Abstract
Some persons who smoke have substituted e-cigarettes for tobacco cigarettes, either completely or partially. What effect does this have on cardiovascular functioning? We conducted a living systematic review on human clinical studies measuring the cardiovascular effects of e-cigarette substitution for smoking. The Scopus, PubMed, and CENTRAL Cochrane Library databases were searched on January 31 and April 29, 2021. Three secondary searches and a grey literature search were conducted. Included study designs were randomized controlled trials, quasi-experimental clinical trials, and cohort studies. Risk of bias and study quality were evaluated with the JBI Critical Appraisal tools and the Oxford Catalogue of Bias. The systematic review covered 25 studies comprising 1810 participants who smoked. Twenty studies were rated at high risk of bias, and five as some concerns. A tabular synthesis by direction of effect was conducted due to heterogeneity in the data. Nearly two-thirds of the test analyses indicated that e-cigarette use had no significance difference compared with tobacco cigarettes on heart rate, blood pressure, and in other cardiovascular tests. In two studies, participants with hypertension experienced a clinically relevant reduction in systolic blood pressure after 1 year of e-cigarette use. E-cigarette substitution incurs no additional cardiovascular risks, and some possible benefits may be obtained, but the evidence is of low to very low certainty. An update search on May 30, 2022 retrieved five studies that did not alter our conclusion.
Registration PROSPERO #CRD42021239094.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
More than 1 billion people worldwide are tobacco users, causing more than 7 million deaths annually [1]. For cardiovascular diseases (CVD), smoking is recognized as one of the principal acquired risk factors for atherosclerotic diseases and contributes to the disease burden of aortic aneurysm (34.6%), peripheral artery disease (26.8%), ischemic heart disease (18.41%), and stroke (14.2%) [2]. Two elements of cigarette smoking contribute to causing CVD: the smoke from combustion and nicotine.
Some researchers, particularly those supporting tobacco harm reduction, hold the position that “most of the harm caused by tobacco use is derived from exposure to combustion products of tobacco” [3]. Others disagree, “the relative contributions of nicotine versus non-nicotine components of TC [tobacco cigarette] smoke are unknown” [4]. The effects of inhaled nicotine are difficult to isolate from the smoke constituents (oral nicotine delivery has been studied) and “understanding the role of nicotine in cardiopulmonary disease is extraordinarily difficult” [5]. Research has shown that nicotine activates the sympathetic nervous system, constricting coronary arteries, reducing coronary blood flow reserve, and causing transient increases in heart rate, blood pressure, and myocardial contractability [5,6,7].
Although smoking risks are well known, cigarette smoking remains widespread. Cigarette smoking is hard to quit. The success rate for cessation is abysmally low, approximately 7% at 6 months [8, 9]. Moreover, adequate tobacco cessation services are not available in many countries [10]. Also, often overlooked, are the pleasurable effects of nicotine [11, 12] that contribute to habituation.
Some people who smoke have tried or are using electronic nicotine delivery systems (ENDS), popularly known as e-cigarettes. ENDS consumption has increased noticeably in the few last years [13]. ENDS may function as a substitute for those who are unwilling or unable to quit [14, 15]. The expected benefits come from the significantly lower exposure to toxicants in ENDS vapor compared to cigarette smoke, including the absence of carbon monoxide [3, 4, 7, 16, 17]. Knowing the health effects of ENDS compared to continued smoking is of primary importance for clinicians and users [18].
Our aim was to conduct a living systematic review with evidence from clinical research on human participants to address the question: “What are the cardiovascular health effects resulting from the substitution of ENDS for conventional cigarettes?”.
Methods
Our research question was framed with the PICO (population, intervention, comparator, outcome) model.
-
Population: adults who smoke cigarettes.
-
Intervention: substitution of ENDS for tobacco cigarettes (TC).
-
Comparator: participants who continued to smoke or within-subject changes for participants who substituted ENDS for smoking.
-
Outcomes: measures of cardiovascular function including blood pressure, heart rate, and other cardiovascular tests.
Our review conforms to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) 2020 guidelines [19], see Online Resource 1 PRISMA 2020 Checklist. The protocol was registered with PROSPERO #CRD42021239094 and published in a peer-reviewed journal [20].
The baseline literature search was conducted on January 31, 2021 and updated through April 29, 2021. The databases searched were Scopus, PUBMED, and CENTRAL Cochrane Library with the start date of 2010. The search syntaxes are displayed in Online Resource 2 Search Syntax. Reference lists of systematic and narrative reviews on the cardiovascular effects of ENDS use published from 2018 through 2020 were examined for additional studies. Citation chasing of the included studies was conducted in Google Scholar. A grey literature search checked 41 cardiovascular medical organizations (Online Resource 3 Grey Literature Searches).
An update (“living”) search was conducted on May 22, 2022 with the start date of March 2021 to allow for indexing lag. The update search was conducted in the PubMed and Scopus databases. Newly published systematic reviews were checked for additional studies. The grey literature search of medical organizations was not performed. The study selection processes of title/abstract review and full paper were conducted with the same procedures as the baseline search. The reference lists of included studies were checked for additional studies.
Study designs included were human subject randomized and non-randomized controlled trials, clinical trials, prospective and retrospective cohort studies, and case-controlled studies. The first process was the exclusion of articles based on titles and abstracts to remove in vitro and animal studies, commentary articles, and false retrievals. Details of the exclusion criteria are available in the published protocol [20].
The second process for study inclusion was a full paper review. Three inclusion criteria were applied. One, studies were limited to the research designs listed above. Two, a study was required to have either a comparator group who smoked combustible tobacco (cigarettes) or a within-subject testing of participants who had substituted ENDS for smoking. Third, the study had to report outcome data or analysis from a cardiovascular test. All three criteria had to be satisfied for a study to be included. The inclusion and exclusion of studies was conducted independently by two reviewers, and discrepancies were resolved by discussion. There were no unresolved disagreements for the title/abstract sorting, and for full paper review, one study was decided by the Project Leader (RO), 95% inter-rater agreement. For the update search, there were no unresolved disagreements on the title/abstract sorting or the full paper review.
The data extraction process was conducted independently by two reviewers with a pre-specified data extraction form drawn from the JBI Manual [21] and the Cochrane Collaboration Handbook [22]. (Online Resource 4, Data Extraction Form). When published data were insufficient or missing, the corresponding author was contacted via email.
