Abstract
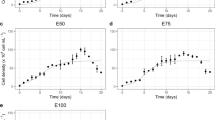
Heavy metal pollution is one of the most severe ecological problems with Lead (Pb) being present in high concentrations. Cyanobacteria growing in polluted sites have evolved different mechanisms to avoid metal toxicity and continue to contribute to the biological activity of the site. The present study was undertaken to understand the mechanism of Pb tolerance in a common diazotroph Desmonostoc muscorum as it tolerated Pb up to 20 mg L-1. During first 48 h, Pb toxicity in this organism resulted in 88% increase in superoxide radicals (SOR) which caused morphological alterations, 37–72% decrease in photosynthetic pigments leading to reduced growth of the organism. The cyanobacterium managed this toxicity by activating antioxidant enzymes superoxide dismutase (SOD) (187% increase) and peroxidase (POD) (181% increase). Simultaneously, increase in the levels of gluthathione (GSH) (53%) and proline (60%), and activity of glutathione reductase (GR) was observed. After 48 h, level of SOR, activities of SOD and POD decreased while the level of GSH and GR remained high. This indicated that Pb toxicity at initial level is managed by the organism by activating both SOD/POD and GSH/GR systems, while later on the organism adopts some other mechanism and is able to manage Pb toxicity by only GSH/GR system.




Similar content being viewed by others
Abbreviations
- A720 :
-
Absorbance at 720 nm
- APX:
-
Ascorbate peroxidase
- AsA:
-
Ascorbic acid
- CAT:
-
Catalase
- Chl a :
-
Chlorophyll a
- EDTA:
-
Ethylenediamine tetra-acetic acid
- GR:
-
Glutathione reductase
- GSH:
-
Reduced glutathione
- GSSG:
-
Glutathione disulphide
- H2O2 :
-
Hydrogen peroxide
- NADPH:
-
Nicotineamide adenine dinucleotide phosphate (reduced)
- NBT:
-
Nitroblue tetrazolium chloride
- POD:
-
Peroxidase
- ROS:
-
Reactive oxygen species
- SEM:
-
Scanning electron microscopy
- SOD:
-
Superoxide dismutase
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Agustina M, Tjahjaningsih W (2021) Assessment of heavy metal (pb) contents in canned crab products by atomic absorption spectrophotometry (AAS). Environ Earth Sci 679(1):012012. https://doi.org/10.1088/1755-1315/679/1/012012
Ahad RIA, Syiem MB (2021) Analyzing dose dependency of antioxidant defense system in the cyanobacterium Nostoc muscorum Meg 1 chronically exposed to Cd2+. Regul Toxicol Pharmacol 456(20):30250–30257. https://doi.org/10.1016/j.cbpc.2020.108950
Ahmad P, Jaleel CA, Salem MA, Nabi G, Sharma S (2010) Roles of enzymatic and nonenzymatic antioxidants in plants during abiotic stress. Crit Rev Biotechnol 30:161–175. https://doi.org/10.3109/07388550903524243
Ajitha V, Sreevidya CP, Sarasan M, Park JC, Mohandas A, Singh ISB, Lee JS (2021) Effects of zinc and mercury on ROS-mediated oxidative stress-induced physiological impairments and antioxidant responses in the microalga Chlorella vulgaris. Environ Sci Pollut Res 28:32475–32492. https://doi.org/10.1007/s11356-021-12950-6
Ali H, Khan E, Ilahi I (2019) Environmental chemistry and ecotoxicology of hazardous heavy metals: environmental persistence toxicity and bioaccumulation. J Chem 1–14. https://doi.org/10.1155/2019/6730305
Arias DM, Garcia J, Uggetti E (2020) Production of polymers by cyanobacteria grown in wastewater: current status, challenges and future perspectives. N Biotechnol 55:46–57. https://doi.org/10.1016/j.nbt.2019.09.001
Baracho DH, Silva JC, Lombardi AT (2019) The effects of copper on photosynthesis and biomolecules yield in Chlorolobion braunii. J Physiol 55(6):1335–1347. https://doi.org/10.1111/jpy.12914
