANE (2017) A functional overview of conservation biological control. Crop Prot 97:145–158. https://doi.org/10.1016/j.cropro.2016.11.008
Billqvist A, Ekbom B (2001a) Effects of host plant species on the interaction between the parasitic wasp Diospilus capito and pollen beetles (Meligethes spp.). Agric For Entomol 3:147–152. https://doi.org/10.1046/j.1461-9563.2001.00099.x
Billqvist A, Ekbom B (2001b) The influence of host plant species on parasitism of pollen beetles (Meligethes spp.) by Phradis morionellus. Entomol Exp Appl 98:41–47. https://doi.org/10.1046/j.1570-7458.2001.00755.x
Borjesdotter D (2000) Barbarea verna and Barbarea vulgaris as host plants for the pollen beetle (Meligethes aeneus). J Agric Sci 134:213–220. https://doi.org/10.1017/s002185969900742x
Bromand B (1990) Diversities in oilseed rape growing in the Western Palearctic Region. IOBC/wprs Bull 13:7–31
Brooks DR, Perry JN, Clark SJ, Heard MS, Firbank LG, Holdgate R, Mason NS, Shortall CR, Skellern MP, Woiwod IP (2008) National-scale metacommunity dynamics of carabid beetles in UK farmland. J Animal Ecol 77:265–274. https://doi.org/10.1111/j.1365-2656.2007.01331.x
Buchi R (1990) Investigations on the use of turnip rape as a trap plant to control oilseed rape pests. IOBC/wprs Bull 13:32–39
Buchi R (2002) Mortality of pollen beetle (Meligethes spp.) larvae due to predators and parasitoids in rape fields and the effect of conservation strips. Agric Ecosyst Environ 90:255–263. https://doi.org/10.1016/s0167-8809(01)00213-4
Büchs W (2003) Predators as biocontrol agents of oilseed rape pests. In: Alford DV (ed) Biocontrol of oilseed rape pests. Blackwell, Oxford, pp 279–298
Büchs W, Alford DV (2003) Predators of oilseed rape pests. In: Alford DV (ed) Biocontrol of oilseed rape pests. Blackwell, Oxford, pp 181–200
Burgio G, Ferrari R, Boriani L, Pozzati M, van Lenteren J (2006) The role of ecological infrastructures on Coccinellidae (Coleoptera) and other predators in weedy field margins within northern Italy agroecosystems. Bull Insectol 59:59–67
Carvell C, Westrich P, Meek WR, Pywell RF, Nowakowski M (2006) Assessing the value of annual and perennial forage mixtures for bumblebees by direct observation and pollen analysis. Apidologie 37:326–340. https://doi.org/10.1051/apido:2006002
Collins KL, Boatman ND, Wilcox A, Holland JM, Chaney K (2002) Influence of beetle banks on cereal, aphid predation in winter wheat. Agric Ecosyst Environ 93:337–350. https://doi.org/10.1016/s0167-8809(01)00340-1
Cook SM, Watts NP, Hunter F, Smart LE, Williams IH (2004) Effects of a turnip rape trap crop on the spatial distribution of Meligethes aeneus and Ceutorhynchus assimilis in oilseed rape. IOBC/wprs Bull 27:199–206
Cook SM, Smart LE, Martin JL, Murray DA, Watts NP, Williams IH (2006) Exploitation of host plant preferences in pest management strategies for oilseed rape (Brassica napus). Entomol Exp Appl 119:221–229. https://doi.org/10.1111/j.1570-7458.2006.00419.x
Cook SM, Jonsson M, Skellern MP, Murray DA, Anderson P, Powell W (2007a) Responses of Phradis parasitoids to volatiles of lavender, Lavendula angustifolia—a possible repellent for their host, Meligethes aeneus. Biocontrol 52:591–598
Cook SM, Khan ZR, Pickett JA (2007b) The use of push-pull strategies in integrated pest management. Annu Rev Entomol 52(1):375–400
Cook SM, Rasmussen HB, Birkett MA, Murray DA, Pye BJ, Watts NP, Williams IH (2007c) Behavioural and chemical ecology underlying the success of turnip rape (Brassica rapa) trap crops in protecting oilseed rape (Brassica napus) from the pollen beetle (Meligethes aeneus). Arthropod Plant Interact 1:57–67. https://doi.org/10.1007/s11829-007-9004-5
Cook SM, Doring TF, Ferguson AW, Martin JL, Skellern MP, Smart LE, Watts NP, Welham SJ, Woodcock C, Pickett JA (2013) Development of an integrated pest management strategy for control of pollen beetles in winter oilseed rape. HGCA project report no. 504. https://cereals.ahdb.org.uk/publications/2013/february/05/development-of-an-integrated-pest-management-strategy-for-control-of-pollen-beetles-in-winter-oilseed-rape.aspx. Accessed November 2017