The quality of studies was assessed by two independent reviewers applying the JBI quality assessment tools [21] and a report of biases drawn from the Oxford Centre for Evidence Based Medicine Catalogue of Bias [23] (Online Resource 5, Bias Report Form). Discrepancies were resolved by discussion; no third person arbitration was required.
The overall rating of the risk of bias for each study was assessed with a rubric that consisted of the JBI score and the biases reports (Online Resource 6, Bias Rating Rubric). Studies were rated as one of three classifications from the Cochrane handbook [22]: low risk of bias, some concerns, and high risk of bias. The rater (RO) was blinded to study outcomes and funders, and the study bias rating was endorsed by the team members.
In addition, the two reviewers independently completed two quality checks for each study. One report noted deviations from protocol (Online Resource 7, Protocol Deviations Report). Another checklist (Online Resource 8, Data discrepancies) identified discrepancies in the reporting of data and statistical significance with the methodology developed by Puljak et al. [24].
All the reports for data extraction and quality assessment are available in the Zenodo data repository: https://zenodo.org/record/4835883#.YPnMqECxUuU.
Per protocol, a tabular synthesis was conducted on test measurements and a narrative summary was compiled of study biases. As anticipated, no meta-analysis was conducted due to the heterogeneity between the studies. These differences across studies included the ENDS nicotine strength and differing ENDS models, wide disparities in study populations in their smoking history and patterns of use, and differing clinical test parameters (acute and widely differing follow-up periods). Furthermore, the high risk of bias in 80% of the studies precluded conducting a meta-analysis [22, 25].
Six analyses were conducted. One sensitivity analyses excluded all studies at high risk of bias. Another analysis was for effect modifications on findings. Three sub-group analyses were conducted for (1) concurrent users of cigarettes (dual users), (2) participants with prior disease conditions, and (3) ENDS use of a duration of 1 year or longer. Finally, the certainty of evidence was evaluated with Grading of Recommendations Assessment, Development and Evaluation (GRADE) [26].
There were a few deviations from our protocol. Due to the length of the review, we excluded the narrative summary of the individual studies. A sensitivity analysis for conflict of interest for industry studies was not conducted, because all industry studies were at high risk of bias. An analysis of effect modifications was added. Because no meta-analysis was conducted, a formal assessment of publication bias could not be performed. The synthesis of the test tabulations as Vote Counting Direction of Effect was added after data collection and before the start of the analysis. This method categorizes the test outcome as showing benefit (improvement in function), harm (decrease in function), or no difference (no significant effect) as a standardized metric that is counted and compared [22].
Results
Study inclusion
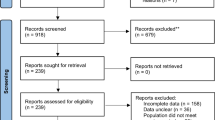
The search results are reported in Fig. 1 PRISMA Search Diagram. Publications excluded at full paper review with the reason for exclusion are listed in Online Resource 9, Excluded Studies.
Our baseline systematic review retrieved 25 studies with 26 publications [27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52]; see Table 1, studies included. No grey literature was found.
The data for the tabular test synthesis and study details are reported in Online Resource 10, Study Evidence Table. The studies were conducted in USA (8), UK (6), Italy (4), Germany (2), and one each from Belgium, Greece, Indonesia, Poland, and South Africa. The participants ranged in age from 18 to 65 comprising 1810 participants who smoked. Four studies were conducted with participants with comorbidities of serious mental illness (1), HIV-positive (1), or hypertension (2). Fourteen studies conducted acute testing; 11 studies presented follow-up data ranging from 5 days to 24 months. Study designs were 14 randomized controlled trials (RCT), 10 quasi-experimental (clinical trials), and 1 cohort study. Twenty studies were rated at high risk of bias, five were rated as some concerns, and no studies were rated as at low risk of bias. The JBI assessment scores, methodological issues, and reporting biases are presented in Online Resource 11, Study Biases.
Five recently published studies were retrieved with the update review; see Fig. 1. One follow-up study reported no significant changes in blood pressure with ENDS use [53], two acute studies found no differences in blood pressure or heart rate between ENDS and TC users [54, 55], and a third acute study reported a higher resting heart rate with tobacco smoking compared to ENDS [56]. The fifth study conducted positron emission tomography/magnetic resonance imaging in matched groups of tobacco users and ENDS users aged 18–30 years; they found no evidence of vascular inflammation [57]. As per protocol, because the findings of these studies did not alter our conclusions, the studies will be incorporated into the updated version of the living systematic review planned for the end of 2024.
Tabular synthesis of cardiovascular tests
Twenty-two studies tested heart rate (HR), 12 with acute testing and 10 with follow-up (one study data not reported [38]). Acute testing both within-subject and comparing ENDS vs. TC yielded mixed findings; see Fig. 2. Eight acute studies found no significant increase in HR with ENDS use within subject [43, 46, 48, 50] or compared to TC [32, 36, 48, 52]. Three acute tests found a significant increase in HR for nicotine ENDS [39, 41, 43], but in one test, the increase was not significantly different than TC [39], and in another, it was a significantly lower increase than TC [41]. One additional acute study finding an increase in HR was seriously compromised due to excessive ENDS exposure [42]. One study reported that acute non-nicotine ENDS use significantly raised HR compared to sham vaping; it conducted excessive ENDS exposure [30]. Another study on non-nicotine ENDS found that TC significantly raised HR in comparison [45].
Eight of the nine follow-up studies demonstrated no significant changes in HR [31, 33,34,35, 44, 47, 49, 51], while one study reported differing outcomes based on the participants’ pack year smoking history [37]; see Fig. 3.
Nineteen studies tested blood pressure (BP), 8 acute studies and 11 follow-up studies. Fifteen studies indicated no significant changes [31,32,33,34, 36,37,38, 40, 41, 45,46,47, 49,50,51]. One acute test found a significant increase in BP, but the increase was not significantly different from TC, and ENDS increases were lower than TC [27]. Two studies showed that TC significantly increased BP, while ENDS did not [41, 45]. One study with five ENDS models found that some significantly increased BP, while others did not, but the finding is compromised due to excessive ENDS exposure [52]. Two follow-up studies found a significant decrease in BP with ENDS use [35, 44]; see Fig. 4.
Four studies assessed the impact of ENDS on arterial stiffness with flow-mediated dilation (FMD) tests. Two acute studies reported significant declines for within-subject measurements, but the difference between ENDS and TC was not significant and the reduction with ENDS was less than TC [27, 28]. One acute study [42] found significant decreases within subject, but the findings were compromised due to excessive ENDS exposure. A fourth study with a 4-week follow-up calculated that females but not males using ENDS had a smaller decrease than participants continuing smoking [37].
