Abstract
Somatic embryogenesis (SE) in the tree fern Cyathea delgadii was first described in 2015 and since then has been used to exploration of this phenomenon in cryptogamic plants. To deepen the knowledge about the hormonal control of SE, stipe explants were cultured on media supplemented with hormone biosynthesis and transport inhibitors (HBTIs). In the presence of 30 µM 2,3,5-triiodobenzoic acid (TIBA), or 40 µM fluridone or 125 µM salicylic acid (SA), somatic embryo production was totally inhibited. The quantitative analysis of the changes in endogenous hormone and sugar contents was conducted every 2 days within 10-day-long initial culture. The results showed that the concentrations of endogenous indole-3-acetic acid (IAA), abscisic acid (ABA), cytokinins (CKs) and soluble sugars were strongly modified either by TIBA and fluridone. Under their influence, the contents of cytokinins such as c-Z, c-ZR, t-Z, t-ZR, KinR were reduced to barely detectable levels. Treatment with SA results in the changes in endogenous IAA and sugar contents. It also modifies the IAA/CKs ratio; however, excluding the first 2 days of culture, the concentrations of ABA and cytokinins were kept on the control level. All HBTIs significantly increased the kinetin (Kin) content. Our work sheds new light on the relationships between the biosynthetic inhibitors and phytohormones and sugars in the process of early SE. It can be helpful to study the role of hormones in acquisition of embryogenic competence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The most critical event in induction of somatic embryogenesis (SE) seems to be the establishment of a favourable hormonal balance within the initial explants cultured in vitro. Several external stimuli, such as plant growth regulators (Ayil-Gutiérrez et al. 2013), osmotic agents (Kikuchi et al. 2006), nutritional components (Pěnčík et al. 2015), amongst others, have been recognised as essential factors in determining both the hormone biosynthesis and the developmental fate of explant cells. Therefore, many previous studies focused on the role of phytohormones on different aspects of SE (Jiménez 2005; Vondráková et al. 2016), but the endogenous hormonal regulation of SE induction (i.e., the phase in which the somatic cells undergo dedifferentiation and acquire an embryogenic competence) is still not well understood.
Hormone biosynthesis and transport inhibitors (HBTIs) are useful compounds for elucidating the effects of different phytohormones on SE (Jiménez 2005). By applying antiauxin or auxin polar transport inhibitors, many investigators have shown the necessity of this group of hormones for embryo maturation and its normal development (Vondráková et al. 2016). It has been also proposed that auxin may act as a trigger in the embryogenic program of plant somatic cells (LoSchiavo et al. 1989; Tokuji and Kuriyama 2003; Nic-Can and Loyla-Vargas 2016). Establishment of an auxin gradient was essential for initiating somatic embryos in Eleutherococcus senticosus Maxim. (Choi et al. 2001) and for inducing stem cell formation within the embryonic callus of Arabidopsis (Su and Zhang 2009). Conversely, it does not appear to be required for the very early stages of direct SE in Daucus carota L. (Tokuji and Kuriyama 2003). Like auxin, cytokinins are key regulators for various aspects of plant growth and development, including somatic embryo production (Jiménez 2005). Together with auxin, they regulate meristem formation in early embryogenesis. However, their importance during the acquisition of embryogenic potential is poorly understood. Tokuji and Kuriyama (2003) showed that cytokinins regulate the early stage of SE in carrot. Somleva et al. (1995) and Sáenz et al. (2010) have found an inverse relationship between cytokinins and the embryogenic response. Recent studies revealed a close link between auxin and ethylene in the mechanism that controls an acquisition of embryogenic competence on the level of specific genes (Nowak et al. 2015). Using salicylic acid (SA), that is considered as an inhibitor of ethylene biosynthesis, both the positive (Luo et al. 2001; Quiroz-Figueroa et al. 2001; Sakhanokho et al. 2009; Mulgund et al. 2012) and negative (Nissen 1994; Hosseini et al. 2011; Kępczyńska and Zielińska 2013) influence of the ethylene on somatic embryo production has been shown. In the case of abscisic acid (ABA), its role in SE initiation is less well known. So far, it has been shown that an accumulation of high levels of ABA inhibits the ability of the explant to undergo SE (Ivanova et al. 1994; Jiménez and Bangerth 2001; Grzyb et al. 2017). In conifers, the decline of ABA level in the presence of the ABA synthesis inhibitor fluridone enhances the transition of proembryogenic masses to early somatic embryos (Farias-Soares et al. 2014). However, some ABA seems to be necessary for the initiation of both direct and indirect SE (Rajasekaran et al. 1987; Kikuchi et al. 2006; Su et al. 2013). Although the biosynthetic inhibitors are often used in study of SE, there are only a few reports describing how the contents of different endogenous hormones are altered by treatment with those substances (Rajasekaran et al. 1987; Nissen 1994; Kikuchi et al. 2006; Ruduś et al. 2009; Farias-Soares et al. 2014).
It is worth noting that the regulatory role of ABA is achieved, in part, by crosstalk with other hormones (Belin et al. 2009), and with sucrose signalling pathways involved in starch biosynthesis. On the other hand, sugars modulate many vital processes, including embryogenesis, via crosstalk with phytohormones (Gibson 2004). For example, the availability of sucrose and glucose helps regulate the transition from growth by cell division to growth by cell expansion and reserve accumulation in developing embryos (Gibson 2004). Moreover, the role of LEAFY COTYLEDON1 (LEC1) gene, that controls embryogenic competence, requires both the auxin and sucrose to promote cell division and embryonic differentiation (Casson and Lindsey 2006). Various exogenous sugars have been used to enhance SE efficiency, indicating that carbon sources play an important role in this process (Yaseen et al. 2013). Despite the importance of sucrose metabolism in maintaining the balance between hexose signals and metabolic paths (Koch 2004), there is only one example of the relationship between the content of phytohormones and endogenous sugars during the SE induction currently displayed (Grzyb et al. 2017).
