Abstract
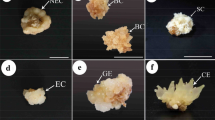
Enhanced somatic embryogenesis and plant regeneration have been obtained using young leaf bases of naked oat (Avena nuda) as explants by including salicylic acid (SA) and carrot embryogenic callus extracts (CECE) in media. A 5- and 4-fold improvement was achieved in somatic embryogenesis and plant regeneration on the corresponding media supplemented with 0.5 mM SA and CECE as compared to control, respectively. Some physiological and biochemical changes were assayed in both embryogenic callus (EC) and non-embryogenic callus (NEC). The results indicated that superoxide dismutase activity was stimulated and catalases and ascorbate peroxidase activities were inhibited, while the O2 - (superoxide anion) content was reduced and the hydrogen peroxide level was promoted in EC compared with NEC. Reduced malondialdehyde content and relative electrolyte leakage were also detected in EC.
Similar content being viewed by others
Abbreviations
- APX:
-
ascorbate peroxidase
- AsA:
-
ascorbic acid
- BA:
-
N6-benzylaminopurine
- CAT:
-
catalases
- CECE:
-
carrot embryogenic callus extracts
- 2,4-D :
-
2,4-dichlorophenoxyacetic acid
- EC:
-
embryogenic callus
- IAA:
-
indole-3-acetic acid
- KT:
-
kinetin
- MDA:
-
malondialdehyde
- MS:
-
Murashige and Skoog
- NAA:
-
α-naphthalene acetic acid
- NEC:
-
non-embryogenic callus
- REL:
-
relative electrolyte leakage
- ROS:
-
reactive oxygen species
- SA:
-
salicylic acid
- SOD:
-
superoxide dismutase
References
Aebi HE, (1983)Catalase. In: Bergmeyer HU, (ed) Methods of Enzymatic Analyses, Vol. 3 Verlag Chemie, Weinheim pp. 273–282
Beers EP, McDowell JM, (2001) Regulation and execution of programmed cell death in response to pathogens, stress and developmental cuesCurr. Opin. Plant Biol. 4: 561–567
Bozhkov PV, Filonova LH, Suarez MF, (2005) Programmed cell death in plant embryogenesisCurr. Top. Dev. Biol. 67: 135–79
Caliskan M, Turet M, Cuming AC, (2004) Formation of wheat ( Triticum aestivum L.) embryogenic callus involves peroxide-generating germin-like oxalate oxidasePlanta. 219: 132–140
Conrath U, Chen Z, Ricigliano JR, Klessig DF, (1995) Two inducers of plant defense responses, 2,6-dichloroisonicotinic acid and salicylic acid, inhibit catalase activities in tobacco Proc. Natl. Acad. Sci. USA. 92: 7143–7147
Cui K, Xing G, Liu X, Xing G, Wang Y, (1999) Effect of hydrogen peroxide on somatic embryogenesis of Lycium barbarum LPlant Sci. 146: 9–16
Desikan R, Mackerness RS-H, Hancock JT, Neill SJ, (2001) Regulation of the Arabidopsis transcriptome by oxidative stressPlant Physiol. 127: 159–172
Doke N, (1983) Involvement of superoxide anion generation in the hypersensitive response of potato tuber tissues to infection with an incompatible race of Phytophthora infestans and to the hyphal wall componentsPhysiol. Plant Pathol. 23: 345–357
Durner J, Klessig DF, (1995) Inhibition of ascorbate peroxidase by salicylic acid and 2,6-dichloroisonicotinic acid, two inducers of plant defense responsesProc. Natl. Acad. Sci. USA. 92: 11312–11316
Gless C, Lörz H, Jähne-Gärtner A, (1998) Transgenic oat plants obtained at high efficiency by microprojectile bombardment of leaf base segmentsJ. Plant Physiol. 152: 151–157
Guerin TF, Guerin PM, (1993) Recent developments in oat molecular biololgyPlant Mol. Biol. Rep. 11: 65–72
Hao L, Xi T, Li XY, Li YY, (1991) Embryogenesis in tissue derived from young leaf of Avena nudaActa Bot. Boreal.-Occident. Sin. 11: 271–275
Heath RL, Packer L, (1968) Photoperoxidation in isolated chloroplastsArch. Biochem. Biophys. 125: 189–198
Hutchinson MJ, Saxena PK, (1996) Acetylsalicylic acid enhances and synchronizes thidiazuron induced somatic embryogenesis in geranium (Pelargonium × hortorum Baily) tissue culturesPlant Cell Rep. 15: 512–515
Levine A, Tenhaken R, Dixon R, Lamb C, (1994) H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance responseCell. 79: 583–593
Libik M, Konieczny R, Pater B, lesak IS, Miszalski Z, (2005) Differences in the activities of some antioxidant enzymes and in H2O2 content during rhizogenesis and somatic embryogenesis in callus cultures of the ice plantPlant Cell Rep. 23: 834–841