Electrocardiogram (ECG) 12-lead testing was conducted in three studies. No significant findings were reported in acute testing [50] or in the follow-up studies [33, 51].
Analyses
A sensitivity analysis was made with the five studies at some risk of bias. Their tests results were in much the same ratios as the studies at high risk of bias.
An assessment of effect modification was performed for age, gender, and smoking history. Only one study had a significant result for females but not males, and observed differing results based on participants’ smoking history. No effect modifications were observed in any other studies.
Three sub-group analyses were calculated. For dual users, only one study compared dual users with exclusive ENDS users; finding that exclusive users had significantly better improvements in HR and BP than dual users [44]. For participants with comorbidities, no significant cardiovascular changes were reported for those with HIV [31] or participants with serious mental illnesses [38]. Only two studies were conducted with participants with prior cardiovascular disease (discussed below). All other studies specifically excluded participants with prior cardiovascular diseases or symptoms. The third analysis was on findings of ENDS use of 1 year or longer. Two studies reported no modification on HR [35, 44], and one study reported no significant changes in either BP or HR [51].
Two studies that measured BP after 1 year of ENDS substitution found clinically relevant reductions in participants with hypertension. Farsalinos et al. [35] conducted a sub-group analysis of 66 participants with elevated BP at baseline. Their average baseline systolic BP 141.2 dropped to 132.4 (p < 0.001), plus they had a significant but small decrease in diastolic BP. A clinically significant reduction in BP was observed by Polosa et al. [44] in a cohort of 43 participants with hypertension. After 1 year, participants using ENDS exclusively experienced a reduction in systolic BP from 140 to 130; dual users also had significant reductions.
The final analysis evaluated the overall confidence in the evidence with the GRADE rating. It was assessed as low to very low. Twelve of the clinical trials were lowered from high certainly to low due to the high risk of bias. Eight clinical trials were lowered from high to very low for high risk of bias plus either imprecision for excessive ENDS exposure or indirectness for testing with discontinued early ENDS models. The one observational study was lowered from low to very low for some concerns of bias. For the four studies at the moderate rating, three had no significant findings and one study had significant findings only for a sub-group. No studies were at low risk of bias; therefore, none were eligible for a higher rating. See Online Resources Document 12, GRADE rating.
Discussion
We found discrepant results regarding the effects of ENDS substitution on cardiovascular outcomes. The findings had no indications that ENDS use is more harmful than smoking. In 14 studies, ENDS substitution induced no significant changes in HR. In the five studies that recorded in a slight increase in HR, one study found that the increase was lower with ENDS compared to tobacco smoking, and in two studies, the increase was not significantly different between them. In 15 studies, ENDS use had no significant effect on BP, and a beneficial effect in three. Of the 55 tests extracted from the studies, 36 tests, just under two-thirds did not show significant changes in cardiovascular function with ENDS use. The GRADE quality of evidence is low to very low.
Study quality issues: methodology
We identified substantive problems with the research designs of many studies.
Blinding was not clear or absent in many studies. Although blinding participants, testers (clinical testers), and assessors (data analysts) is the standard, blinding participants is not always possible in ENDS clinical research. Participants certainly see the difference between ENDS and cigarettes or the lack of vapor with sham vaping. Nevertheless, nicotine levels can be masked. In the 14 RCTs, seven did not specify the blinding of the treaters and nine did not report blinding of assessors.
Ascertainment bias was common in the studies. It can be introduced when participants in a clinical trial differ from the target population [58]. For gender, males were frequently dominant in the participant populations; three studies were conducted with only male participants, and nine studies had a substantially higher percentage of male participants. Conversely, one study had all female participants and three had a substantially higher percentage of female participants. Patterns of cigarette use by participants varied. Three studies included participants smoking less than 10 cigarettes a day, and fourteen studies included participants smoking more than 20 cigarettes a day (heavy smoking). Only one study included smoking history in their analysis, and half the studies did not report the smoking history of the participants. For studies on cardiovascular functioning, the age of participants is critical; the age for CVD risk is ≥ 40 years [59]. Four studies included participants exclusively over age 40; nine studies included participants over 40 but with an average age under 40; and six studies had no participants over age 40. The divergent factors of gender, age, and patterns of cigarette use limit the generalizability of the findings.
Different ENDS models are an issue with evidence from early studies. ENDS models are continuously evolving, so findings based on the first devices may not be applicable to current ones [60]. Five studies were conducted with the now discontinued “cig-a-like” models; they complicate the generalizability of study findings [61].
Finally, bias occurs when acute studies are conducted with vaping protocols that do not replicate how individuals use ENDS in real life [62]. The standard laboratory exposure is 15 puffs in 10 min based on the widely accepted Penn State Electronic Cigarette Dependence Index [63]. An excessive exposure protocol was conducted in three studies.
Study quality issues: reporting
Omissions and errors in reporting in the studies led to serious concerns for potential bias (see Online Document 11, Bias table).
Changes from the protocol of a study should be disclosed. Substantial deviations from protocol may be an indicator of data dredging [64]. Thirteen studies either did not report a protocol or the protocol was not published, thereby limiting an examination for missing data [65, 66] or the identification of changes in the designation of primary and secondary outcomes [67, 68]. Of the 12 studies with a published protocol, nine had deviations that were not acknowledged in the published study; two of which had substantial deviations.
Reporting errors occur when there are discrepancies within the article between the data reported in the abstract, discussion, tables, figures, or conclusions [24]. Ten publications had data discrepancies. These errors distorted the presentation of data, including in the conclusions.
Bias may occur from how findings are reported. In nine studies, their discussions and conclusions were presented with spin bias. This occurs when authors, intentionally or unintentionally, emphasize p values [69] or secondary endpoints without clinical relevance [70, 71]. Some studies failed to discuss all their data or framed non-significant tests as substantive evidence. Furthermore, authors engaged in one-sided reference bias or “all’s well” literature bias by citing only studies that supported their results while omitting those opposing them [72].
Finally, studies that fail to find any significant findings may push researchers into spin bias with speculations, forgetting that non-significant results are important research findings [73]. In our review, demonstrating that ENDS have no significant difference in cardiovascular effects compared to TC is highly relevant.