Čuljak TG, Pernar R, Juran I, Ančić M, Bažok R (2016) Impact of oilseed rape crop management systems on the spatial distribution of Brassicogethes aeneus (Fabricius 1775): implications for integrated pest management. Crop Prot 89:129–138
Daniel C, Dierauer H, Clerc M (2013) The potential of silicate rock dust to control pollen beetles (Meligethes spp.). IOBC/wprs Bull 96:47–55
Defra (2010) A framework for the practical application of semiochemicals in field crops: Defra Project PS2113 final report (authored by Pickett JA, Powell W, Smart LE, Cook SM). http://sciencesearch.defra.gov.uk/Document.aspx?Document=PS2113_9792_FRP.pdf. Accessed November 2017
Defra (2013) Oilseed rape ecology—crop and margin management to encourage natural enemies for crop protection and biodiversity: Defra Project IF0139 final report (authored by Cook, SM, Ferguson, AW, Watts NP, Skellern MP). http://randd.defra.gov.uk/Default.aspx?Menu=Menu&Module=More&Location=None&ProjectID=15611&FromSearch=Y&Status=3&Publisher=1&SearchText=IF0139&SortString=ProjectCode&SortOrder=Asc&Paging=10%20-%20Description. Accessed November 2017
Defra (2016) Field margins for biocontrol and biodiversity across crop rotations. Final report (authored by Cook SM, Baverstock J, Skellern MP, Pell JK). http://randd.defra.gov.uk/Default.aspx?Menu=Menu&Module=More&Location=None&ProjectID=17807&FromSearch=Y&Status=3&Publisher=1&SearchText=IF01122&SortString=ProjectCode&SortOrder=Asc&Paging=10%20-%20Description. Accessed November 2017
Denys C, Tscharntke T (2002) Plant-insect communities and predator-prey ratios in field margin strips, adjacent crop fields, and fallows. Oecologia 130:315–324. https://doi.org/10.1007/s004420100796
Döring TF, Skellern M, Watts N, Cook SM (2012) Colour choice behaviour in the pollen beetle Meligethes aeneus (Coleoptera: Nitidulidae). Physiol Entomol 37:360–378. https://doi.org/10.1111/j.1365-3032.2012.00850.x
Dorn B, Jossi W, Humphrys C, Hiltbrunner J (2014) Screening of natural products in the laboratory and the field for control of pollen beetles. J Appl Entomol 138:109–119. https://doi.org/10.1111/jen.12086
Drapela T, Moser D, Zaller J, Frank T (2008) Spider assemblages in winter oilseed rape affected by landscape and site factors. Ecography 31:254–262. https://doi.org/10.1111/j.0906-7590.2008.5250.x
Drapela T, Frank T, Heer X, Moser D, Zaller JG (2011) Landscape structure affects activity density, body size and fecundity of Pardosa wolf spiders (Araneae: Lycosidae) in winter oilseed rape. Eur J Entomol 108:609–614
Eigenbrode SD, Birch AN, Lindzey S, Meadow R, Snyder WE (2016) A mechanistic framework to improve understanding and applications of push-pull systems in pest management. J Appl Ecol 53:202–212. https://doi.org/10.1111/1365-2664.12556
Ekbom B (1997) Host plant choice by pollen beetles (Meligethes spp.). Vaxtskyddsnotiser 61:87–91
Ekbom B (1998) Clutch size and larval performance of pollen beetles on different host plants. Oikos 83:56–64
Ekbom B, Borg A (1996) Pollen beetle (Meligethes aeneus) oviposition and feeding preference on different host plant species. Entomol Exp Appl 78:291–299
Ekbom B, Popov SYA (2004) Host plant affects pollen beetle (Meligethes aeneus) egg size. Physiol Entomol 29:118–122. https://doi.org/10.1111/j.0307-6962.2004.0373.x
Ellis S, Berry P (2012) Re-evaluating thresholds for pollen beetle in oilseed rape. HGCA publication PR495. AHDB-HGCA, Kenilworth
Eurostat (2016) Agriculture database. http://ec.europa.eu/eurostat/web/agriculture/data/database. Accessed October 2016; last accessed November 2017
Ferguson AW, Skellern MP, Johnen A, von Richthofen J-S, Watts NP, Bardsley E, Murray DA, Cook SM (2016) The potential of decision support systems to improve risk assessment for pollen beetle management in winter oilseed rape. Pest Manag Sci 72:609–617. https://doi.org/10.1002/ps.4069 doi
Frank T (1997) Species diversity of ground beetles (Carabidae) in sown weed strips and adjacent fields. Biol Agric Hortic 15:297–307
Frank T (1998) Slug damage and numbers of the slug pests, Arion lusitanicus and Deroceras reticulatum, in oilseed rape grown beside sown wildflower strips. Agric Ecosyst Environ 7:67–78. https://doi.org/10.1016/S0167-8809(97)00108-4