Bates B, Waldern RP, Teare ID (1973) Rapid determination of free proline for water stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Bazihizina N, Taiti C, Marti L, Rodrigo-Moreno A, Spinelli F, Giordano C, Caparrotta S, Gori M, Azzarello E, Mancuso S (2014) Zn2+ induced changes at the root level account for the increased tolerance of acclimated tobacco plants. J Exp Bot 65:4931–4942. https://doi.org/10.1093/jxb/eru251
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44(1):276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Bennett A, Bogorad L (1973) Complementary chromatic adaptation in a filamentous blue-green alga. J Cell Biol 58(2):419–435. https://doi.org/10.1083/jcb.58.2.419
Bhat MA, Singh DP, Khattar JIS, Singh RS (2021) Toxicological effect of pendimethalin on some physiological parameters of the diazotrophic cyanobacterium Desmonostoc muscorum PUPCCC405.10. J Appl Biol Biotechnol 9(4):10–18. https://doi.org/10.7324/JABB.2021.9402
Boden JS, Konhauser KO, Robbins LJ, Sánchez-Baracaldo P (2021) Timing the evolution of antioxidant enzymes in cyanobacteria. Nat Commun 12(1):1–12. https://doi.org/10.1038/s41467-021-24396-y
Carlberg I, Mannervik B (1985) Glutathione reductase. Methods Enzymol 113:484–490. https://doi.org/10.1016/S0076-6879(85)13062-4
Chakraborty S, Mishra AK (2021) Effects of zinc toxicity on the nitrogen-fixing cyanobacteria Anabaena sphaerica ultrastructural, physiological and biochemical analyses. Environ Sci Pollut Res 28:33292–33306. https://doi.org/10.1007/s11356-021-12882-1
Chattergee P, Biswas S, Biswas AK (2018) Sodium chloride primed seeds modulate glutathione metabolism in legume cultivars under NaCl stress. Am J Plant Physiol 13:8–12. https://doi.org/10.3923/ajpp.2018.8.22
Choudhary M, Jetley UK, Abash Khan M, Zutshi S, Fatma T (2007) Effect of heavy metal stress on proline, malondialdehyde and superoxide dismutase activity in the cyanobacterium Spirulina platensis-S5. Ecotoxicol Environ Saf 66:204–209. https://doi.org/10.1016/j.ecoenv.2006.02.002
DalCorso G (2012) Heavy metal toxicity in plants. In: Furini A (ed) Plants and heavy metals. Springer, pp 1–25. https://doi.org/10.1007/978-94-007-4441-7_1
Danouche M, EI Ghachtouli N, El Baouchi A, El Arroussi H (2020) Heavy metals phycoremediation using tolerant green microalgae: enzymatic and non-enzymatic antioxidant systems for the management of oxidative stress. J Environ Chem Engg 8(5):104460. https://doi.org/10.1016/j.jece.2020.104460
Das K, Roychoudhury A (2014) Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front Environ Science 2:53. https://doi.org/10.3389/fenvs.2014.00053
de Lima AA, de Carvalho MA, de Souza SA, Dias PM, da Silva Filho RG, de Meirelles Saramago CS, de Melo Bento CA, Hofer E (2012) Heavy metal tolerance (cr, ag and hg) in bacteria isolated from sewage. Braz J Microbiol 43(4):1620–1631. https://doi.org/10.1590/s1517-838220120004000047
Dhuldhaja U, Pandyab U, Singh S (2018) Anti-oxidative response of cyanobacterium Anabaena sp. strain PCC 7120 to arsenite as(III). J Microbiol 87:848–856. https://doi.org/10.1134/S0026261718060097
Dutta S, Bhadury P (2020) Effect of arsenic on exopolysaccharide production in a diazotrophic cyanobacterium. J Appl Phycol 32:2915–2926. https://doi.org/10.1007/s10811-020-02206-0
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77. https://doi.org/10.1016/0003-9861(59)90090-6
Elstner EF, Heupel A (1976) Inhibition of nitrite formation from hydroxyl ammonium chloride: a simple assay for superoxide dismutase. Anal Biochem 70:616–620. https://doi.org/10.1016/0003-2697(76)90488-7
Etesami H (2018) Bacterial mediated alleviation of heavy metal stress and decreased accumulation of metals in plant tissues: mechanisms and future prospects. Ecotoxicol Environ Saf 147:175–219. https://doi.org/10.1016/j.ecoenv.2017.08.032