Comparison with other systematic reviews
Other systematic reviews have included data from study designs from in vitro, animal, biomarkers, and cross-sectional surveys. We excluded these study designs, because direct evidence for human health effects cannot be obtained from them. First of all, cross-sectional studies cannot prove causation, because the temporal sequence of ENDS use and disease outcomes cannot be established. In vitro studies “still do not fully represent complex in vivo systems and may not directly translate to adverse effects relevant to disease outcomes” [60; see also 74]. Plus, the predictive capacity of in vitro testing is unknown [60]. In animal tests on ENDS (almost all with rats), exposures are performed with intra-tracheally or nasally administered liquids, or whole-body aerosol exposure, protocols that do not reflect human exposure levels [75]. Finally, biomarkers are surrogate outcomes [76]. Their role is prominent in screening but remains limited in indicating disease [77]. Furthermore, the predictive value of biomarkers for tobacco-related diseases has yet to be verified [78, 79].
Bearing in mind the critical limitations of these study designs, we compared our findings with the five recent systematic reviews on cardiovascular outcomes.
Martinez-Morata, Sanchez, Shimbo and Navas-Acien [6] with 13 studies, concluded that ENDS may cause elevations in BP. Their evidence included ENDS use by people who do not smoke, so do not reflect substitution effects. No quality appraisal was performed.
Kennedy, van Schalkwyk, McKee and Pisinger [80] analysed 38 studies, including 6 animal and 8 in vitro studies. The authors excluded studies which they deemed to have a conflict of interest. They concluded that 90% of the studies found potentially harmful effects on the cardiovascular system. Yet, 6 of 11 studies at moderate–high risk of bias had conclusions that were supportive of ENDS use, because their negative effects were lower than TC. Their assessments of issues with the randomization and blinding in studies were very similar to ours.
The Goniewicz et al.’s [81] meta-analysis was calculated based on three cross-sectional population studies on myocardial infarction, coronary heart disease, and stroke. They concluded that there were no significant differences in cardiovascular outcomes between former smokers who transitioned to ENDS compared to current exclusive smokers.
Two systematic reviews conducted a meta-analysis of BP and HR outcomes. Garcia et al. [62] examined 19 studies and presented conclusions similar to ours that acute ENDS exposure increases HR and BP less than TC. Similar findings were reported by Skotsimara et al. [82] with a meta-analysis of 14 studies, although their conclusion focused on negative effects. Both reviews contained some concerns of imprecision, did not identify clinically relevant results, and neither conducted a quality assessment.
Future research
Initial findings in this systematic review show promise for harm reduction with ENDS substitution for smoking for persons with hypertension. A reduction of 10 mmHg in SBP significantly reduces the risk for major cardiovascular events with a relative risk of 0.80 (95% CI 0.77–0.83) [83]. Two studies, N = 109, rated at some concerns of bias found a mean reduction ~ 10 mmHg for participants with hypertension who switched to ENDS for 1 year. This potential beneficial effect deserves further study with clinical testing and medical record cohort studies, preferably longer term.
Based on this review, we offer these recommendations for future ENDS research:
-
Reconsider the value of acute testing. “Acute studies of effects of ENDS in humans may not reflect long term effects” [84]. Nonetheless, for RCTs conducting acute ENDS exposure, administer a validated puff protocol such as the Pennsylvania University standard [63].
-
Carry out blinding procedures where feasible. A good example is the study by Veldheer et al. [49] for its processes for nicotine content labeling and participant identification.
-
Account for the ascertainment bias factors specific to smoking: smoking history which includes quit attempts and patterns of use which includes dual use. These participant profiles should be considered for both recruiting and in analyses.
-
Eliminate reporting errors. Data discrepancies in a study article should be rooted out by the authors, or failing that, peer reviewers.
-
Clearly differentiate in the Discussion and conclusions between statistical significance and clinical relevance, because a numerical change in a clinical test measurement may not indicate a change in health status. For example, a change in heart rate from a baseline reading of 65.7 (± 7.7) to 71.6 (± 8.1) 5 min after ENDS use is not of clinical relevance but generates a p < 0.05 [study 39].
-
And a respectful reminder: publishing a protocol is a must for transparency and confidence.
Limitations
The studies retrieved for the review have many limitations. The large majority, 80%, were rated at high risk of bias, and no studies were at low risk. Nearly half of the studies were conducted with small sample sizes. These and other issues resulted in the GRADE assessment of the evidence to be at low to very low certainty. In addition, acute effects contribute little to understanding health outcomes, nor do significant p values indicate clinically relevant outcomes. Finally, there were few long-term studies.
For the systematic review, our analysis method, vote counting of direction of effect, is limited. It does not provide data on the magnitude of the effect size, nor of its clinical relevance. This method does not represent the relative sizes of the studies, and many of the studies had a small number of participants [22].
To achieve the highest quality, this review is PRISMA 2020 compliant [19] and meets all AMSTAR2 standards [85]. Unfortunately, the most rigorous methodology cannot overcome the limitations caused by the poor quality of the available studies.
Conclusions
The large majority of studies, nearly two-thirds, found no significant changes in heart rate or blood pressure with ENDS substitution for smoking. Where ENDS use increased the heart rate, in most cases the increase was lower compared to smoking. One-year use of ENDS resulted in improvements in hypertension for 109 participants in two studies. Our conclusion is that ENDS substitution is not more harmful to health than continued smoking and may have some limited benefits. Our confidence in these conclusions is low to very low.
Furthermore, based on our quality and bias assessments, the current research needs to be read with a jaundiced eye. For future studies, we urge researchers to pay close attention to their research designs and reporting of data. More quality evidence is certainly needed to inform if ENDS substitution is a worthwhile option for harm reduction for persons who smoke.
Data availability
All data generated or analysed during this review and included in the published article and Online Resources.
Code availability
Not applicable.