Frank SD (2010) Biological control of arthropods using banker plant systems: past progress and future directions. Biol Control 52:8–16
Frank T, Nentwig W (1995) Ground dwelling spiders (Araneae) in sown weed strips and adjacent fields. Acta Oecol 16:179–193
Frank T, Drapela T, Moser D, Zaller JG (2010) Insect pests and spiders in oilseed rape and their response to site and landscape factors. In: Williams IH (ed) Biocontrol-based integrated management of oilseed rape pests. Springer, London, pp 285–304
Free JB, Williams IH (1978a) A survey of the damage caused to crops of oil-seed rape (Brassica napus L.) by insect pests in south-central England and their effect on seed yield. J Agric Sci 90:417–424
Free JB, Williams IH (1978b) Responses of the pollen beetle Meligethes aeneus, and the seed weevil Ceutorhynchus assimilis, to oilseed rape Brassica napus, and other plants. J App Ecol 15:761–774
Friederichs K (1920) Untersuchungen über Rapsglanzkäfer in Mecklrnburg. J Appl Entomol 7:1–36
Gillespie MA, Gurr GM, Wratten SD (2016) Beyond nectar provision: the other resource requirements of parasitoid biological control agents. Entomol Exp Appl 159:207–221
Gladbach DJ, Holzschuh A, Scherber C, Thies C, Dormann CF, Tscharntke T (2011) Crop-noncrop spillover: arable fields affect trophic interactions on wild plants in surrounding habitats. Oecologia 166:433–441. https://doi.org/10.1007/s00442-010-1870-3
Gurr GM, Wratten SD, Barbosa P (2000) Success in conservation biological control of arthropods. In: Gurr GM, Wratten SD (eds) Biological control: measures of success. Klewer Academic, Dordrecht, pp 105–132
Gurr GM, Wratten SD, Altieri MA (2004) Ecological engineering for pest management: advances in habitat manipulation for arthropods. CSIRO, Collingwood
Haaland C, Naisbit RE, Berster L-F (2011) Sown wildflower strips for insect conservation: a review. Insect Conserv and Divers 4(1):60–80
Hansen LM (2004) Economic damage threshold model for pollen beetles (Meligethes aeneus F.) in spring oilseed rape (Brassica napus L.) crops. Crop Prot 23:43–46. https://doi.org/10.1016/S0261-2194(03)00167-4
Harenberg A (1997) Auswirkungen extensiv geführter Anbausysteme in verschiedenen Fruchtfolgen (Raps-, Zuckerrübenfruchtfolge) und einer selbstbegrünenden Daucerbrache auf Spinnen (Arachnida: Araneae). PhD thesis, University of Rostock, Germany
Haschek C, Drapela T, Schuller N, Fiedler K, Frank T (2012) Carabid beetle condition, reproduction and density in winter oilseed rape affected by field and landscape parameters. J Appl Entomol 136:665–674. https://doi.org/10.1111/j.1439-0418.2011.01694.x
Hatt S, Lopes T, Boeraeve F, Chen J, Francis F (2017) Pest regulation and support of natural enemies in agriculture: experimental evidence of within field wildflower strips. Ecol Eng 98:240–245
Hatt S, Uyttenbroeck R, Lopes Chen JL, Piqueray J, Monty A, Francis F (in press) Effect of flower traits and hosts on the abundance of parasitoids in perennial multiple species wildflower strips sown within oilseed rape (Brassica napus) crops. Arthropod Plant Interact. https://doi.org/10.1007/s11829-017-9567-8
Hervé MR (2017) Breeding for insect resistance in oilseed rape: challenges, current knowledge and perspectives. Plant Breeding. https://doi.org/10.1111/pbr.12552
Hervé MR, Cortesero AM (2016) Potential for oilseed rape resistance in pollen beetle control. Arthropod Plant Interact 10:463–475. https://doi.org/10.1007/s11829-016-9438-8
Hokkanen HMT (1989) Biological and agrotechnical control of the rape blossom beetle Meligethes aeneus (Coleoptera, Nitidulidae). Acta Ecol Fennica 53:25–29
Hokkanen HMT (1991) Trap cropping in insect pest management. Annu Rev Entomol 36:119–138. https://doi.org/10.1146/annurev.en.36.010191.001003
Hokkanen HMT (1993) Overwintering survival and spring emergence in Meligethes aeneus—effects of body weight, crowding, and soil treatment with Beauveria bassiana. Entomol Exp Appl 67:241–246
Hokkanen HMT (2008) Biological control methods of insect pests in oilseed rape. EPPO Bull 38:104–109