The tree fern Cyathea delgadii Sternb. is the first cryptogamic plant to be used as a model system for studying SE (Mikuła et al. 2015b; Domżalska et al. 2017; Grzyb et al. 2017). It is also one of the few plant species in which direct SE can be induced on plant growth regulator-free medium (Mikuła et al. 2015a). The hormonal balance that allows induction of embryogenic potential in C. delgadii is achieved by long-term etiolation of donor plantlets (Mikuła et al. 2015a). It reduces a high concentration of phytohormone ABA—the main factor inhibiting the explant ability to SE (Grzyb et al. 2017). In response to an explant excision, a dramatic reduction in the content of several cytokinins, indole-3-acetic acid (IAA) and ABA happen. It initiates the whole cascade of events leading to the acquisition of embryogenic competence. Among them the sudden increase in the concentration of soluble sucrose is considered to be the switch to phase in which certain epidermal cells regain their potential to division and to develop into somatic embryos. In this way, the explants of juvenile etiolated stipes of C. delgadii undergo restructuring to generate embryogenic cells during 10 days of initial culture (Grzyb et al. 2017). These studies have shown that the balance between the endogenous IAA and cytokinin content is particularly related to the induction phase of SE. In the present work, the hormone biosynthesis and transport inhibitors, such as TIBA, fluridone and SA, were used to examine their impact on the somatic embryo production in C. delgadii. The effect of these treatments on endogenous contents of IAA, cytokinins, ABA and sugars in stipe explants is presented.
Materials and methods
Plant material and tissue culture
The somatic embryo-derived sporophytes of C. delgadii were cultured on agar medium containing half-strength Murashige and Skoog’s (1962) macro- and micro-nutrients and a full complement of vitamins (1/2MS), supplemented with 2% (w/v) sucrose; 0.7% plant agar (Duchefa Biochemie); pH 5.8. The cultures were maintained in constant darkness, in a climatic chamber at +24 ± 1 °C. Five-month-old etiolated plantlets that had developed 4–5 leaves, were used as a source of explants (Mikuła et al. 2015a, b). Stipe explants measuring 2.5 mm in length were excised from the first frond of etiolated sporophytes and cultured on 1/2MS medium with 1% (w/v) sucrose (control), and supplemented with TIBA (4, 8, 10, 12, 16, 20 and 30 µM) or fluridone (1, 10, 20, 30 and 40 µM) or SA (1, 5, 10, 25, 50, 75, 100 and 125 µM), in darkness. TIBA was dissolved in 0.1% (v/v) dimethyl sulfoxide (DMSO). Medium supplemented with DMSO, that was used as additional control, did not inhibit the SE.
The efficiency of SE was expressed as the percentage response of explants, calculated as follows: (number of explants that formed somatic embryos/total number of explants) × 100; and as the number of somatic embryos per responding explant. Somatic embryos were counted under a stereo microscope (Olympus SZH, Japan) following 2 months of culture.
Sample preparation for HPLC analysis
The samples were collected every 2 days within 10-day-long culture. Immediately after collection, the stipe explants (about 80 explants per sample; 35 mg of FW) were frozen in liquid nitrogen, lyophilised and homogenised in an ice-cold mixture of methanol, water, and formic acid (15/4/1) according to Dobrev and Kamínek (2002). Internal isotopic standard mixture consisting of deuterated IAA and Kin labelled with nitrogen 15N was added to each sample. The prepared extract was fractionated using solid phase extraction (SPE) columns Oasis MCX (Waters). According to HPLC method of Štefančič et al. (2007), Żur et al. (2015), and Hura et al. (2016), three fractions were collected: “acidic” for IAA and ABA analyses, “basic” for cytokinins analyses, and “flow through” for carbohydrates analyses, as provided by Grzyb et al. (2017). All chemicals for sample preparation and HPLC were bought in the Sigma-Aldrich Sp. z.o. (Poznan, Poland), HPLC standards (normal and stable isotope labelled) were bought in the Olchemim (Olomouc, Czech Republic).
Quantification of IAA and ABA
The fraction containing IAA and ABA was eluted from SPE column according to Żur et al. (2015). The samples were analysed using a Supelco Ascentis RP-Amide column (7.5 cm × 2.1 mm, 2.7 μm) as has been previously described (Grzyb et al. 2017). The HPLC apparatus used was Agilent Technologies 1290 Infinity equipped with Agilent Technologies 6460 Triple Quad LC/MS with Jet Stream. Two most abundant ions were monitored (MRM—multiple reaction monitoring mode) and they were: IAA—176.1 primary, 130.3, 77.2 secondary; D-IAA (deuterated IAA used as internal standard)—181.1 primary, 134.7, 81.4 secondary; ABA—265.2 primary, 229.1, 247.1 secondary. Agilent Technologies Mass Hunter 5 software was used for apparatus control, data collection, and processing.
Quantification of cytokinins
Cytokinins such as c-Z, c-ZR, iPA,t-Z, t-ZR, Kin and KinR were separated from the samples. Their sum is defined as the total concentration of cytokinins (CKs). In order to assess hormonal changes introduced by HBTIs, the phytohormone balance was expressed by the ratios of IAA to total amount of CKs, and ABA to CKs or IAA.
Fractions of cytokinins were flushed out from the SPE column after collecting IAA and ABA, collected using procedure described earlier (Grzyb et al. 2017) and analysed using the same chromatographic system and HPLC column as described above. The monitored ions were as follows: c/tZ—220.2 primary, 136.3, 202.3, secondary; c/t ZR—352.4 primary, 220.3, 136.3, secondary; Kin—216.2 primary, 188.3, 148.3 secondary; KinR—348.3 primary, 148.3, 216.3 secondary; iPA—204.2 primary, 136.3, 148.3 secondary; Kin-15N (heavy nitrogen labelled Kin used as internal standard)—220.1 primary, 192.3, 152.3 secondary.