References
World Health Organization (2020) Tobacco. https://www.who.int/news-room/fact-sheets/detail/tobacco. Accessed 16 Dec 2020
Institute for Health Metrics and Evaluation (2020) GBD compare Viz Hub (2020). https://vizhub.healthdata.org/gbd-compare/
Benowitz NL, Fraiman JB (2017) Cardiovascular effects of electronic cigarettes. Nat Rev Cardiol 14:447–456. https://doi.org/10.1038/ncardio.2017.36
MacDonald A, Middlekauff HR (2019) Electronic cigarettes and cardiovascular health: what do we know so far? Vasc Health Risk Manag 15:159–174. https://doi.org/10.2147/VHRM.S175970
Tarran R, Barr RG, Benowitz NL, Bhatnagar A, Chu HW, Dalton P et al (2021) E-cigarettes and cardiopulmonary health. Functon 2(2):zqab004. https://doi.org/10.1093/function/zqab004
Martinez-Morata I, Sanchez TR, Shimbo D, Navas-Acien A (2020) Electronic cigarette use and blood pressure endpoints: a systematic review. Curr Hypertens Rep 23(1):2. https://doi.org/10.1007/s11906-020-01119-0
Middlekauff HR, Park J, Moheimani RS (2014) Adverse effects of cigarette and noncigarette smoke exposure on the autonomic nervous system: mechanisms and implications for cardiovascular risk. J Am Coll Cardiol 64(16):1740–1750. https://doi.org/10.1016/j.jacc.2014.06.1201
Babb S, Malarcher A, Schauer G, Asman K, Jamal A (2017) Quitting smoking among adults—United States, 2000–2015. Morb Mortal Wkly Rep 65(52):1457–1464. https://doi.org/10.15585/mmwr.mm6552a1
United States Department of Health and Human Services (2020) Smoking cessation: a report of the Surgeon General. US Department of Health and Human Services, Washington DC
World Health Organization (2019) WHO report on the global tobacco epidemic, 2019: offer help to quit tobacco use. WHO, Geneva
Benowitz NL (2010) Nicotine addiction. N Engl J Med 362(24):2295–2303. https://doi.org/10.1056/NEJMra0809890
Niaura R (2016) Re-thinking nicotine and its effects. Truth Initiative. http://truthinitiative.org/sites/default/files/ReThinking-Nicotine.pdf. Accessed 8 Feb 2021
Jones L (15 September 2019) Vaping: how popular are e-cigarettes? BBC News. https://www.bbc.com/news/business-44295336. Accessed 2 Feb 2021
Green SH, Bayer R, Fairchild AL (2016) Evidence, policy, and e-cigarettes—will England reframe the debate? NEMJ 374(14):1301–1303. https://doi.org/10.1056/NEJMp1601154
McNeill A, Brose LS, Calder R, Bauld L, Robson D (2018) Evidence review of e-cigarettes and heated tobacco products 2018. A report commissioned by Public Health England. PHE, London
Goniewicz ML, Smith DM, Edwards KC, Blount BC, Cladwell KL, Feng J, Wang L, Christensen C, Ambrose B, Borek N et al (2018) Comparison of nicotine and toxicant exposure in users of electronic cigarettes and combustible cigarettes. JAMA Netw Open 1(8):e185937. https://doi.org/10.1001/jamanetworkopen.2018.5937
National Academies of Sciences Engineering and Medicine (2018) Public health consequences of ecigarettes. National Academies Press, Washington DC
Fairchild AL, Bayer R, Lee JS (2019) The e-cigarette debate: what counts as evidence? Am J Public Health 109(7):1000–1006. https://doi.org/10.2105/AJPH.2019.305107
Page MJ, McKenzie JE, Bossuyt PM, Boutron J, Hoffman TC, Mulrow CD, Shamseer L, Tetlaff JM, Akl EA, Brennan SE et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/bmj.n71
O’Leary R, Qureshi MA, La Rosa GRM, Vernooij RWM, Odimegwu DC, Bertino G, Polosa R (2021) Respiratory and cardiovascular health effects of e-cigarette substitution: protocol for two living systematic reviews. JMIR Res Protoc 10(5):e29084. https://doi.org/10.2196/29084
Aromataris E, Munn Z (eds) (2020) JBI manual for evidence synthesis. JBI Global. https://wiki.jbi.global/display/MANUAL/Downloadable+PDF+-+current+version. Accessed 5 Jul 2021
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (eds) (2020) Cochrane handbook for systematic reviews of interventions version 6.1. Cochrane Collaborative, London
Centre for Evidence Based Medicine (2019) Catalogue of bias. https://catalogofbias.org/. Accessed 8 Feb 2021
Puljak L, Riva N, Parmelli E, González-Lorenzo M, Moja L, Pieper D (2020) Data extraction methods: an analysis of internal reporting discrepancies in single manuscripts and practical advice. J Clin Epidemiol 117:158–164. https://doi.org/10.1016/j.clinepi.2019.09.003
Johnson BT, Hennessy EA (2019) Systematic reviews and meta-analyses in the health sciences: best practice methods for research synthesis. Soc Sci Med 233:237–251. https://doi.org/10.1016/j.socscimed.2019.05.035
Guyatt G, Oxman AD, Sultan S, Glasziou P, Akl EA, Alonso-Coelle P, Kunz R, Brozek J, Montori V, Jaeschke R et al (2011) GRADE guidelines: 9. Rating up the quality of evidence. J Clin Epidemiol 64(12):1311–1316. https://doi.org/10.1016/j.jclinepi.2011.06.004
Biondi-Zoccai G, Sciarretta S, Bullen C, Nocella C, Violi F, Loffredo L, Pignatelli P, Perri L, Peruzzi M, Marullo AGM et al (2019) Acute effects of heat-not-burn, electronic vaping, and traditional tobacco combustion cigarettes: the Sapienza University of Rome-Vascular Assessment of Proatherosclerotic Effects of Smoking (SUR-VAPES) 2 randomized trial. J Am Heart Assoc 8(6):e010455. https://doi.org/10.1161/JAHA.118.010455
Carnevale R, Sciarretta S, Violi F, Nocella C, Loffredo L, Perri L, Peruzzi M, De Falco E, Chimenti I, Valenti V, Biondi-Zoccai G, Frati G (2016) Acute impact of tobacco vs electronic cigarette smoking on oxidative stress and vascular function. Chest 150(3):606–612. https://doi.org/10.1016/j.chest.2016.04.012
Mastrangeli S, Carnevale R, Cavarretta E, Sciarette S, Peruzzi M, Marullo AGM, De Falco E, Chimenti I, Valenti V, Bellen C, Roever L, Frati G, Biondi-Zoccai G (2018) Predictors of oxidative stress and vascular function in an experimental study of tobacco versus electronic cigarettes: a post hoc analysis of the SUR-VAPES 1 study. Tob Induc Dis 16:18. https://doi.org/10.18332/tid/89975