Hokkanen HMT, Menzler-Hokkanen (2017) Use of entomopathogenic fungi in insect pest management of Brassica oilseed crops. In: Reddy GVP (ed) Integrated management of insect pests on Canola and other Brassica oilseed crops. CAB International, Boston, pp 373–382
Hokkanen HMT, Granlund H, Husberg G-B, Markkula M (1986) Trap crops used successfully to control Meligethes aeneus (Coleoptera: Nitidulidae). Ann Entomol Fenn 52:115–120
Hokkanen HMT, Husberg G-B, Söderblom M (1988) Natural enemy conservation for the integrated control of the rape blossom beetle Meligethes aeneus F. Ann Agric Fenn 27:281–294
Hopkins RJ, Ekbom B (1999) The pollen beetle Meligethes aeneus changes egg production rate to match host quality. Oecologia 120:274–278. https://doi.org/10.1007/pl00008823
Horstmann K (1981) Revision der Europäischen Tersilochinen II (Hymenoptera: Ichneumonidae). Spixiana 4:1–76
Husberg GB, Hokkanen HMT (2001) Effects of Metarhizium anisopliae on the pollen beetle Meligethes aeneus and its parasitoids Phradis morionellus and Diospilus capito. Biocontrol 46:261–273. https://doi.org/10.1023/a:1011479616787
Johnen A, von Richthofen J-S (2013) The decision-support system proPlant expert: a computer-based tool for integrated pest management in Europe. IOBC/wprs Bull 96:99–105
Jönsson M, Anderson P (2007) Emission of oilseed rape volatiles after pollen beetle infestation; behavioural and electrophysiological responses in the parasitoid Phradis morionellus. Chemoecology 17:201–207. https://doi.org/10.1007/s00049-007-0379-7
Jourdheuil P (1960) Influence de quelques facteurs écologiques sur les fluctuations de population d’une biocénose parasitaire: Étude relative á quelque Hyménoptères (Ophioninae, Diospilinae, Euphorinae) parasites de divers Coléoptères inféodès aux crucifères. Ann Épiphyt 11:445–658
Juhel AS, Barbu CM, Franck P, RogerEstrade J, Butier A, Bazot M et al. (2017) Characterization of the pollen beetle, Brassicogethes aeneus, dispersal from woodlands to winter oilseed rape fields. PLoS ONE 12(8):e0183878. https://doi.org/10.1371/journal.pone.0183878
Kaasik R, Kovács G, Kaart T, Metspalu L, Williams IH, Veromann E (2014a) Meligethes aeneus oviposition preferences, larval parasitism rate and species composition of parasitoids on Brassica nigra, Raphanus sativus and Eruca sativa compared with on Brassica napus. Biol Control 69:65–71. https://doi.org/10.1016/j.biocontrol.2013.11.002
Kaasik R, Kovacs G, Toome M, Metspalu L, Veromann E (2014b) The relative attractiveness of Brassica napus, B. rapa, B. juncea and Sinapis alba to pollen beetles. Biocontrol 59:19–28. https://doi.org/10.1007/s10526-013-9540-0
Klingenberg A, Ulber B (1994) Untersuchungen zum Auftreten dar Tersilochinae (Hym., Ichneumonidae) als Larvalparasitoide einiger Rapsschädlinge im Raum Göttingen 1990 und 1991 und zu deren Schlupfabundanz nach unterschiedlicher Bodenbearbeitung. J App Entomol 117:287–299. https://doi.org/10.1111/j.1439-0418.1994.tb00737.x
Kraus P, Kromp B (2002) Parasitization rates of the oilseed rape pests Ceutorhynchus napi, Ceutorhynchus pallidactylus (Coleoptera, Curculionidae) and Meligethes aeneus (Coleoptera, Nitidulidae) by ichneumonids in several localities of eastern Austria. IOBC/wprs Bull 25:117–122
Krooß S (1996) Staphyliniden in abgestuft extensiv bewirtschafteten Agrarökosystemen: Populations-dynamik, Gemainschaftsstruktur und Bedeutung als Prädatoren. PhD thesis, University of Göttingen, Germany
Lu Z-X, Zhu P-Y, Gurr GM, Zheng X-S, Read DMY, Heong K-L, Yang Y-J, Xu H-X (2014) Mechanisms for flowering plants to benefit arthropod natural enemies of insect pests: prospects for enhanced use in agriculture. Insect Sci 21:1–12
Luka H, Pfiffner W, Wyss E (1998) Amara ovata und Amara similata (Coleoptera, Carabidae), zwei phytophage Laufkäferarten in Rapsfelden. Mitt der Schweizer Entomol Gesellschaft 71:125–131
Mansion-Vaquié A, Ferrante M, Cook SM, Pell JK, Lövei GL (2017) Manipulating field margins to increase predation intensity in winter wheat (Triticum eastivum) fields in Denmark. J Appl Entomol. https://doi.org/10.1111/jen.12385