Farooqui A, Suhail S, Zeeshan M (2017) Cadmium induced oxidative stress and biochemical responses in cyanobacterium Nostoc muscorum. Russ J Plant Physiol 64(1):124–132. https://doi.org/10.1134/S102144371701006X
Fatma T, Khan MA, Choudhary M (2007) Impact of environmental pollution on cyanobacterial proline content. J Appl Phycol 19(6):625–629. https://doi.org/10.1007/s10811-007-9195-2
Fornazier RF, Ferreira RR, Pereira GJG, Molina SMG, Smith RJ, Lea PJ, Azevedo RA (2002) Cadmium stress in sugarcane callus cultures: effect on antioxidant enzymes. Plant Cell Tissue Organ Culture 71:125–131. https://doi.org/10.1023/A:1019917705111
Gahagen HE, Holm RE, Abeles FB (1968) Effect of ethylene on peroxidase activity. Physiol Plant 21:1270–1279. https://doi.org/10.1111/j.1399-3054.1968.tb07358.x
Gest N, Gautier H, Stevens R (2013) Ascorbate as seen through plant evolution: the rise of a successful molecule? J Exp Bot 64:33–53. https://doi.org/10.1093/jxb/ers297
Ghoneim MM, El-Desoky HS, El-Moselhy KM, Amer A, Abou El-Naga EH, Mohamedein LI, Al-Prol AE (2014) Removal of cadmium from aqueous solution using marine green algae, Ulva lactuca. Egypt J Aquat Res 40(3):235–242. https://doi.org/10.1016/j.ejar.2014.08.005
González-Henao S, Ghneim-Herrera T (2021) Heavy metals in soils and the remediation potential of bacteria associated with the plant microbiome. Front Environ Sci 9:604216. https://www.frontiersin.org/articles/https://doi.org/10.3389/fenvs.2021.604216/full
Goswami S, Ahad RIA, Syiem MB (2019) Expression of copper toxicity in the rice-field cyanobacterium Anabaena oryzae Ind4. Eur Asian J Bio Sci 13(1):57–67. https://doi.org/10.1016/j.ejar.2014.08.005
Gottesfeld P, Tirima S, Anka SM, Fotso A, Nota MM (2019) Reducing lead and silica dust exposures in small-scale mining in northern Nigeria. Ann Work Expo Health 63(1):1–8. https://doi.org/10.1093/annweh/wxy095
Holm G (1954) Chlorophyll mutations in barley. Acta Agric Scand 4:457–471. https://doi.org/10.1080/00015125409439955
Hossain MA, Hoque MA, Burritt DJ, Fujita M (2014) Proline protects plants against abiotic oxidative stress: biochemical and molecular mechanisms. In: Ahmad P (ed) Oxidative damage to plants: Antioxidant networks and signaling. Academic Press, pp 477–522. https://doi.org/10.1016/B978-0-12-799963-0.00016-2
Hu J, Lin B, Yuan M, Lao Z, Wu K, Zeng Y, Fan H (2019) Trace metal pollution and ecological risk assessment in agricultural soil in Dexing Pb/Zn mining area China. Environ Geochem Health 41(2):967–980. https://doi.org/10.1007/s10653-018-0193-x
Hussein MH, Hamouda RA, Elhadary AM, Abuelmagd MA, Ali S, Rizwan M (2019) Characterization and chromium biosorption potential of extruded polymeric substances from Synechococcus mundulus induced by acute dose of gamma irradiation. Environ Sci Pollut Res 26:31998–32012. https://doi.org/10.1007/s11356-019-06202-x
Ishikawa T, Shigeoka S (2008) Recent advances in ascorbate biosynthesis and the physiological significance of ascorbate peroxidise in photosynthesizing organisms. Biosci Biotechnol Biochem 72:1143–1154. https://doi.org/10.1271/bbb.80062
Jarosławiecka A, Piotrowska-Seget Z (2014) Lead resistance in micro-organisms. Microbiol 160(1):12–25. https://doi.org/10.1099/mic.0.070284-0
Kashyap M, Anand V, Ghosh A, Kiran B (2021) Superintending Scenedesmus and Chlorella sp. with lead and cobalt tolerance governed via stress biomarkers. Water Supply 21(5):2387–2399. https://doi.org/10.2166/ws.2021.065
Khattar JIS, Parveen S, Singh Y, Singh DP, Gulati A (2015) Intracellular uptake and reduction of hexavalent chromium by the cyanobacterium Synechocystis sp. PUPCCC 62. J Appl Phycol 27(2):827–837. https://doi.org/10.1007/s10811-014-0374-7
