Abstract
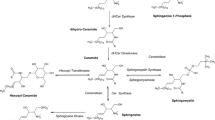
A novel sensitive high-performance liquid chromatography-fluorescence detection (HPLC-FLD) method was developed for real-time monitoring of relative sphingomyelin synthase (SMS) activity based on the measurement of a fluorescent ceramide (Cer) analog and its metabolite, a fluorescent sphingomyelin (CerPCho) analog, in plasma. Analyses were conducted using HPLC-FLD following a protein precipitation procedure. The chromatographic separations were carried out on an Agilent C18 RP column (150 × 4.6 mm, 5 μm) based on a methanol—0.1 % trifluoroacetic acid aqueous solution (88:12, by vol) elution at a flow-rate of 1 mL/min. The limit of quantification in plasma was 0.05 μM for both the fluorescent Cer analog and its metabolite. Significant differences in the fluorescent Cer analog and its metabolite concentration ratio at 5 min were found between vehicle control group and three D2 (a novel SMS inhibitor) dose groups (P < 0.05). Dose-dependent effects (D2 doses: 0, 2.5, 5, 10 mg/kg) were observed. Our method could be used to detect relative SMS activity in biochemical assays and to screen potential SMS inhibitors in vivo. D2 was found to be a potent SMS inhibitor in vivo, and may have a potential antiatherosclerotic effect, which is under further study. D609 was also selected as another model SMS inhibitor to validate our newly developed method.





Similar content being viewed by others
Abbreviations
- AS:
-
Atherosclerosis
- C6-NBD-Cer:
-
6-((N-(7-nitrobenz-2-oxa-1.3-diazol-4-yl) amino) hexanoyl)-sphingosine
- C6-NBD-CerPCho:
-
N-(6-(7-nitro-2-1, 3-benzoxadiazol-4-yl) amino) hexanoyl)-sphingosine -1-phosphocholine
- D2:
-
(2-(2-(benzyloxy)phenyl)-2-(phenylamino) acetonitrile)
- DAG:
-
Diacylglycerol
- HPLC-FLD:
-
High-performance liquid chromatography-fluorescence detection
- PtdCho:
-
Phosphatidylcholine
- CerPCho:
-
Sphingomyelin
- SMS:
-
Sphingomyelin synthase
- D609:
-
Tricyclodecane-9-yl-xanthogenate
References
Xu F, Ji J, Li L, Chen R, Hu W (2007) Activation of adventitial fibroblasts contributes to the early development of atherosclerosis: a novel hypothesis that complements the “Response-to-injury hypothesis” and the “Inflammation hypothesis”. Med Hypotheses 69:908–912
Jiang XC, Paultre F, Pearson TA, Reed RG, Francis CK, Lin M, Berglund L, Tall AR (2000) Plasma sphingomyelin level as a risk factor for coronary artery disease. Arterioscler Thromb Vasc Biol 20:2614–2618
Marathe S, Choi Y, Leventhal AR, Tabas I (2000) Sphingomyelinase converts lipoproteins from apolipoprotein E knockout mice into potent inducers of macrophage foam cell formation. Arterioscler Thromb Vasc Biol 20:2607–2613
Guyton JR, Klemp KF (1996) Development of the lipid-rich core in human atherosclerosis. Arterioscler Thromb Vasc Biol 16:4–11
Schissel SL, Tweedie-Hardman J, Rapp JH, Graham G, Williams KJ, Tabas I (1996) Rabbit aorta and human atherosclerotic lesions hydrolyze the sphingomyelin of retained low-density lipoprotein. Proposed role for arterial-wall sphingomyelinase in subendothelial retention and aggregation of atherogenic lipoproteins. J Clin Invest 98:1455–1464
Hanada K, Hara T, Nishijima M, Kuge O, Dickson RC, Nagiec MM (1997) A mammalian homolog of the yeast LCB1 encodes a component of serine palmitoyltransferase, the enzyme catalyzing the first step in sphingolipid synthesis. J Biol Chem 272:32108–32114