Mauchline AL, Birkett MA, Woodcock CM, Pickett JA, Osborne JL, Powell W (2008) Electrophysiological and behavioural responses of the pollen beetle, Meligethes aeneus, to volatiles from a non-host plant, lavender, Lavandula angustifolia (Lamiaceae). Arthropod Plant Interact 2:109–115. https://doi.org/10.1007/s11829-008-9038-3
Mauchline AL, Cook SM, Powell W, Osborne JL (2013) Effects of non-host plant odour on Meligethes aeneus during immigration to oilseed rape. Entomol Exp Appl 46:313–320
Mauchline AL, Cook SM, Powell W, Chapman JW, Osborne JL (2017) Predicting migratory flight behaviour of the pollen beetle Meligethes aeneus (.) Pest Manage Sci 73:1076–1082. https://doi.org/10.1002/ps.4550
Mauchline AL, Hervé MR, Cook SM (in press) Semiochemical-based alternatives to synthetic toxicant insecticides for pollen beetle management. Arthropod Plant Interact. https://doi.org/10.1007/s11829-017-9569-6
Meek B, Loxton D, Sparks T, Pywell R, Pickett H, Nowakowski M (2002) The effect of arable field margin composition on invertebrate biodiversity. Biol Conserv 106:259–271. https://doi.org/10.1016/S0006-3207(01)00252-X
Menzler-Hokkanen I, Hokkanen HMT (2005) Developing entomopathogenic nematode delivery systems for biological control of oilseed rape pests. IOBC/wprs Bull 28:19–22
Moser D, Drapela T, Zaller JG, Frank T (2009) Interacting effects of wind direction and resource distribution on insect pest densities. Basic Appl Ecol 10:208–215. https://doi.org/10.1016/j.baae.2008.03.008
Nilsson C (1988) Pollen beetles (Meligethes aeneus) and flowering in rape. Swed J Agric Res 18:113–118
Nilsson C (2003) Parasitoids of the pollen beetles. In: Alford DV (ed) Biocontrol of oilseed rape pests. Blackwell, Oxford, pp 73–85
Nilsson C (2010) Impact of soil tillage on parasitoids of oilseed rape pest. In: Williams IH (ed) Biocontrol-based integrated management of oilseed rape pests. Springer, London, pp 305–312
Nilsson C, Andreasson B (1987) Parasitoids and predators attacking pollen beetles (Meligethes aeneus F.) in spring and winter rape in southern Sweden. IOBC/wprs Bull 10:64–73
Osbourne P (1960) Observations on the natural enemies of Meligethes aeneus. (F.) and M. viridescens (F.) [Coleoptera: Nitidulidae]. Parasitology 50:91–110
Ouvrard P, Hicks DM, Mouland M, Nicholls JA, Baldock KC, Goddard MA, Kunin WE, Potts SG, Thieme T, Veromann E, Stone GNGN. (2016) Molecular taxonomic analysis of the plant associations of adult pollen beetles (Nitidulidae: Meligethinae), and the population structure of Brassicogethes aeneus. Genome 59:1101–1116. https://doi.org/10.1139/gen-2016-0020
Pavela R (2011) Insecticidal and repellent activity of selected essential oils against of the pollen beetle, Meligethes aeneus (Fabricius) adults. Ind Crops Prod 34:888–892
Pickett CH, Bugg RL (1998) Enhancing biological control: habitat management to promote natural enemies of agricultural pests. University of California Press, Berkeley
Piper R, Williams IH (2004) Incidence and feeding activity of epigeic, predatory invertebrates within winter oilseed rape in the UK, with comparisons between integrated and conventional crop management. Bull OILB/SROP 27:281–288
Potting RPJ, Perry JN, Powell W (2005) Insect behavioural ecology and other factors affecting the control efficacy of agro-ecosystern diversification strategies. Ecol Model 182:199–216. https://doi.org/10.1016/j.ecolmodel.2004.07.017
PURE (2013) Deliverable 10.5: recommendation of manipulation of field margins (authored by Begg, GS, Cook, SM & Lövei, G). http://www.pure-ipm.eu/sites/default/files/content/files/D10.5_vfinal.pdf. Accessed November 2017
Ramsden MW, Kendall SL, Ellis SA, Berry PM (2017) A review of economic thresholds for invertebrate pests in UK arable crops. Crop Prot 96:30–43. https://doi.org/10.1016/j.cropro.2017.01.009
Rebek EJ, Sadof CS, Hanks LM (2006) Influence of floral resource plants on control of an armored scale pest by the parasitoid Encarsia citrina (Craw.) (Hymenoptera: Aphelinidae). Biol Control 37:320–328. https://doi.org/10.1016/j.biocontrol.2005.10.009