Quantification of soluble sugars
The “flow through” fraction collected from the SPE column was lyophilised and reconstituted according to the procedure described by Janeczko et al. (2010). Samples were analysed using Agilent Technologies 1200 HPLC equipped with an amperometric detector ESA Coulochem II Analytical Cell 5040 with gold electrode. A Hamilton RCX-10 250 × 4.1 mm (Hamilton, Reno, NV, USA) HPLC column was used. The mobile phase, flow rate of HPLC column and the detector settings have been applied according to Grzyb et al. (2017). Agilent Technologies ChemStation B04 software was used for apparatus control, data collection and processing.
Statistical analysis
Data for the effect of HBTIs on somatic embryo production are presented as mean ± SD for three biological replications each consisting of 48 explants, and these were tested by means of one-way ANOVA analysis of variance and Fisher’s least significant difference (LSD) procedure using Statgraphics Plus software. Significance was set at a P value of less than 0.05. Data for endogenous hormone and sugar contents are presented as mean ± SD for three replicates (35 mg FW, i.e., about 80 explants, was considered as one replicate). Differences in the concentration of hormones and sugars between the control and HBTIs treatments were analysed using the Student’s t test for each day separately. Only a return of P < 0.05 was designated as being statistically significant. Statistical analysis was performed for each type of HBTIs separately.
Results
Effect of TIBA, fluridone and SA on the somatic embryo production in C. delgadii
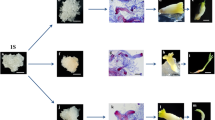
After 2 months of culture on control medium, 78.5% of explants were able to produce somatic embryo-derived sporophytes showing an efficiency of almost 25 embryos per responding explant (Figs. 1a, 2a). Those sporophytes developed the first leaf, shoot apex and primordium of the second leaf, as well as root primordium (Fig. 1b). Although the culture was kept in the dark, the first leaf of the juvenile sporophyte was clearly greenish, but the blades of the fronds were characteristically coiled in the crosier at all times (Fig. 1a, b).
Somatic embryogenesis (SE) of Cyathea delgadii and its disturbances. a–e Cultures kept in the dark for 2 months. a Numerous somatic embryo-derived sporophytes obtained on ½ MS medium. b Details of juvenile sporophyte development; greenish crosier of the first frond and root on the base of sporophyte are visible. The inset shows red autofluorescence of chlorophyll in crosier cells induced by blue-violet light (blue-violet light; BV filter 400–440 nm). c Stipe explant after 2 months of culture in the presence of 30 µM TIBA without any cell divisions and d with gametophyte-like structures. e Whitish somatic embryos formed in the presence of 10 µM fluridone (details on inset). f Whitish frond of sporophyte (1 and 2) developed in darkness on the medium supplemented with fluridone. Green primordium (5), frond (4) and leaf blade (3) formed after transferred of sporophyte to fluridone-free medium and kept under photoperiodic conditions (5-month-old plantlet). R root, 1st first leaf, 2nd second leaf
Efficiency of somatic embryogenesis in C. delgadii after 2 months of culture on control medium (1/2MS medium supplemented with 1% sucrose) (a) or in the presence of TIBA (b), fluridone (c) or SA (d); in darkness. HBTIs hormone biosynthesis and transport inhibitors, TIBA 2,3,5-triiodobenzoic acid. Values marked with the same letter do not differ significantly at the 0.05 level according to Fisher’s least significant difference (LSD’s) test. Data represent mean ± standard deviation of three independent experiments, each consisting of 48 explants
The capacity to produce somatic embryos gradually declined with increasing concentrations of TIBA and fluridone (Fig. 2b, c). The percentage of responding explants and the number of somatic embryos were about 2-fold less in explants treated with 10 µM TIBA or fluridone than in controls. The embryogenic capacity was totally lost in the presence of 30 µM TIBA or 40 µM fluridone. There were no specific cell divisions for somatic embryo formation observed (Fig. 1c, d). The somatic embryos that were formed on the explants treated with a smaller dose of fluridone were unable to synthesise functional chlorophyll as indicated by their pallid appearance (Fig. 1e). It led to the formation of sporophytes with completely white fronds. The sporophytes, after being transferred to fluridone-free medium and exposed to light, produced normally developed green leaves with expanded leaf blades (Fig. 1f).
When SA was used at concentrations of 1–5 µM, there were no significant differences in the percentage of responding explants observed, and only a slight decrease in the number of somatic embryos was achieved (Fig. 2d). Concentrations of SA between 50 and 100 µM resulted in a dramatic reduction in the embryogenic capacity of C. delgadii stipe explants. The medium supplemented with 125 µM SA completely inhibited SE. The gametophyte-like structures were formed sporadically at the end of first month of SA-treated culture (Fig. 1d).
Effect of TIBA, fluridone and SA on the content of endogenous IAA, ABA, cytokinins and sugars
In order to study the involvement of endogenous IAA, ABA, cytokinins and sugars in SE induction of C. delgadii, TIBA, fluridone and SA were used at concentrations that totally inhibited somatic embryo production, i.e., 30, 40 and 125 µM, respectively. When these HBTIs were added to the induction medium, the content of all phytohormones studied diminished rapidly in excised explants during 2 days of culture (Fig. 3a–c; Supplementary Fig. 1). In the case of explants cultured on control medium, minimal levels of these hormones were achieved 2 days later.
The effect of SA, fluridone and TIBA on endogenous a IAA, b ABA, c CKs and d Kin contents in the stipe explants of C. delgadii during 10 days of culture. ABA abscisic acid, CKs the sum of t-Z, c-Z, t-ZR, c-ZR, Kin, KinR and iPA contents, IAA indole-3-acetic acid, Kin kinetin, TIBA 2,3,5-triiodobenzoic acid. Values marked with asterisk differ significantly from the control at the 0.05 level according to Student’s t test. Data represent mean ± standard deviation of three independent experiments, each consisting of 48 explants
The use of TIBA contributed to about a 2-fold reduction in the concentration of IAA and CKs (Fig. 3a, c). A total reduction in the content of c-Z, c-ZR, t-Z, t-ZR, KinR was also observed (Supplementary Fig. 1). Moreover, almost a 4-fold increase in ABA content between days 6 and 8 of culture was found (Fig. 3b). TIBA also reduced the IAA/CKs ratio and showed an increasing trend in the ABA/CKs and ABA/IAA ratios peaking at day 8 of culture (Fig. 4a–c). Another characteristic feature of the TIBA treatment was complete consumption of sugars on day 2 (Fig. 4d–f). The sugar concentrations increased gradually over the next 6 days of culture.