Yeboah J, McNamara C, Jiang XC, Tabas I, Herrington DM, Burke GL, Shea S (2010) Association of plasma sphingomyelin levels and incident coronary heart disease events in an adult population: multi-ethnic study of atherosclerosis. Arterioscler Thromb Vasc Biol 30:628–633
Kinnunen PK, Holopainen JM (2002) Sphingomyelinase activity of LDL: a link between atherosclerosis, Cer, and apoptosis? Trends Cardiovas Med 12:37–42
Li Z, Fan Y, Liu J, Li Y, Huan C, Bui HH, Kuo MS, Park TS, Cao G, Jiang XC (2012) Impact of sphingomyelin synthase 1 deficiency on sphingolipid metabolism and atherosclerosis in mice. Arterioscler Thromb Vasc Biol 32:1577–1584
Liu J, Huan C, Chakraborty M, Zhang H, Lu D, Kuo MS, Cao G, Jiang XC (2009) Macrophage sphingomyelin synthase 2 deficiency decreases atherosclerosis in mice. Circ Res 105:295–303
Liu J, Zhang H, Li Z, Hailemariam TK, Chakraborty M, Jiang K, Qiu D, Bui HH, Peake DA, Kuo MS, Wadgaonkar R, Cao G, Jiang XC (2009) Sphingomyelin synthase 2 is one of the determinants for plasma and liver sphingomyelin levels in mice. Arterioscler Thromb Vasc Biol 29:850–856
Park TS, Panek RL, Mueller SB, Hanselman JC, Rosebury WS, Robertson AW, Kindt EK, Homan R, Karathanasis SK, Rekhter MD (2004) Inhibition of sphingomyelin synthesis reduces atherogenesis in apolipoprotein E-knockout mice. Circulation 110:3465–3471
Dong J, Liu J, Lou B, Li Z, Ye X, Wu M, Jiang XC (2006) Adenovirus-mediated overexpression of sphingomyelin synthases 1 and 2 increases the atherogenic potential in mice. J Lipid Res 47:1307–1314
Ding T, Li Z, Hailemariam T, Mukherjee S, Maxfield FR, Wu MP, Jiang XC (2008) SMS overexpression and knockdown: impact on cellular sphingomyelin and diacylglyCerol metabolism, and cell apoptosis. J Lipid Res 49:376–385
Hailemariam TK, Huan C, Liu J, Li Z, Roman C, Kalbfeisch M, Bui HH, Peake DA, Kuo MS, Cao G, Wadgaonkar R, Jiang XC (2008) Sphingomyelin synthase 2 deficiency attenuates NFkappaB activation. Arterioscler Thromb Vasc Biol 28:1519–1526
Li Z, Hailemariam TK, Zhou H, Li Y, Duckworth DC, Peake DA, Zhang Y, Kuo MS, Cao G, Jiang XC (2007) Inhibition of sphingomyelin synthase (SMS) affects intracellular sphingomyelin accumulation and plasma membrane lipid organization. Biochim Biophys Acta 1771:1186–1194
Liu G, Wang W, Sun G, Ma X, Liu Z, Yang J (2008) Nystatin interferes with the effects of N-methyl-N′-nitro-N-nitrosoguanidine on sphingolipid metabolism in human FL cells. Lipids 43(9):867–875
Tafesse FG, Huitema K, Hermansson M, van der Poel S, van den Dikkenberg J, Uphoff A, Somerharju P, Holthuis JC (2007) Both sphingomyelin synthases SMS1 and SMS2 are required for sphingomyelin homeostasis and growth in human HeLa cells. J Biol Chem 282:17537–17547
Yeang C, Varshney S, Wang R, Zhang Y, Ye D, Jiang XC (2008) The domain responsible for sphingomyelin synthase (SMS) activity. Biochim Biophys Acta 1781:610–617
Luberto C, Hannun YA (1998) Sphingomyelin synthase, a potential regulator of intracellular levels of Cer and diacylglyCerol during SV40 transformation—does sphingomyelin synthase account for the putative, phosphatidylcholine-specific phospholipase C? J Biol Chem 273:14550–14559