Robinson KA, Jonsson M, Wratten SD, Wade MR, Buckley HL (2008) Implications of floral resources for predation by an omnivorous lacewing. Basic Appl Ecol 9:172–181. https://doi.org/10.1016/j.baae.2007.01.002
Rouabah A, Villerd J, Amiaud B, Plantureux S, Lasserre-Joulin F (2015) Response of carabid beetles diversity and size distribution to the vegetation structure within differently managed field margins. Agric Ecosyst Environ 200:21–32. https://doi.org/10.1016/j.agee.2014.10.011
Rusch A, Valantin-Morison M, Sarthou JP, Roger-Estrade J (2010) Integrating crop and landscape management into new crop protection strategies to enhance biological control of oilseed rape insect pests. In: Williams IH (ed) Biocontrol-based integrated management of oilseed rape pests. Springer, Dordrecht, pp 415–448
Rusch A, Valantin-Morison M, Sarthou J-P, Roger-Estrade J (2011) Multi-scale effects of landscape complexity and crop management on pollen beetle parasitism rate. Landscape Ecol 26:473–486. https://doi.org/10.1007/s10980-011-9573-7
Rusch A, Valantin-Morison M, Roger-Estrade J, Sarthou J-P (2012a) Local and landscape determinants of pollen beetle abundance in overwintering habitats. Agric For Entomol 14:37–47. https://doi.org/10.1111/j.1461-9563.2011.00547.x
Rusch A, Valantin-Morison M, Roger-Estrade J, Sarthou JP (2012b) Using landscape indicators to predict high pest infestations and successful natural pest control at the regional scale. Landscape Urban Plan 105:62–73. https://doi.org/10.1016/j.landurbplan.2011.11.021
Rusch A, Suchail S, Valantin-Morison M, Sarthou J-P, Roger-Estrade J (2013a) Nutritional state of the pollen beetle parasitoid Tersilochus heterocerus foraging in the field. Biocontrol 58:17–26. https://doi.org/10.1007/s10526-012-9463-1
Rusch A, Valantin-Morison M, Sarthou JP, Roger-Estrade J (2013b) Effect of crop management and landscape context on insect pest populations and crop damage. Agric Ecosyst Environ 166:118–125. https://doi.org/10.1016/j.agee.2011.05.004
Scheid BE, Thies C, Tscharntke T (2011) Enhancing rape pollen beetle parasitism within sown flower fields along a landscape complexity gradient. Agric For Entomol 13:173–179. https://doi.org/10.1111/j.1461-9563.2010.00516.x
Schernéy F (1959) Unsere laufkäfer. Ziemsen, Wittenberg
Schernéy F (1961) Beiträge zur biologie und ökonomischen bedeutung räuberisch lebender käferarten. Z Angew Zool 48:163–175
Schlein O, Büchs W, Niepold F (2006) Proof of pest larval consumption by predators using the PCR-technique and microscopical gut dissection. In: Proceedings of the international symposium on integrated pest management in oilseed rape. Göttingen, Germany, 3–5 April 2006
Schneider G, Krauss J, Riedinger V, Holzschuh A, Steffan-Dewenter I (2015) Biological pest control and yields depend on spatial and temporal crop cover dynamics. J App Ecol 52:1283–1292. https://doi.org/10.1111/1365-2664.12471
Shelton AM, Badenes-Perez FR (2006) Concepts and applications of trap cropping in pest management. Annu Rev Entomol 51:285–308. https://doi.org/10.1146/annurev.ento.51.110104.150959
Shelton AM, Nault BA (2004) Dead-end trap cropping: a technique to improve management of the diamondback moth, Plutella xylostella (Lepidoptera: Plutellidae). Crop Prot 23:497–503
Skellern MP, Cook SM (in press) The potential of crop management practices to reduce pollen beetle damage in oilseed rape. Arthropod Plant Interact. https://doi.org/10.1007/s11829-017-9571-z
Skellern MP, Welham SJ, Watts NP, Cook SM (2017) Meteorological and landscape influences on pollen beetle Meligethes aeneus immigration into oilseed rape crops: a model to aid development of effective pest monitoring systems. Agr Ecosyst Environ 241:150–159. https://doi.org/10.1016/j.agee.2017.03.008
Skellern MP, Clark SJ, Ferguson AW, Watts NP, Cook SM Banker plant bonuses? The benefits and risks of including brassica plants in field margins to promote conservation biocontrol of specialist pests (submitted)
Sotherton NW (1984) The distribution and abundance of predatory arthropods overwintering on farmland. Ann Appl Biol 105:423–429. https://doi.org/10.1111/j.1744-7348.1984.tb03068.x