Chaumont M, van de Borne P, Bernard A, Van Muylem A, Ddeprez G, Ullmo J, Starczewska E, Briki R, de Hemptinne Q, Zaher W, Debbas N (2019) Fourth generation e-cigarette vaping induces transient lung inflammation and gas exchange disturbances: results from two randomized clinical trials. Am J Physiol Lung Moi Physiol 316(5):L705–L719. https://doi.org/10.1152/ajplung.00492.2018
Cioe PA, Mercurio AN, Lechner W, Costantino CC, Tidy JW, Eissenberg T, Kahler CW (2020) A pilot study to examine the acceptability and health effects of electronic cigarettes in HIV-positive smokers. Drug Alcohol Depend 206:107678. https://doi.org/10.1016/j.drugalcdep.2019.107678
Cobb CO, Lopez AA, Soule EK, Yen MS, Rumsey H, Lester Scholtes R, Rudy AK, Lipato T, Guy M, Eissenberg T (2019) Influence of electronic cigarette liquid flavors and nicotine concentration on subjective measures of abuse liability in young adult cigarette smokers. Drug Alcohol Depend 203:27–34. https://doi.org/10.1016/j.drugalcdep.2019.05.024
Cravo AS, Bush J, Sharma G, Savioz R, Martin C, Craige S, Walele T (2016) A randomised, parallel group study to evaluate the safety profile of an electronic vapour product over 12 weeks. Regul Toxicol Pharmacol 81(Suppl 1):S1–S14. https://doi.org/10.1016/j.yrtph.2016.10.003
D’Ruiz CD, O’Connell G, Graff DW, Yan XS (2017) Measurement of cardiovascular and pulmonary function endpoints and other physiological effects following partial or complete substitution of cigarettes with electronic cigarettes in adult smokers. Regul Toxicol Pharmacol 87:36–53. https://doi.org/10.1016/j.yrtph.2017.05.002
Farsalinos K, Cibella F, Caponnetto P, Campagna D, Morjaria JB, Battaglia E, Caruso M, Russo C, Polosa R (2016) Effect of continuous smoking reduction and abstinence on blood pressure and heart rate in smokers switching to electronic cigarettes. Intern Emerg Med 11(1):85–94. https://doi.org/10.1007/s11739-015-1361-y
Franzen KF, Willig J, Cayo Talavera S, Meusel M, Sayk F, Reppel M, Dalhoff K, Martensen K, Droemann D (2018) E-cigarettes and cigarettes worsen peripheral and central hemodynamics as well as arterial stiffness: a randomized, double-blinded pilot study. Vasc Med 23(5):419–425. https://doi.org/10.1177/1358863X18779694
George J, Hussain M, Vadiveloo T, Ireland S, Hopkinson P, Struthers AD, Donnan PT, Kahn F, Lang CC (2019) Cardiovascular effects of switching from tobacco cigarettes to electronic cigarettes. J Am Coll Cardiol 74(25):3112–3120. https://doi.org/10.1016/j.jacc.2019.09.067
Hickling LM, Perez-Iglesias R, McNeill A, Dawkins L, Moxham J, Ruffell T, Sendt KV, McGuire P (2019) A pre-post pilot study of electronic cigarettes to reduce smoking in people with severe mental illness. Psychol Med 49(6):1033–1040. https://doi.org/10.1017/s0033291718001782
Hiler M, Breland A, Spindle T, Maloney S, Lipato T, Karaoghlanian N, Shihadeh A, Lopez A, Ramôa C, Eisenberg T (2017) Electronic cigarette user plasma nicotine concentration, puff topography, heart rate, and subjective effects: influence of liquid nicotine concentration and user experience. Exp Clin Psychopharmacol 25(5):380–392. https://doi.org/10.1037/pha0000140
Ikonomidis I, Katogiannis K, Kostelli G, Kourea K, Kyriahou A, Tsoumani M, Andreadou I, Lambadiari V, Plotas P, Thymis I, Tsantes AE (2020) Effects of electronic cigarette on platelet and vascular function after four months of use. Food Chem Toxicol 141:111389. https://doi.org/10.1016/j.fct.2020.111389
Kerr DMI, Brooksbank KJM, Taylor RG, Pinel K, Rios FJ, Touyz RM, Delles C (2019) Acute effects of electronic and tobacco cigarettes on vascular and respiratory function in healthy volunteers: a cross-over study. J Hypertens 37(1):154–166. https://doi.org/10.1097/HJH.0000000000001890
Kuntic M, Oelze M, Steven S, Krőller-Schőn S, Stamm P, Kalinovic S, Frenis K, Vujacic-Mirski K, Jimenez MTB, Kvandova M et al (2020) Short-term e-cigarette vapour exposure causes vascular oxidative stress and dysfunction: evidence for a close connection to brain damage and a key role of the phagocytic NADPH oxidase (NOX-2). Eur Heart J 41(26):2472–2483. https://doi.org/10.1093/eurheartj/ehz772
Nides MA, Leischow SJ, Bhatter M, Simmons M (2014) Nicotine blood levels and short-term smoking reduction with an electronic nicotine delivery system. Am J Health Behav 38(2):265–274. https://doi.org/10.5993/AJHB.38.2.12
Polosa R, Morjaria JB, Caponnetto P, Battaglia E, Russo C, Ciampi C, Adams G, Bruno C (2016) Blood pressure control in smokers with arterial hypertension who switched to electronic cigarettes. Int J Environ Res Public Health 13(11):1123. https://doi.org/10.3390/ijerph13111123
Sumartiningsih S, Lin HF, Lin JC (2019) Cigarette smoking blunts exercise-induced heart rate response among young adult male smokers. Int J Environ Res Public Health 16(6):1032. https://doi.org/10.3390/ijerph16061032
Szołtysek-Bołdys I, Sobczak A, Zielińska-Danch W, Bartoń A, Koszowski B, Kośmider L (2014) Influence of inhaled nicotine source on arterial stiffness. Przegla̧d Lekarski 71(11):572–575
van Staden SR, Groenewald M, Engelbrecht R, Becker PJ, Hazelhurst LT (2013) Carboxyhaemoglobin levels, health and lifestyle perceptions in smokers converting from tobacco cigarettes to electronic cigarettes. S Afr Med J 103(11):865–868. https://doi.org/10.7196/samj.6887
Vansickel AR, Cobb CO, Weaver MF, Eissenberg TE (2010) A clinical laboratory model for evaluating the acute effects of electronic “cigarettes”: nicotine delivery profile and cardiovascular and subjective effects. Cancer Epidemiol Biomark Prev 19(8):1945–1953. https://doi.org/10.1158/1055-9965.EPI-10-0288
Veldheer S, Yingst J, Midya V, Hummer B, Lester C, Krebs N, Hrabovsky S, Wilherm A, Liao J, Yen MS, Cobb C, Eissenberg T, Foulds J (2019) Pulmonary and other health effects of electronic cigarette use among adult smokers participating in a randomized controlled smoking reduction trial. Addict Behav 91:95–101. https://doi.org/10.1016/j.addbeh.2018.10.041