Kratz WA, Myers J (1955) Nutrition and growth of several blue green algae. Amer J Bot 42(3):282–287. https://doi.org/10.1002/j.1537-2197.1955.tb11120.x
Kushwaha A, Hans N, Kumar S, Rani R (2018) A critical review speciation, mobilization and toxicity of lead in soil-microbe-plant system and bioremediation strategies. Ecotoxicol Environ Saf 147:1035–1045. https://doi.org/10.1016/j.ecoenv.2017.09.049
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
Leung PTY, Yi AX, Ip JCH, Mak SST, Leung KMY (2017) Photosynthetic and transcriptional responses of the marine diatom Thalassiosira pseudonana to the combined effect of temperature stress and copper exposure. Mar Pollut Bull 124(2):938–945. https://doi.org/10.1016/j.marpolbul.2017.03.038
Li YP, Wang SL, Nan ZR, Zang F, Sun HL, Zhang Q (2019) Accumulation, fractionation and health risk assessment of fluoride and heavy metals in soil-crop systems in Northwest China. Sci Total Environ 663:307–314. https://doi.org/10.1016/j.scitotenv.2019.01.257
Lim LB, Priyantha N, Lu Y, Zaidi NAHM (2019) Adsorption of heavy metal lead using Citrus grandis (Pomelo) leaves as low-cost adsorbent. Desalin Water Treat 166:44–52. https://doi.org/10.5004/dwt.2019.24620
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Mitra A, Chatterjee S, Kataki S, Rastogi RP, Gupta DK (2021) Bacterial tolerance strategies against lead toxicity and their relevance in bioremediation application. Environ Sci Pollut Res 28:14271–14284. https://doi.org/10.1007/s11356-021-12583-9
Mo L, Yang Y, Zhao D, Qin L, Yuan B, Liang N (2022) Time-dependent toxicity and health effects mechanism of cadmium to three green algae. Int J Environ Res Public Health 19:10974. https://doi.org/10.3390/ijerph191710974
Munagamage T, Rathnayake IV, Pathiratne A, Megharaj M (2020) Comparison of sensitivity of tropical freshwater microalgae to environmentally relevant concentrations of cadmium and hexavalent chromium in three types of growth media. Bull Environ Contam Toxicol 105:397–404. https://doi.org/10.1007/s00128-020-02950-6
Nagajyoti PC, Lee KD, Sreekanth TVM (2010) Heavy metals, occurrence and toxicity for plants: a review. Environ Chem Lett 8:199–216. https://doi.org/10.1007/s10311-010-0297-8
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Nowicka B (2022) Heavy metal–induced stress in eukaryotic algae mechanisms of heavy metal toxicity and tolerance with particular emphasis on oxidative stress in exposed cells and the role of antioxidant response. Enviorn Sci Pollut Res 29:16860–16911. https://doi.org/10.1007/s11356-021-18419-w
Oser BL (1979) Hawks physiological chemistry. McGraw Hill N.Y. USA. pp 702–705. https://doi.org/10.1038/161830b0
Pandey N, Patel A, Tiwari S, Prasad SM (2022) Differential response of copper nanoparticles and ionic copper on growth, chlorophyll fluorescence, oxidative stress, and antioxidant machinery of two paddy field cyanobacteria. Ecotoxicol 31:933–947. https://doi.org/10.1007/s10646-022-02553-3
Parveen S, Khattar JIS, Singh DP (2015) The cyanobacterium Synechocystis sp. PUPCCC 62 a potential candidate for biotransformation of cr (VI) to cr (III) in the presence of sulphate. Environ Sci Pollut Res 22(14):10661–10668. https://doi.org/10.1007/s11356-015-4260-x
Patel A, Tiwari S, Prasad SM (2018) Toxicity assessment of arsenate and arsenite on growth, chlorophyll a fluorescence and antioxidant machinery in Nostoc muscorum. Ecotoxicol Environ Saf 157:369–379. https://doi.org/10.1016/j.ecoenv.2018.03.056
Patel A, Tiwari S, Prasad SM (2021) Arsenate and arsenite-induced inhibition and recovery in two diazotrophic cyanobacteria Nostoc muscorum and Anabaena sp. study on time-dependent toxicity regulation. Environ Sci Pollut Res 1–17. https://doi.org/10.1007/s11356-021-13800-1