Thieme T, Heimbach U, Muller A (2010) Chemical control of insect pests and insecticide resistance in oilseed rape. In: Williams IH (ed) Biocontrol-based integrated management of oilseed rape pests. Springer, London, pp 131–335
Thies C, Tscharntke T (1999) Landscape structure and biological control in agroecosystems. Science 285:893–895. https://doi.org/10.1126/science.285.5429.893
Thies C, Tscharntke T (2010) Biological rape pest control in spatio-temporally changing landscapes. In: Williams IH (ed) Biocontrol-based integrated management of oilseed rape pests. Springer, London, pp 273–284
Thies C, Steffan-Dewenter I, Tscharntke T (2003) Effects of landscape context on herbivory and parasitism at different spatial scales. Oikos 101:18–25. https://doi.org/10.1034/j.1600-0706.2003.12567.x
Thies C, Steffan-Dewenter I, Tscharntke T (2008) Interannual landscape changes influence plant–herbivore–parasitoid interactions. Agric Ecosyst Environ 125:266–268. https://doi.org/10.1016/j.agee.2007.12.011
Thomas MB, Wratten SD, Sotherton NW (1991) Creation of island habitats in farmland to manipulate populations of beneficial arthropods—predator densities and emigration. J App Ecol 28:906–917. https://doi.org/10.2307/2404216
Tscharntke T, Klein AM, Kruess A, Steffan-Dewenter I, Thies C (2005) Landscape perspectives on agricultural intensification and biodiversity–ecosystem service management. Ecol Lett 8:857–874. https://doi.org/10.1111/j.1461-0248.2005.00782.x
Tschumi M, Albrecht M, Collatz J, Dubsky V, Entling MH, Najar-Rodriguez AJ, Jacot K (2016) Tailored flower strips promote natural enemy biodiversity and pest control in potato crops. J Appl Ecol 53:1169–1176
Ulber B, Williams IH, Klukowski Z, Luik A, Nilsson C (2010) Parasitoids of oilseed rape pests in Europe: key species for conservation biocontrol. In: Williams IH (ed) Biocontrol-based integrated management of oilseed rape pests. Springer, London, pp 45–76
Valantin-Morison M, Meynard J-M, Dore´ T (2007) Effects of crop management and surrounding field environment on insect incidence in organic winter oilseed rape (Brassica napus L.). Crop Prot 26:1108–1120
Veromann E, Luik A, Metspalu L, Williams IH (2006) Key pests and their parasitoids on spring and winter oilseed rape in Estonia. Entomol Fenn 17:400–404
Veromann E, Saarniit M, Kevvai R, Luik A (2009) Effect of crop management on the incidence of Meligethes aeneus Fab. and their larval parasitism rate in organic and conventional winter oilseed rape. Agron Res 7:548–554
Veromann E, Metspalu L, Williams IH, Hiiesaar K, Mand M, Kaasik R, Kovacs G, Jogar K, Svilponis E, Kivimagi I, Ploomi A, Luik A (2012) Relative attractiveness of Brassica napus, Brassica nigra, Eruca sativa and Raphanus sativus for pollen beetle (Meligethes aeneus) and their potential for use in trap cropping. Arthropod Plant Interact 6:385–394. https://doi.org/10.1007/s11829-012-9191-6
Veromann E, Toome M, Kanaste A, Kaasik R, Copolovici L, Flink J, Kovacs G, Narits L, Luik A, Niinemets U (2013) Effects of nitrogen fertilization on insect pests, their parasitoids, plant diseases and volatile organic compounds in Brassica napus. Crop Prot 43:79–88. https://doi.org/10.1016/j.cropro.2012.09.001
Veromann E, Kaasik R, Kovács G, Metspalu L, Williams IH, Mänd M (2014) Fatal attraction: search for a dead-end trap crop for the pollen beetle (Meligethes aeneus). Arthropod Plant Interact 8:373–381. https://doi.org/10.1007/s11829-014-9325-0
Vickery JA, Feber RE, Fuller RJ (2009) Arable field margins managed for biodiversity conservation: a review of food resource provision for farmland birds. Agric Ecosyst Environ 133:1–13. https://doi.org/10.1016/j.agee.2009.05.012
Vinatier F, Gosme M, Valantin-Morison M (2012) A tool for testing integrated pest management strategies on a tritrophic system involving pollen beetle, its parasitoid and oilseed rape at the landscape scale. Landscape Ecol 27:1421–1433. https://doi.org/10.1007/s10980-012-9795-3
Vinatier F, Gosme M, Valantin-Morison M (2013) Explaining host–parasitoid interactions at the landscape scale: a new approach for calibration and sensitivity analysis of complex spatio-temporal models. Landscape Ecol 28:217–231. https://doi.org/10.1007/s10980-012-9822-4