Walele T, Sharma G, Savioz R, Martin C, Williams J (2016) A randomised, crossover study on an electronic vapour product, a nicotine inhalator and a conventional cigarette. Part B: Safety and subjective effects. Regul Toxicol Pharmacol 74:193–199. https://doi.org/10.1016/j.yrtph.2015.12.004
Walele T, Bush J, Koch A, Savioz R, Martin C, O’Connell G (2018) Evaluation of the safety profile of an electronic vapour product used for two years by smokers in a real-life setting. Regul Toxicol Pharmacol 92:226–238. https://doi.org/10.1016/j.yrtph.2017.12.010
Yan XS, D’Ruiz C (2015) Effects of using electronic cigarettes on nicotine delivery and cardiovascular function in comparison with regular cigarettes. Regul Toxicol Pharmacol 71(1):24–34. https://doi.org/10.1016/j.yrtph.2014.11.004
Arnold MJ, Nollen NL, Mayo MS, Ahluwalia JS, Leavens EL, Zhang G, Rice M, Pulvers K (2021) Harm reduction associated with dual use of cigarettes and e-cigarettes in Black and Latino smokers: secondary analyses from a randomized controlled e-cigarette switching trial. Nicotine Tob Res 23(11):1972–1976. https://doi.org/10.1093/ntr/ntab069
Dimitriadis K, Narkiewicz K, Leontsinis I, Konstantinidid D, Mihas C, Andrikou I, Thomopoulos C, Tousoulis D, Tsioufis K (2022) Acute effects of electronic and tobacco cigarette smoking on sympathetic nerve activity and blood pressure in humans. Int J Environ Res Public Health 19(6):3237. https://doi.org/10.3390/ijerph19063237
Gernun S, Franzen KF, Mallock N, Benthien J, Luch A, Mortensen K, Drőmann D, Pogarell O, Rüther T, Rabenstein A (2022) Cardiovascular functions and arterial stiffness after JUUL use. Tob Induc Dis 20:34. https://doi.org/10.18332/tid/144317
Sumartiningsih S, Rahayu S, Handoyo E, Lin J-C, Lim CL, Starczewski M, Fuchs PX, Kus C-H (2022) Systemic lactate elevation induced by tobacco smoking during rest and exercise is not associated with nicotine. Int J Environ Res Public Health 19(5):2902. https://doi.org/10.3390/ijerph19052902
Sahota A, Naidu S, Jacobi A, Giannarelli C, Woodward M, Fayad ZA, Mani V (2021) Atherosclerosis inflammation and burden in young adult smokers and vapers measured by PET/MR. Atherosclerosis 325:110–116. https://doi.org/10.1016/j.atherosclerosis.2021.03.021
Spencer EA, Brassey J (2017) Ascertainment bias. Catalogue of bias. https://catalogofbias.org/biases/ascertainment-bias/. Accessed 12 Jan 2021
Rodgers JL, Jones J, Bolleddu SI, Vanthenapalli S, Rodgers LE, Shah K, Karia K, Panguluri SK (2019) Cardiovascular risks associated with gender and aging. J Cardiovasc Dev Dis 6(2):19. https://doi.org/10.3390/jcdd6020019
Lauterstein D, Savidge M, Chen Y, Weil R, Yeager RP (2020) Nonanimal toxicology testing approaches for traditional and deemed tobacco products in a complex regulatory environment: limitations, possibilities, and future directions. Toxicol In Vitro 62:104684. https://doi.org/10.1016/j.tiv.2019.104684
Traboulsi H, Cherian M, Abou Rjeili M, Preterooti M, Bourbeau J, Smith BM, Eidelman DH, Baglole CJ (2020) Inhalation toxicology of vaping products and implications for pulmonary health. Int J Mol Sci 21(10):3495. https://doi.org/10.3390/ijms21103495
Garcia PD, Gornbein JA, Middlekauff HR (2020) Cardiovascular autonomic effects of electronic cigarette use: a systematic review. Clin Auton Res 30(6):507–519. https://doi.org/10.1007/s10286-020-00683-4
Penn State College of Medicine (2015) Penn State Nicotine Dependence Index. Center for Research on Tobacco and Health. https://research.med.psu.edu/smoking/dependence-index/. Accessed 29 Nov 2021
Erasmus A, Holman B, Ioannidis J (2020) Data-dredging bias. Catalogue of bias. https://catalogofbias.org/biases/data-dredgingbias/. Accessed 29 Nov 2021
Richards G, Onakpoya I (2019) Reporting biases. Catalogue of bias. http://www.catalogueofbiases.org/reportingbiases. Accessed 12 Jan 2021
Ayorinde AA, Williams I, Mannion R, Song F, Skrybant M, Lilford RJ, Chen Y-F (2020) Assessment of publication bias and outcome reporting bias in systematic reviews of health services and delivery research: a meta-epidemiological study. PLoS ONE 15(1):e0227580. https://doi.org/10.1371/journal.pone.0227580
Jones CW, Keil LG, Holland WC, Caughey MC, Platts-Mills TF (2015) Comparison of registered and published outcomes in randomized controlled trials: a systematic review. BMC Med 13:282. https://doi.org/10.1186/s12916-015-0520-3
Jonsson U, Alaie I, Parling T, Arnberg FK (2014) Reporting of harms in randomized controlled trials of psychological interventions for mental and behavioral disorders: a review of current practice. Contemp Clin Trials 38(1):1–8. https://doi.org/10.1016/j.cct.2014.02.005
Turrentine M (2017) It’s all how you “spin” it: interpretive bias in research findings in the obstetrics and gynecology literature. Obstet Gynecol 129(2):239–242. https://doi.org/10.1097/aog.0000000000001818
Heneghan C, Spencer EA (2017) Biases of rhetoric. Catalogue of bias. https://catalogofbias.org/biases/biasesofrhetoric. Accessed 12 Jan 2021
Mahtani KR, Chalmers I, Nunan D (2019) Spin bias. Catalogue of bias. https://catalogofbias.org/biases/spin-bias/. Accessed 12 Jan 2021
Spencer EA, Brassey J, Heneghan C (2017) One-sided reference bias. Catalogue of bias. https://www.catalogofbias.org/biases/one-sided-reference-bias. Accessed 12 Jan 2021
Meerpohl JJ, Schell LK, Bassler D, Gallus S, Kleijnen J, Kulig M, La Vecchia C, Maršic A, Ravaud P, Reis A (2015) Evidence-informed recommendations to reduce dissemination bias in clinical research: conclusions from the OPEN (Overcome failure to Publish nEgative fiNdings) project based on an international consensus meeting. BMJ Open 5(5):e006666. https://doi.org/10.1136/bmjopen-2014-006666
Benowitz NL, Burbank AD (2016) Cardiovascular toxicity of nicotine: implications for electronic cigarette use. Trends Cardiovasc Med 26(6):515–523. https://doi.org/10.1016/j.tcm.2016.03.001