Ratios between various phytohormones (a–c) and concentrations of soluble sugars (d–f) during 10 days of initial culture on control medium (1/2MS medium supplemented with 1% sucrose) or in the presence of SA, fluridone or TIBA. ABA abscisic acid, CKs the sum of t-Z, c-Z, t-ZR, c-ZR, Kin, KinR and iPA contents, IAA indole-3-acetic acid, SA salicylic acid, TIBA 2,3,5-triiodobenzoic acid. Values marked with asterisk differ significantly from the control at the 0.05 level according to Student’s t test. Data represent mean ± standard deviation of three independent experiments, each consisting of 48 explants
When fluridone was added to the induction medium, the ABA and CKs contents showed a 2-fold increase and a 2-fold reduction, respectively, compared to control explants (Fig. 3b, c). These changes resulted in a 1- to 3-fold increase in the value of all hormone ratios (Fig. 4a–c). The sucrose concentration showed an increasing trend in parallel with a reduction in glucose and fructose content (Fig. 4d–f). At 4 days after commencing culture, the content of all sugars gradually diminished.
Application of SA practically did not affect the concentrations of ABA and CKs after day 4 of culture (Fig. 3b, c). Thus, the ABA/CKs and ABA/IAA ratios were similar to those observed in control explants throughout the duration of culture (Fig. 4b, c). The most important change noticed was 2.5-fold decrease in the value of IAA/CKs ratio starting on day 4 of culture (Fig. 4a). Significant changes in sugar content were also observed (Fig. 4d–f).
Regardless of the type of HBTIs added to the medium (excluding an effect of fluridone at day 2), the concentration of Kin was much higher than in control plant material throughout the entire length of the culture period (Fig. 3d).
Discussion
Modifications of the culture conditions through the inclusion of biosynthetic inhibitors affecting the polar transport of auxin and different hormone synthesis have been extensively used to investigate their role during somatic embryo production and development in spermatophytes. Here, HBTIs were used for the first time as a tool to investigate hormonal control of SE in ferns.
Effect of HBTIs on somatic embryo production
The auxin polar transport inhibitor TIBA competes with the auxin binding site on the efflux carriers. Its effect on SE depends on the genotype (Laublin et al. 1991) and explant type (Choi et al. 1997), as well as the dosage. TIBA at a concentration of 0.5 µM greatly increased the number of somatic embryos during direct SE of Oncidium (Chen and Chang 2004). At a concentration of 4–40 µM, TIBA was found to be inhibitory for the SE of Eleutherococcus senticosus, Panax ginseng C.A.Meyer, Picea abies (L.) H. Karst, and Arachis hypogaea L. (Laublin et al. 1991; Choi et al. 1997, 2001). In C. delgadii, a dramatic reduction in embryo-forming capacity was correlated with an increase in TIBA concentration. In the presence of 30 µM TIBA, somatic embryo production was completely inhibited.
Application of fluridone revealed that ABA is needed not only to promote maturation and normal development of somatic embryos (Rajasekaran et al. 1982; Nickle and Yeung 1994; Senger et al. 2001; Su et al. 2013), but also to the acquisition of embryogenic competence. Rajasekaran et al. (1987) and Su et al. (2013) demonstrated the inhibitory effect of fluridone at concentrations ranging from 0.1 to 3 µM on SE of Pennisetum purpureum Schum and Arabidopsis. In carrot stress-inducible SE, embryo formation was inhibited by 100 µM fluridone (Kikuchi et al. 2006). Our study showed that 40 µM fluridone is sufficient for total inhibition of SE in C. delgadii. It is worth emphasising that the inhibitory effect of fluridone can be nullified by the simultaneous application of fluridone and ABA, confirming the important role of this hormone in SE induction (Kikuchi et al. 2006). A significant problem with fluridone application is its inhibitory effect on carotenoid synthesis (Popova 1995). Lack of carotenoids results in a block in membrane formation and the concurrent blanching of plantlets. Here, our results indicate that this negative effect is irreversible. The newly formed leaves of blanched plantlets of C. delgadii transferred to fluridone-free medium and subjected to light became green and fully functional.
An effect of SA on SE induction appears to be strongly species- or genotype-dependent. In Coffea arabica suspension culture, picomolar concentrations of SA resulted in a 2-fold increase in cellular growth and SE efficiency, but 1 µM inhibited these processes (Quiroz-Figueroa et al. 2001). Incorporation of 7.2 µM SA in the induction medium increased the percentage of SE in ten genotypes of Pinus roxburghii (Chir pine), but concentrations between 14.5 and 36.3 µM had a toxic effect and resulted in the browning of explants (Malabadi et al. 2008). It was also shown that the inclusion of SA in the induction medium at concentrations of 40, 75–100, 150 and 500 µM enhanced SE in callus cultures of Coffea canephora P ex Fr., Hedychium bousigonianum Pierre ex Gagnep., Astragalus adsurgens Pall., and the formation of embryogenic callus in Avena nuda L., respectively (Luo et al. 2001; Hao et al. 2006; Kumar et al. 2007; Sakhanokho et al. 2009). In contrast to these findings, our results showed that the number of somatic embryos of C. delgadii diminished with increasing concentrations of SA. When SA was used at a concentration of 125 µM, SE was completely inhibited. Our findings are consistent with those obtained for Daucus carota, where 100 µM SA almost totally suppressed somatic embryo production (Hosseini et al. 2009).
The impact of biosynthetic inhibitors on the endogenous hormone and sugar contents
How the contents of different phytohormones are disturbed under the influence of inhibitor treatment is still an open problem. Following application of biosynthetic inhibitors in concentrations that stopped an expression of embryogenic totipotency in stipe explants of C. delgadii, it was possible to demonstrate the changes in content of endogenous IAA, ABA and cytokinins during first 10 days of initial culture. Selected phytohormones are well known as the key regulators of plant cell division and differentiation (Nic-Can and Loyola-Vargas 2016). Their signals may also affect sucrose metabolism (Lee and Huang 2013).