Lowry DH, Rosebrough HJ, Farr AL, (1951) Protein measurement with Folin phenol reagentJ. Biol. Chem. 193: 262–275
Luo JP, Jiang ST, Pan LJ, (2001) Enhanced somatic embryogenesis by salicylic acid of Astragalus adsurgens Pall.: relationship with H2O2 production and H2O2-metabolizing enzyme activitiesPlant Sci. 161: 125–132
Marco A de, Roubelakis-Angelakis KA, (1996a) The complexity of enzymic control of hydrogen peroxide concentration may affect the regeneration potential of plant protoplastsPlant Physiol. 110: 137–145
Marco A de, Roubelakis-Angelakis KA, (1996b) Hydrogen peroxide plays a bivalent role in the regeneration of protoplastsJ. Plant Physiol. 149: 109–114
Misra HP, Fridovich I, (1977) Superoxide dismutase: a photochemical augmentation assayArch. Biochem. Biophys. 181: 308–312
Mordhorst AP, Toonen MAJ, de Vries SC, (1997) Plant embryogenesisCrit. Rev. Plant Sci. 16: 535–576
Murashige T, Skoog F, (1962) A revised medium for rapid growth and bioassays with tobacco culturesPhysiol. Plant 15: 473–497
Neill SJ, Desikan R, Clarke A, Hurst RD, Hancock JT, (2002a) Hydrogen peroxide and nitric oxide as signalling molecules in plantsJ. Exp. Bot. 53: 1237–1247
Neill SJ, Desikan R, Hancock JT, (2002b) Hydrogen peroxide signallingCurr. Opin. Plant Biol. 5: 388–395
Patterson BD, Macrae EA, Ferguson IB, (1984) Estimation of hydrogen peroxide in plant extracts using titanium (IV)Anal. Biochem. 134: 487–492
Papadakis AI, Roubelakis-Angelakis KA, (2002) Is oxidative stress responsible for plant protoplast recalcitrance? Plant PhysiolBiochem. 40: 549–559
Papadakis AI, Siminis CI, Roubelakis-Angelakis KA, (1999) Generation of active oxygen species in tobacco and grapevine protoplastsPlant Physiol. 121: 197–205
Papadakis AI, Siminis CI, Roubelakis-Angelakis KA, (2001) Reduced antioxidant machinery correlates with suppression of totipotency in plant protoplastsPlant Physiol. 126: 434–444
Pius J, George L, Eapen S, Rao PS, (1993) Enhanced plant regeneration in pearl millet (Pennisetum americanum) by ethylene inhibitors and cefotaximePlant Cell, Tiss. Org. Cult. 32: 91–96
Polle A, Chakrabati K, Schürmann W, Rennenber H, (1990) Composition and properties of hydrogen peroxide decomposing systems in extracellular and total extracts from needles of Norway spruce (Picea abies L., Karst.)Plant Physiol. 94: 312–319
Rao MV, Paliyath G, Ormrod DP, Murr DP, Watkins CB, (1997) Influence of salicylic acid on H2O2 production, oxidative stress, and H2O2-metabolizing enzymes: salicylic acid mediated oxidative damage requires H2O2Plant Physiol. 115: 137–149
Raskin I, (1992) Role of salicylic acid in plantsAnnu. Rev. Plant Physiol. Plant Mol. Biol. 43: 439–463
Roustan JP, Latche A, Fallot J, (1990) Inhibition of ethylene production and stimulation of carrot somatic embryogenesis by salicylic acidBiol. Plant. 32: 273–276
Siminis CI, Kanellis AK, Roubelakis-Angelakis KA, (1994) Catalase is differentially expressed in dividing and nondividing protoplastsPlant Physiol. 105: 1375–1383
Szalai G, Janda T, Paldi E, Szigeti Z, (1996) Role of light in post-chilling symptoms in maizeJ. Plant Physiol. 148: 378–383
Torbert KA, Rines HW, Kaeppler HF, (1998) Genetically engineering elite oat cultivarsCrop Sci. 38: 1685–1687
Zheng QS, Ju B, Liang LK, Xiao XH, (2005) Effects of antioxidants on the plant regeneration and GUS expressive frequency of peanut (Arachis hypogaea) explants by Agrobacterium tumefacien Plant Cell, Tiss. Org. Cult. 81: 83–90
Acknowledgement
We thank Stephen J. Herbert (University of Massachusetts) for comments on the manuscript. This research was supported by a grant from the Natural Science Foundation of Liaoning Province (20021022), P. R. China to L.H.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hao, L., Zhou, L., Xu, X. et al. The role of salicylic acid and carrot embryogenic callus extracts in somatic embryogenesis of naked oat (Avena nuda). Plant Cell Tiss Organ Cult 85, 109–113 (2006). https://doi.org/10.1007/s11240-005-9052-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-005-9052-4