Pikula KS, Zakharenko AM, Aruoja V, Golokhvast KS, Tsatsakis AM (2019) Oxidative stress and its biomarkers in microalgal ecotoxicology. Curr Opin Toxicol 13:8–15. https://doi.org/10.1016/j.cotox.2018.12.006
Pinto E, Sigaud-kutner TC, Leitao MA, Okamoto OK, Morse D, Colepicolo P (2003) Heavy metal–induced oxidative stress in algae. J Phycol 39(6):1008–1018. https://doi.org/10.1111/j.0022-3646.2003.02-193
Piotrowska-Niczyporuk A, Bajguz A, Talarek M, Bralska M, Zambrzycka E (2015) The effect of lead on the growth, content of primary metabolites, and antioxidant response of green alga Acutodesmus obliquus (Chlorophyceae). Environ Sci Pollut Res 22:19112–19123. https://doi.org/10.1007/s11356-015-5118-y
Pourrut B, Shahid M, Dumat C, Winterton P, Pinelli E (2011) Lead uptake, toxicity and detoxification in plants. In: Whitacre DM (ed) Reviews of environmental contamination and toxicology. Springer, New York LLC, pp 113–136. https://doi.org/10.1007/978-1-4419-9860-6_4
Qiu C, Wang W, Zhang Y, Zhou GJ, Bi Y (2022) Response of antioxidant enzyme activities of the green microalga Chlorococcum sp. AZHB to Cu2+ and Cd2+ stress. Sustainability 14(16):10320. https://doi.org/10.3390/su141610320
Qu M, Li W, Zhang C, Huang B, Zhao Y (2015) Assessing the pollution risk of soil chromium based on loading capacity of paddy soil at a regional scale. Sci Rep 5:1–8. https://doi.org/10.1038/srep18451
Rahman Z, Singh VP (2019) The relative impact of toxic heavy metals (THMS) (arsenic (As), cadmium (Cd), chromium (Cr)(VI), mercury (Hg) and lead (Pb)) on the total environment: an overview. Environ Monit Asses 191:419. https://doi.org/10.1007/s10661-019-7528-7
Rejeb KB, Abdelly C, Savoure A (2014) How reactive oxygen species and proline face stress together. Plant Physiol Biochem 80:278–284. https://doi.org/10.1016/j.plaphy.2014.04
Ren R, Li Z, Zhang L, Zhou H, Jiang X, Liu Y (2021) Enzymatic and nonenzymatic antioxidant systems impact the viability of cryopreserved Paeonia suffruticosa pollen. Plant Cell Tissue Organ Culture 144(1):233–246. https://doi.org/10.1007/s11240-020-01794-6
Sharma P, Dubey RS (2005) Lead toxicity in plants. Brazilian J Plant Physiol 17:35–52. https://doi.org/10.1590/S1677-04202005000100004
Shen L, Chen R, Wang J, Fan L, Cui L, Zhang Y, Zeng W (2021) Biosorption behavior and mechanism of cadmium from aqueous solutions by Synechocystis sp. PCC6803. RSC Adv 11(30):18637–18650. https://doi.org/10.1039/D1RA02366G
Shivagangaiah CP, Sanyal D, Dasgupta S, Banik A (2021) Phycoremediation and photosynthetic toxicity assessment of lead by two freshwater microalgae Scenedesmus acutus and Chlorella pyrenoidosa. Physiol Plant 173(1):246–258. https://doi.org/10.1111/ppl.13368
Singh DP, Khattar JIS, Kaur G, Gupta M, Singh Y (2015) Effect of pretilachlor on nitrogen uptake and assimilation by the cyanobacterium Desmonostoc muscorum PUPCCC 405.10. Acta Physiol Plant 37:177. https://doi.org/10.1007/s11738-015-1923-7
Sirikhachornkit A, Niyogi KK (2010) Antioxidants and photo-oxidative stress responses in plants and algae. In: Rebeiz CA, Benning C, Bohnert HJ et al (eds) The chloroplast. Springer, Netherlands, Dordrecht, pp 379–396. https://doi.org/10.1007/978-90-481-8531-3_24
Sirunyan AM, Tumasyan A, Adam W, Ambrogi F, Asilar E, Bergauer T, Brandstetter J, Dragicevic M, Erö J, Del Valle AE, Flechl M (2019) Search for vector-like quarks in events with two oppositely charged leptons and jets in proton-proton collisions at √s = 13 TeV. Eur Phys J C 79:1–31. https://doi.org/10.1140/epjc/s10052-019-6855-8