The earlier works showed the inhibitory effect of TIBA on the somatic embryo development by blocking the polar auxin transport (Schiavone and Cooke 1987; Liu et al. 1993). Some investigators have also suggested the necessity of polar auxin transport for initiation of embryo formation (Choi et al. 1997, 2001; Su and Zhang 2009). On the contrary, studies by Tokuji and Kuriyama (2003) showed that it is not needed in the induction of direct SE in carrot. Ramarosandratana and Van Staden (2004) hypothesised that the reduction of embryonal-suspensor mass initiation in Picea abies L. Karst. following application of TIBA resulted from an increase of internal auxin concentration rather than disruption of auxin gradient. However, our study clearly showed that exogenously applied TIBA at a concentration of 30 µM contributes to about a 2-fold reduction in the IAA content compared to a non-treated control. Furthermore, there was even a 4-fold increase in the level of ABA observed between days 6 and 8 of culture. TIBA also strongly modified the content of cytokinins and the ratios between all phytohormones studied here. To the best of our knowledge, this is the first report describing the relationship between TIBA treatment and endogenous hormone contents. We conclude that loss of ability to SE by explant treated with TIBA may be a direct result of the ABA accumulation in response to a stress.
When an early SE of C. delgadii was examined by the use of fluridone, we can clearly see that ABA interferes with auxin metabolism. In stipe explants treated with fluridone, the concentrations of endogenous IAA and ABA are maintained at slightly lower, and at an almost 2-fold greater concentration, respectively, when compared with control explants. In young leaf explants of Pennisetum purpureum Schum. that were cultured in the presence of fluridone at concentration higher than 5 mg/l for 5 days, both the IAA and ABA levels decreased (Rajasekaran et al. 1987). These fluctuations are consistent with the trend presented for proembryogenic tissue of Araucaria angustifolia (Bertol.) (Farias-Soares et al. 2014) and for 14-day-old explants of cotyledonary somatic embryo of Medicago sativa L. (Ruduś et al. 2009). The negative effect of fluridone on the content of endogenous ABA was also shown in carrot explants (Kikuchi et al. 2006). It is difficult to explain why the C. delgadii explants treated with fluridone contain 2-fold more endogenous ABA than control explants. Our analysis showed that fluridone, like TIBA, not only disrupts the contents of IAA and ABA, but also reduces the contents of some endogenous cytokinins (such as c-Z, c-ZR, t-Z, t-ZR, KinR) to barely detectable levels.
SA is a stress-related phytohormone that functions as an important signalling molecule involved in the embryogenic response. Its activity induces an increase in the endogenous concentration of hydrogen peroxide (H2O2), and it has been proposed that it signals the initiation of SE (Luo et al. 2001). However, prolonging SA treatment can generate overproduction of H2O2 (Rao et al. 1997), and consequently inhibit SE. The inhibitory effect of SA on somatic embryo production in C. delgadii may also be related to the inhibition of the ethylene synthesis, as it was suggested by other authors (Romani et al. 1989; Quiroz-Figueroa et al. 2001; Hosseini et al. 2011; Mulgund et al. 2012). By using SA we revealed significant decrease in the content of endogenous IAA that was observed almost throughout the culture period. It is worth to note that the concentrations of ABA and cytokinins were lower than in control explants only during first 2 days of culture; however, the values of the hormonal ratios remained unchanged.
Our results provide evidence that by HBTIs treatment a significant increase in endogenous Kin content happens. Surprisingly, the high levels of Kin were reached under the influence of SA during first 6 days of culture. It may be the result of cellular changes involving DNA degradation or intensive oxidative metabolism (Barciszewski et al. 2007). It was shown that Kin is synthesised via the production of furfural, an oxidative damage product of DNA deoxyribose, and it is quenched by the adenine base (Barciszewski et al. 2000). Some data indicate its strong antioxidant properties (Olsen et al. 1999; Barciszewski et al. 2000; Żur et al. 2015).
In view of the interactions between hormone and sugar response pathways (Gibson 2004), we showed that treatment with HBTIs affects glucose, fructose and sucrose contents in stipe explants of C. delgadii. All HBTIs investigated caused significant changes to endogenous sugar profiles, the most important being the absence of a short-term increase in sucrose concentration at day 6 of culture. This rapid increase in sucrose content is considered as a switch to the SE expression phase in C. delgadii (Grzyb et al. 2017).
Conclusions
In the present investigation we showed that TIBA, fluridone and SA at concentrations 30 µM, 40 µM and 125 µM, respectively, totally suppress somatic embryo production in C. delgadii. Both TIBA and fluridone strongly influence the levels of endogenous IAA, ABA and cytokinins as well as the hormone ratios. Under their influence, the contents of c-Z, c-ZR, t-Z, t-ZR, KinR were reduced to barely detectable levels. Treatment with SA results in the changes in IAA content and the IAA/CKs ratio that are particularly related to the SE induction. The imbalances in phytohormone level are responsible for the modification of sugar contents, including sucrose—the main factor triggering embryogenesis in C. delgadii.
Our work sheds new light on the relationship between the treatment with biosynthetic inhibitors and the changes in phytohormone and sugar levels. It is worth to emphasise that this is the first study on influence of HBTIs on SE in cryptogams. It can be helpful to study the role of hormones in acquisition of totipotency.
Author contribution statement
MG designed and performed the experiments. AK carried out HPLC separation. MG and AM wrote the manuscript. All authors read and approved the final manuscript.