Wang G, Liu W, Song W (2019) Toxicity assessment of electronic cigarettes. Inhal Toxicol 31(7):259–273. https://doi.org/10.1080/08958378.2019.1671558
Boyd CM, Singh S, Varadhan R, Weiss CO, Sharma R, Bass EB (2012) Puhan MA (2012) Methods for benefit and harm assessment in systematic reviews. Agency for Healthcare Quality and Research, Washington DC
Institute of Medicine (2012) Scientific standards for studies on modified risk tobacco products. The National Academies Press, Washington DC
Chang CM, Cheng YC, Cho TM, Mishina EV, Del Valle-Pinero AY, van Bermmel DM, Hatsukami DK (2019) Biomarkers of potential harm: summary of an FDA-sponsored public workshop. Nicotine Tob Res 21(1):3–13. https://doi.org/10.1093/ntr/ntx273
Hu Q, Hou H (2015) Tobacco smoke exposure biomarkers. CRC Press, Boca Raton
Kennedy CD, van Schalkwyk MCI, McKee M, Pisinger C (2019) The cardiovascular effects of electronic cigarettes: a systematic review of experimental studies. Prev Med 127:105770. https://doi.org/10.1016/j.ypmed.2019.105770
Goniewicz ML, Miller CR, Sutanto E, Li D (2020) How effective are electronic cigarettes for reducing respiratory and cardiovascular risk in smokers? A systematic review. Harm Reduct J 17(1):91. https://doi.org/10.1186/s12954-020-00440-w
Skotsimara G, Antonopoulos AS, Oikonomou E, Siasos G, Ioakeimidis N, Tsalamandris S, Charalambour G, Galiatsatos N, Vlachopoulos C, Tousoulis D (2019) Cardiovascular effects of electronic cigarettes: a systematic review and meta-analysis. Eur J Prev Cardiol 26(11):1219–1228. https://doi.org/10.1177/2047487319832975
Ettehad D, Emdin CA, Kiran A, Anderson SG, Callender T, Emberson J, Chalmers J, Rodgers A, Rahimi K (2016) Blood pressure lowering for prevention of cardiovascular disease and death: a systematic review and meta-analysis. Lancet 387(10022):957–967. https://doi.org/10.1016/S0140-6736(15)01225-8
Benowitz NL, St Helen G, Liakoni E (2021) Clinical pharmacology of electronic nicotine delivery systems (ENDS): implications for benefits and risks in the promotion of the combusted tobacco endgame. J Clin Pharmacol 61(Suppl 2):S18–S36. https://doi.org/10.1002/jcph.1915
Shea BJ, Reeves BC, Wells G, Thuku M, Hamel C, Moran J, Moher D, Tugwell P, Welch V, Kristjansson E, Henry DA (2017) AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 358:j4008. https://doi.org/10.1136/bmj.j4008
Acknowledgements
We gratefully thank Prof. Gaetano Bertino for his support and project oversight as Scientific Director.
Funding
This investigator-initiated study was sponsored by ECLAT srl, a research based spin-off company of the University of Catania, with the help of a grant from the Foundation for a Smoke-Free World Inc., a US nonprofit 501(c)(3) private foundation. The contents, selection, and presentation of facts, as well as any opinions expressed herein are the sole responsibility of the authors and under no circumstances shall be regarded as reflecting the positions of the funders.
Author information
Authors and Affiliations
Contributions
GR—formal analysis, investigation, methodology, validation, writing—original draft, and writing—reviewing and editing. RV—formal analysis, investigation, methodology, validation, writing—original draft, and writing—reviewing and editing. MQ—investigation, and writing—reviewing and editing. RP—conceptualization, funding acquisition, resources, and writing—reviewing and editing. RO—conceptualization, data curation, formal analysis, investigation, methodology, project administration, resources, software, supervision, validation, visualization, writing—original draft, and writing—reviewing and editing.
Corresponding author
Ethics declarations
Conflict of interest
RP is full tenured professor of Internal Medicine at the University of Catania (Italy) and Medical Director of the Institute for Internal Medicine and Clinical Immunology at the same University. He has received grants from U-BIOPRED and AIR-PROM, Integral Rheumatology & Immunology Specialists Network (IRIS), Founda- tion for a Smoke Free World, Pfizer, GlaxoSmithKline, CV Therapeu- tics, NeuroSearch A/S, Sandoz, Merk Sharp & Dohme, Boehringer Ingelheim, Novartis, Arbi Group Srl., Duska Therapeutics, Forest Laboratories and Ministero dell Universita’ e della Ricerca (MUR) Bando PNRR 3277/2021, CUP E63C22000900006, funded by NextGenerationEU, the European Union (EU) economic recovery package. He is founder of the Center for Tobacco Prevention and Treatment (CPCT) at the University of Catania and of the Center of Excellence for the Acceleration of Harm Reduction at the same university. He receives consultancy fees from Pfizer, Boehringer Ingelheim, Duska Therapeutics, Forest Laboratories, CV Therapeutics, and Sermo Inc. He is being paid textbook royalties from Elsevier. He is also involved in a patent application for ECLAT Srl. He is a pro bono scientific advisor for Lega Italiana Anti Fumo (LIAF) and the International Network of Nicotine Consumers Organizations (INNCO); and he is Chair of the European Technical Committee for Standardization on “Requirements and test methods for emissions of electronic cigarettes” (CEN/TC 437; WG4).
Consent to participate and for publication
Not applicable.
Ethics approval
Consent for data collection was obtained during enrollment in the original studies.
Human and animal rights
This systematic review does not contain studies conducted by the authors.
Data repository
Data extraction forms, the JBI forms, Bias assessment forms, and Data discrepancy reports are available at Zenodo https://zenodo.org/record/4835883#.YPnMqECxUuU.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
La Rosa, G., Vernooij, R., Qureshi, M. et al. Clinical testing of the cardiovascular effects of e-cigarette substitution for smoking: a living systematic review. Intern Emerg Med 18, 917–928 (2023). https://doi.org/10.1007/s11739-022-03161-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-022-03161-z