Smedley PL, Nicolli HB, Macdonald DMJ, Barros AJ, Tullio JO (2002) Hygrogeochemistry of arsenic and other inorganic contitutents in ground waters from La Pampa. Argentina Appl Geochem 17:259–284. https://doi.org/10.1016/S0883-2927(01)00082-8
Tamaki S, Mochida K, Suzuki K (2021) Diverse biosynthetic pathways and protective functions against environmental stress of antioxidants in microalgae. Plants 10:1250. https://doi.org/10.3390/plants10061250
Tiwari S, Patel A, Prasad SM (2020) Phytohormone up-regulates the biochemical constituent, exopolysaccharide and nitrogen metabolism in paddy-field cyanobacteria exposed to chromium stress. BMC Microbiol 20:20. https://doi.org/10.1186/s12866-020-01799-3
Van Loon LC (1971) Tobacco polyphenoloxidase. A specific staining method indicating non-identity with peroxidase. Phytochemistry 10:503–507. https://doi.org/10.1016/S0031-9422(00)94689-2
Verbruggen N, Hermans C (2008) Proline accumulation in plants: a review. Amino Acids 35:753–759. https://doi.org/10.1007/s00726-008-0061-6
Verma N, Prasad SM (2021) Regulation of redox homeostasis in cadmium stressed rice field cyanobacteria by exogenous hydrogen peroxide and nitric oxide. Sci Rep 11(1):1–15. https://doi.org/10.1038/s41598-021-82397-9
Verma N, Pandey A, Tiwari S, Prasad SM (2021) Calcium mediated nitric oxide responses: Acquisition of nickel stress tolerance in cyanobacterium Nostoc muscorum ATCC 27893. Biochem Biophys Rep 26:100953. https://doi.org/10.1016/j.bbrep.2021.100953
Wang S, Li Q, Huang S, Zhao W, Zheng Z (2021) Single and combined effects of microplastics and lead on the freshwater algae Microcystis aeruginosa. Ecotoxicol Environ Saf 208:111664. https://doi.org/10.1016/j.ecoenv.2020.111664
Woodbury W, Spencer AK, Stahmann MA (1971) An improved procedure using ferricyanide for detecting catalase isozymes. Anal Biochem 44:301–305. https://doi.org/10.1016/0003-2697(71)90375-7
Zhang J, Li Q, Yv Z, Zhang J, Lu G, Lu Z, Dang Z, Guo C (2019) Bioaccumulation and distribution of cadmium by Burkholderia cepacia GYP1 under oligotrophic condition and mechanism analysis at proteome level. Ecotoxicol Environ Saf 176:162–169. https://doi.org/10.1016/j.ecoenv.2019.03.091
Acknowledgements
The authors are grateful to Head and Coordinator, FIST of DST, DRS-SAP-II of UGC, Department of Botany, Punjabi University, Patiala, for providing laboratory and other infrastructure facilities.
Funding
The authors did not receive any financial support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
Davinder Pal Singh and Jasvirinder Singh conceived the idea, provided the general concept and inputs for each section of the manuscript, and drafted part of the manuscript. Minakshi Chandel, Kirti Sharma and Manzoor performed the experiments and wrote the manuscript after consulting the relevant literature. Jasvirinder and Davinder edited, compiled, and finalized the draft manuscript. Finally, all the authors read and approved it for publication.
Corresponding author
Ethics declarations
Financial or non-financial interests
There are no financial or non-financial interests.
Ethical approval
The present research did not involve any human participants and/or animals. No data/figure/table has been taken from any source which requires permission.
Informed consent
All the authors have gone through the contents of the manuscript and give their consent to submit it for publication.
Competing interests
The authors declare that there are no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chandel, M., Khattar, J.S., Singh, D.P. et al. Pb toxicity at initial level is managed by Desmonostoc muscorum PUPCCC 405.10 by activating antioxidant defense system. Biologia 78, 3725–3736 (2023). https://doi.org/10.1007/s11756-023-01535-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-023-01535-y