Abbreviations
- ABA:
-
Abscisic acid
- c/t-Z:
-
Cis/trans zeatin
- c/t-ZR:
-
Cis/trans-zeatin riboside
- CKs:
-
Total concentration of cytokinins
- Fluridone:
-
1-Methyl-3-phenyl-5-[3-trifluoromethyl (phenyl)]-4-(1H)-pyridinone
- FW:
-
Fresh weight
- HBTIs:
-
Hormone biosynthesis and transport inhibitors
- HPLC:
-
High-performance liquid chromatography
- IAA:
-
Indole-3-acetic acid
- iPA:
-
Isopentenyladenosine
- Kin:
-
Kinetin
- KinR:
-
Kinetin riboside
- SA:
-
Salicylic acid
- SE:
-
Somatic embryogenesis
- TIBA:
-
2,3,5-Triiodobenzoic acid
References
Ayil-Gutiérrez B, Galaz-Avalos RM, Peña-Cabrera E, Loyola-Vargas VM (2013) Dynamics of the concentration of IAA and some of its conjugates during the induction of somatic embryogenesis in Coffea canephora. Plant Signal Behav 8:1–10
Barciszewski J, Siboska G, Rattan SIS, Clark BFC (2000) Occurrence, biosynthesis and properties of kinetin (N 6 -furfuryladenine). Plant Growth Regul 32:257–265
Barciszewski J, Massino F, Clark BFC (2007) Kinetin—a multiactive molecule. Int J Biol Macromol 40:182–192. https://doi.org/10.1016/j.ijbiomac.2006.06.024
Belin C, Megies C, Hauserová E, Lopez-Molina L (2009) Abscisic acid represses growth of the Arabidopsis embryonic axis after germination by enhancing auxin signaling. Plant Cell 21:2253–2268. https://doi.org/10.1105/tpc.109.067702
Casson SA, Lindsey K (2006) The turnip mutant of Arabidopsis reveals that LEAFY COTYLEDON1 expression mediates the effects of auxin and sugars to promote embryonic cell identity. Plant Physiol 142:526–541. https://doi.org/10.1104/pp.106.080895
Chen JT, Chang WC (2004) TIBA affects the induction of direct somatic embryogenesis from leaf explants of Oncidium. Plant Cell Tiss Org Cult 79:315–320. https://doi.org/10.1007/s11240-004-4613-5
Choi YE, Kim HS, Soh WY, Yang DC (1997) Developmental and structural aspects of somatic embryos formed on medium containing 2,3,5,-triiodobenzoic acid. Plant Cell Rep 16:738–744. https://doi.org/10.1007/s002990050312
Choi YE, Katsumi M, Sano H (2001) Triiodobenzoic acid, an auxin polar transport inhibitor, suppresses somatic embryo formation and postembryonic shoot/root development in Eleutherococcus senticosus. Plant Sci 160:1183–1190. https://doi.org/10.1016/S0168-9452(01)00357-0
Dobrev PI, Kamínek M (2002) Fast and efficient separation of cytokinins from auxin and abscisic acid and their purification using mixed-mode solid-phase extraction. J Chromatogr A 950:21–29. https://doi.org/10.1016/S0021-9673(02)00024-9
Domżalska L, Kędracka-Krok S, Jankowska U, Grzyb M, Sobczak M, Rybczyński JJ, Mikuła A (2017) Proteomic analysis of stipe explants reveals differentially expressed proteins involved in early direct somatic embryogenesis of the tree fern Cyathea delgadii Sternb. Plant Sci 258:61–76. https://doi.org/10.1016/j.plantsci.2017.01.017
Farias-Soares FL, Steiner N, Schmidt ÉC, Pereira MLT, Rogge-Renner GD, Bouzon ZL, Floh ESI, Guerra MP (2014) The transition of proembryogenic masses to somatic embryos in Araucaria angustifolia (Bertol.) Kuntze is related to the endogenous contents of IAA, ABA and polyamines. Acta Physiol Plant 36:1853–1865. https://doi.org/10.1007/s11738-014-1560-6
Gibson SI (2004) Sugar and phytohormone response pathways: navigating a signalling network. J Exp Bot 55:253–264. https://doi.org/10.1093/jxb/erh048
Grzyb M, Kalandyk A, Waligórski P, Mikuła A (2017) The content of endogenous hormones an sugars in the process of early somatic embryogenesis in the tree fern Cyathea delgadii Sternb. Plant Cell Tiss Org Cult. https://doi.org/10.1007/s11240-017-1185-8
Hao L, Zhou L, Xu X, Cao J, Xi T (2006) The role of salicylic acid and carrot embryogenic callus extracts in somatic embryogenesis of naked oat (Avena nuda). Plant Cell Tiss Organ Cult 85:109–113. https://doi.org/10.1007/s11240-005-9052-4
Hosseini SS, Mashayekhi K, Alizadek M (2009) Ehtylene production and somatic embryogenesis of carrot explants as affected by salicylic acid treatments. Am J Agric Environ Sci 6:539–545
Hosseini SS, Mashayekhi K, Alizadeh M, Ebrahimi P (2011) Effect of salicylic acid on somatic embryogenesis and chlorogenic acid levels of carrot (Daucus carota cv. Nantes) explants. J Ornam Hortic Plants 1:105–113
Hura T, Dziurka M, Hura K, Ostrowska A, Dziurka K (2016) Different allocation of carbohydrates and phenolics in dehydrated leaves of triticale. J Plant Physiol 202:1–9. https://doi.org/10.1016/j.jplph.2016.06.018
Ivanova A, Velcheva M, Denchev P, Atanassov A, Van Onckelen HA (1994) Endogenous hormone levels during direct somatic embryogenesis in Medicago falcata. Physiol Plant 92:85–89. https://doi.org/10.1111/j.1399-3054.1994.tb06658.x
Janeczko A, Biesaga-Kościelniak J, Oklest’kova J, Filek M, Dziurka M, Szarek-Łukaszewska G, Kościelniak J (2010) Role of 24-epibrassinolide in wheat production: physiological effects and uptake. J Agron Crop Sci 196:311–321. https://doi.org/10.1111/j.1439-037X.2009.00413.x
Jiménez VM (2005) Involvement of plant hormones and plant growth regulators on in vitro somatic embryogenesis. Plant Growth Regul 47:91–110. https://doi.org/10.1007/s10725-005-3478-x