Wäckers F, van Rijn P, Bruin J (2005) Plant-provided food for carnivorous insects: a protective mutualism and its applications. Cambridge University Press, Cambridge
Warner DJ (2001) The potential of Carabidae in the control of insect pests of winter oilseed rape. PhD thesis, University of Hertfordshire, UK
Warner DJ, Allen-Williams LJ, Warrington S, Ferguson AW, Williams IH (2008) Implications for conservation biocontrol of spatio-temporal relationships between carabid beetles and coleopterous pests in winter oilseed rape. Agric For Entomol 10:375–387. https://doi.org/10.1111/j.1461-9563.2008.00391.x
Williams IH (2010) The major insect pests of oilseed rape in Europe and their management: an overview. In: Williams IH (ed) Biocontrol-based integrated management of oilseed rape pests. Springer, London, pp 1–43
Williams IH, Cook SM (2010) Crop location by oilseed rape pests and host location by their parasitoids. In: Williams IH (ed) Biocontrol-based Integrated management of oilseed rape pests. Springer, London, pp 215–244
Williams IH, Free JB (1978) The feeding and mating behaviour of pollen beetles (Meligethes aeneus Fab.) and seed weevils (Ceutorhynchus assimilis Payk.) on oil-seed rape (Brassica napus L.). J Agr Sci 91:453–459
Williams IH, Frearson DJT, Barari H, McCartney A (2007) First field evidence that parasitoids use upwind anemotaxis for host-habitat location. Entomol Exp Appl 123:299–307. https://doi.org/10.1111/j.1570-7458.2007.00551.x
Williams IH, Ferguson AW, Kruus M, Veromann E, Warner DJ (2010) Ground beetles as predators of oilseed rape pests: incidence, spatio-temporal distributions and feeding. In: Williams IH (ed) Biocontrol-based integrated management of oilseed rape pests. Springer, London, pp 115–150
Wyrostkiewicz K, Blazejewska A (1985) Isurgus heterocerus Thoms. I. morionellus Holmgr. (Hym: Ichneumonidae) parazytoidy larw slodyszka rzepakowego—Meligethes aeneus F. (Col.: Nitidulidae). Polskie Pismo Entomol 55:391–404
Zaller JG, Moser D, Drapela T, Schmöger C, Frank T (2008a) Effect of within-field and landscape factors on insect damage in winter oilseed rape. Agric Ecosyst Environ 123:233–238. https://doi.org/10.1016/j.agee.2007.07.002
Zaller JG, Moser D, Drapela T, Schmöger C, Frank T (2008b) Insect pests in winter oilseed rape affected by field and landscape characteristics. Basic Appl Ecol 9:682–690. https://doi.org/10.1016/j.baae.2007.10.004
Zaller J, Moser D, Drapela T, Schmoger C, Frank T (2009) Parasitism of stem weevils and pollen beetles in winter oilseed rape is differentially affected by crop management and landscape characteristics. Biocontrol 54:505–514
Zimmer CT, Köhler H, Nauen R (2014) Baseline susceptibility and insecticide resistance monitoring in European populations of Meligethes aeneus and Ceutorhynchus assimilis collected in winter oilseed rape. Entomol Exp Appl 150:279–288. https://doi.org/10.1111/eea.12162
Zlof V (2008) Recommendations and conclusions of the Ad hoc EPPO Workshop on insecticide resistance of Meligethes spp. (pollen beetle) on oilseed rape. EPPO Bull 38:65–67. https://doi.org/10.1111/j.1365-2338.2008.01182.x
Acknowledgements
This review was funded by the UK Defra Health & Safety Executive (Chemicals Regulation Directorate) Project PS2141. Rothamsted Research receives strategic funding from the UK Biotechnology and Biological Sciences Research Council (BBSRC). SMC is part-funded by research programme NE/N018125/1 LTS-M ASSIST—Achieving Sustainable Agricultural Systems, funded by BBSRC and NERC. We sincerely thank the two anonymous reviewers for their comments which helped to improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Heikki Hokkanen.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Skellern, M.P., Cook, S.M. Prospects for improved off-crop habitat management for pollen beetle control in oilseed rape. Arthropod-Plant Interactions 12, 849–866 (2018). https://doi.org/10.1007/s11829-018-9598-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-018-9598-9