Amtmann E (1996) The antiviral, antitumoural xanthate D609 is a competitive inhibitor of phosphatidylcholine-specific phospholipase C. Drugs Exp Clin Res 22:287–294
Tschaikowsky K, Schmidt J, Meisner M (1998) Modulation of mouse endotoxin shock by inhibition of phosphatidylcholine-specific phospholipase C. J Pharmacol Exp Ther 285:800–804
Adibhatla RM, Hatcher JF, Gusain A (2012) Tricyclodecan-9-yl-xanthogenate (D609) mechanism of actions: a mini-review of literature. Neurochem Res 37:671–679
Wang N, Lv X, Su L, Zhao B, Zhang S, Miao J (2006) D609 blocks cell survival and induces apoptosis in neural stem cells. Bioorg Med Chem Lett 16:4780–4783
Schutze S, Potthoff K, Machleidt T, Berkovic D, Wiegmann K, Kronke M (1992) TNF activates NF-κB by phosphatidylcholine-specific phospholipase-C-induced acidic sphingomyelin breakdown. Cell 71:765–776
Gusain A, Hatcher JF, Adibhatla RM, Wesley UV, Dempsey RJ (2012) Anti-proliferative effects of tricyclodecan-9-yl-xanthogenate (D609) involve cer and cell cycle inhibition. Mol Neurobiol 45:455–464
Deng X, Sun H, Gao X, Gong H, Lu W, Chu Y, Zhou L, Ye D (2012) Development, validation, and application of a novel method for mammalian sphingomyelin synthase activity measurement. Anal Lett 45:1581–1589
Deng X, Lin F, Zhang Y, Li Y, Zhou L, Lou B, Li Y, Dong J, Ding T, Jiang X, Wang R, Ye D (2013) Identification of small molecule sphingomyelin synthase inhibitors. Eur J Med Chem 73C:1–7
Kornhuber J, Muehlbacher M, Trapp S, Pechmann S, Friedl A, Reichel M, Muehle C, Terfloth L, Groemer TW, Spitzer GM, Liedl KR, Gulbins E, Tripal P (2011) Identification of novel functional inhibitors of acid sphingomyelinase. PLOS One 6(8):e23852
Lee DH, Kim SH, Ahn KH, Kim SK, Choi JM, Ji JE, Won JH, Park YH, Lim C, Kim S, Kim DK (2011) Identification and evaluation of neutral sphingomyelinase 2 inhibitors. Arch Pharm Res 34:229–236
Wang S-H, Yang W-B, Liu Y-C, Chiu Y-H, Chen C-T, Kao P-F, Lin C-M (2011) A potent sphingomyelinase inhibitor from Cordyceps mycelia contributes its cytoprotective effect against oxidative stress in macrophages. J Lipid Res 52:471–479
Mehta G, Modugu NR (2013) Concise approach to the carbocyclic core of the naturally occurring sphingomyelinase inhibitor scyphostatin. J Org Chem 78:3367–3373
Zama K, Mitsutake S, Watanabe K, Okazaki T, Igarashi Y (2012) A sensitive cell-based method to screen for selective inhibitors of SMS1 or SMS2 using HPLC and a fluorescent substrate. Chem Phys Lipids 165:760–768
Fischbeck A, Krueger M, Blaas N, Humpf H-U (2009) Analysis of sphingomyelin in meat based on hydrophilic interaction liquid chromatography coupled to electrospray ionization-tandem mass spectrometry (HILIC-HPLC-ESI-MS/MS). J Agric Food Chem 57:9469–9474
Acknowledgments
We gratefully acknowledge the financial support from the grant of the Science and Technology Research Key Projects, the Science and Technology Commission of Shanghai Municipality (No.11431920102).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Huang, T., Li, X., Hu, S. et al. Analysis of Fluorescent Ceramide and Sphingomyelin Analogs: A Novel Approach for in Vivo Monitoring of Sphingomyelin Synthase Activity. Lipids 49, 1071–1079 (2014). https://doi.org/10.1007/s11745-014-3940-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-014-3940-5