Jiménez VM, Bangerth F (2001) Hormonal status of maize initial explants and of the embryogenic and non-embryogenic callus cultures derived from them as related to morphogenesis in vitro. Plant Sci 160:247–257. https://doi.org/10.1016/S0168-9452(00)00382-4
Kępczyńska E, Zielińska S (2013) The role of endogenous ethylene in carbohydrate metabolism of Medicago sativa L. somatic embryos in relation to their regenerative ability. J Plant Growth Regul 32:191–199. https://doi.org/10.1007/s00344-012-9288-2
Kikuchi A, Sanuki N, Higashi K, Koshiba T, Kamada H (2006) Abscisic acid and stress treatment are essential for the acquisition of embryogenic competence by carrot somatic cells. Planta 223:637–645. https://doi.org/10.1007/s00425-005-0114-y
Koch K (2004) Sucrose metabolism: regulatory mechanisms and pivotal roles in sugar sensing and plant development. Curr Opin Plant Biol 7:235–246. https://doi.org/10.1016/j.pbi.2004.03.014
Kumar V, Ramakrishna A, Ravishankar GA (2007) Influence of different ethylene inhibitors on somatic embryogenesis and secondary embryogenesis from Coffea canephora P ex Fr. Vitr Cell Dev Biol - Plant 43:602–607. https://doi.org/10.1007/s11627-007-9067-0
Laublin G, Saini HS, Cappadocia M (1991) In vitro plant regeneration via somatic embryogenesis from root culture of some rhizomatous irises. Plant Cell Tiss Organ Cult 27:15–21. https://doi.org/10.1007/BF00048200
Lee S-T, Huang W-L (2013) Cytokinin, auxin, and abscisic acid affects sucrose metabolism conduce to de novo shoot organogenesis in rice (Oryza sativa L.) callus. Bot Stud 54:5 http://www.as-botanicalstudies.com/content/54/1/5
Liu CM, Xu ZH, Chua NH (1993) Auxin polar transport is essential for the establishment of bilateral symmetry during early plant embryogenesis. Plant Cell 5:621–630
LoSchiavo F, Pitto L, Giuliano G, Torti G, Nuti-Ronchi V, Marazziti D, Vergara R, Orselli S, Terzi M (1989) DNA methylation of embryogenic carrot cell cultures and its variations as caused by mutation, differentiation, hormones and hypomethylating drugs. Theor Appl Genet 77:325–331. https://doi.org/10.1007/BF00305823
Luo J, Jiang S, Pan L (2001) Enhanced somatic embryogenesis by salicylic acid of Astragalus adsurgens Pall.: relationship with H2O2 production and H2O2-metabolizing enzyme activities. Plant Sci 161:125–132. https://doi.org/10.1016/S0168-9452(01)00401-0
Malabadi RB, da Silva TJA, Nataraja K (2008) Salicylic acid induces somatic embryogenesis from mature trees of Pinus roxburghii (Chir pine) using TCL technology. Tree For Sci Biotechnol 2:34–39
Mikuła A, Pożoga M, Grzyb M, Rybczyński JJ (2015a) An unique system of somatic embryogenesis in the tree fern Cyathea delgadii Sternb.: the importance of explant type, and physical and chemical factors. Plant Cell Tiss Organ Cult 123:467–478. https://doi.org/10.1007/s11240-015-0850-z
Mikuła A, Pożoga M, Tomiczak K, Rybczyński JJ (2015b) Somatic embryogenesis in ferns: a new experimental system. Plant Cell Rep 34:783–794. https://doi.org/10.1007/s00299-015-1741-9
Mulgund GS, Meti NT, Malabadi RB, Nataraja K, Kumar SV (2012) Role of salicylic acid on conifer somatic embryogenesis. Res Biotechnol 3:57–61
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Nic-Can GI, Loyola-Vargas VM (2016) The role of the auxins during somatic embryogenesis. In: Loyola-Vargas VM, Ochoa-Alejo N (eds) Somatic embryogenesis: fundamental aspects and applications. Springer, Cham, pp 171–182. https://doi.org/10.1007/978-3-319-33705-0_10
Nickle TC, Yeung EC (1994) Further evidence of a role for abscisic acid in conversion of somatic embryos of Daucus carota. In Vitro Cell Dev Biol Plant 30:96–103. https://doi.org/10.1007/BF02632136
Nissen P (1994) Stimulation of somatic embryogenesis in carrot by ethylene: effects of modulators of ethylene biosynthesis and action. Physiol Plant 92:397–403
Nowak K, Wójcikowska B, Gaj MD (2015) ERF022 impacts the induction of somatic embryogenesis in Arabidopsis through the ethylene-related pathway. Planta 241:967–985. https://doi.org/10.1007/s00425-014-2225-9
Olsen A, Siboska GE, Clark BF, Rattan SIS (1999) N(6)-furfuryladenine, kinetin, protects against fenton reaction-mediated oxidative damage to DNA. Biochem Biophys Res Commun 265:499–502. https://doi.org/10.1006/bbrc.1999.1669
Pěnčík A, Turečková V, Paulišić S, Rolčík J, Strnad M, Mihaljević S (2015) Ammonium regulates embryogenic potential in Cucurbita pepo through pH-mediated changes in endogenous auxin and abscisic acid. Plant Cell Tiss Organ Cult 122:89–100. https://doi.org/10.1007/s11240-015-0752-0
Popova L (1995) Effect of fluridone on plant development and stress-induced accumulation in Vicia faba L. plants. Bulg J Plant Physiol 21:42–50
Quiroz-Figueroa F, Méndez-Zeel M, Larqué-Saavedra A, Loyola-Vargas VM (2001) Picomolar concentrations of salicylates induce cellular growth and enhance somatic embryogenesis in Coffea arabica tissue culture. Plant Cell Rep 20:679–684. https://doi.org/10.1007/s002990100386
Rajasekaran K, Vine J, Mullins MG (1982) Dormancy in somatic embryos and seeds of Vitis: changes in endogenous abscisic acid during embryogeny and germination. Planta 154:139–144. https://doi.org/10.1007/BF00387907
Rajasekaran K, Hein MB, Vasil IK (1987) Endogenous abscisic acid and indole-3-acetic acid and somatic embryogenesis in cultured leaf explants of Pennisetum purpureum Schum.: effects in vivo and in vitro of glyphosate, fluridone, and paclobutrazol. Plant Physiol 84:47–51. https://doi.org/10.1104/pp.84.1.47
Ramarosandratana AV, Van Staden J (2004) Effects of auxins and 2,3,5-triiodobenzoic acid on somatic embryo initiation from Norway spruce zygotic embryos (Picea abies). Plant Cell Tiss Org Cult 79:105–107
Rao MV, Paliyath G, Ormrod DP, Murr DP, Watkins CB (1997) Influence of salicylic acid on H2O2 production, oxidative stress, and H2O2-metabolizing enzymes. Salicylic acid-mediated oxidative damage requires H2O2. Plant Physiol 115:137–149. https://doi.org/10.1104/pp.115.1.137
Romani RJ, Hess BM, Leslie CA (1989) Salicylic acid inhibition of ethylene production by apple discs and other plant tissues. J Plant Growth Regul 8:63–69. https://doi.org/10.1007/BF02024927
Ruduś I, Weiler E, Kępczynska E (2009) Do stress-related phytohormones, abscisic acid and jasmonic acid play a role in the regulation of Medicago sativa L. somatic embryogenesis? Plant Growth Regul 59:159–169. https://doi.org/10.1007/s10725-009-9399-3
Sáenz L, Azpeitia A, Oropeza C, Jones LH, Fuchsova K, Spichal L, Strnad M (2010) Endogenous cytokinins in Cocos nucifera L. in vitro cultures obtained from plumular explants. Plant Cell Rep 29:1227–1234. https://doi.org/10.1007/s00299-010-0906-9
Sakhanokho HF, Rajasekaran K, Kelley RY (2009) Somatic embryogenesis in Hedychium bousigonianum. Hortic Sci 44:1487–1490
Schiavone FM, Cooke TJ (1987) Unusual patterns of somatic embryogenesis in domesticated carrot: developmental effects of exogenous auxins and auxin transport inhibitors. Cell Diff 21:53–62
Senger S, Mock H-P, Conrad U, Manteuffel R (2001) Immunomodulation of ABA function affects early events in somatic embryo development. Plant Cell Rep 20:112–120. https://doi.org/10.1007/s002990000290
Somleva MM, Kapchina V, Alexieva V, Golovinsky E (1995) Anticytokinin effects on in vitro response of embryogenic and nonembryogenic genotypes of Dactylis glomerata L. Plant Growth Regul 16:109–112. https://doi.org/10.1007/BF00029530
Štefančič M, Štampar F, Veberič R, Osterc G (2007) The levels of IAA, IAAsp and some phenolics in cherry rootstock “GiSelA 5” leafy cuttings pretreated with IAA and IBA. Sci Hortic (Amsterdam) 112:399–405. https://doi.org/10.1016/j.scienta.2007.01.004
Su YH, Zhang XS (2009) Auxin gradients trigger de novo formation of stem cells during somatic embryogenesis. Plant Signal Behav 4:574–576. https://doi.org/10.4161/psb.4.7.8730
Su YH, Su YX, Liu YG, Zhang XS (2013) Abscisic acid is required for somatic embryo initiation through mediating spatial auxin response in Arabidopsis. Plant Growth Regul 69:167–176. https://doi.org/10.1007/s10725-012-9759-2
Tokuji Y, Kuriyama K (2003) Involvement of gibberellin and cytokinin in the formation of embryogenic cell clumps in carrot (Daucus carota). J Plant Physiol 160:133–141. https://doi.org/10.1078/0176-1617-00892
Vondráková Z, Krajňáková J, Fischerová L, Vágner M, Eliášová K (2016) Physiology and role of plant growth regulators in somatic embryogenesis. In: Park Y-S, Bonga J, Moon H-K (eds) Vegetative propagation of forest trees. National Institute of Forest Science (NIFoS), Seoul, pp 123–169
Yaseen M, Ahmad T, Sablok G, Standardi A, Hafiz IA (2013) Review: role of carbon sources for in vitro plant growth and development. Mol Biol Rep 40:2837–2849. https://doi.org/10.1007/s11033-012-2299-z
Żur I, Dubas E, Krzewska M, Waligórski P, Dziurka M, Janowiak F (2015) Hormonal requirements for effective induction of microspore embryogenesis in triticale (×Triticosecale Wittm.) anther cultures. Plant Cell Rep 34:47–62. https://doi.org/10.1007/s00299-014-1686-4
Acknowledgements
The authors thank dr. Piotr Waligórski (The Franciszek Górski Institute of Plant Physiology, PAS, Cracow, Poland) for help with HPLC analysis and Professor Jan J. Rybczyński (PAS Botanical Garden CBDC in Powsin, Warsaw, Poland) for valuable advice and creative discussions during preparation of manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
Polish National Center for Science (2011/03/B/NZ9/02472).
Additional information
Communicated by M. Lambardi.
Electronic supplementary material
Below is the link to the electronic supplementary material.

11738_2017_2577_MOESM1_ESM.jpg
Supplementary Fig. 1 The effect of SA, fluridone and TIBA on the concentrations of various endogenous cytokinins in the stipe explants of C. delgadii during 10 days of culture. c-Z cis zeatin, c-ZR cis zeatin riboside, iPA isopentenyladenosine, KinR kinetin riboside, t-Z trans zeatin, t-ZR trans zeatin riboside. Values marked with asterisk differ significantly from the control at the 0.05 level according to Student’s t test. Data represent mean ± standard deviation of three independent experiments, each consisting of 48 explants. (JPEG 491 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Grzyb, M., Kalandyk, A. & Mikuła, A. Effect of TIBA, fluridone and salicylic acid on somatic embryogenesis and endogenous hormone and sugar contents in the tree fern Cyathea delgadii Sternb.. Acta Physiol Plant 40, 1 (2018). https://doi.org/10.1007/s11738-017-2577-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-017-2577-4