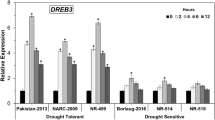
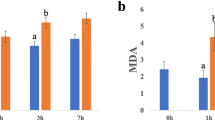
Abstract
Drought or water deficit stress is one of the main environmental stresses affecting plants, resulting in reduced productivity and crop loss. Black pepper, a major spice cultivated across the globe, is drought sensitive and water stress often results in plant death. The present study compared the difference in physiological parameters: relative water content (RWC) and cell membrane leakage, and also analyzed the differential expression of 11 drought responsive genes in drought tolerant and drought sensitive black pepper genotypes. Tolerant black pepper genotype exhibited significantly higher RWC and lower cell membrane leakage 10 days after stress induction than the sensitive genotype. The relative expressions of the 11 selected drought responsive genes were normalized against ubiquitin and RNA-binding protein which was identified as the most stable reference genes in black pepper under the present experimental condition using the RefFinder software. Dehydrin showed the highest transcript accumulation in both the black pepper genotypes under drought stress condition and the relative expression of the gene was higher in the tolerant genotype compared to the susceptible. Similar pattern of higher relative expression was also observed in the stress responsive gene, osmotin. The membrane protein aquaporin and the transcription factor bZIP were relatively down-regulated in the tolerant genotype. The differential expression of these important drought responsive genes in tolerant genotype of black pepper indicates its further usefulness in developing varieties with improved water stress tolerance.



Similar content being viewed by others
References
Aharon R, Shahak Y, Wininger S, Bendov R, Kapulnik Y, Galili G (2003) Overexpression of a plasma membrane aquaporin in transgenic tobacco improves plant vigor under favorable growth conditions but not under drought or salt stress. Plant Cell 15:439–447
Altinkut A, Kazan K, Ipekci Z, Gozukirmizi N (2001) Tolerance to paraquat is correlated with the traits associated with water stress tolerance in segregating F2 populations of barley and wheat. Euphytica 121:8186
Al-Whaibi MH (2011) Plant heat-shock proteins: a mini review. J King Saud Univ Sci 23:139–150
Andersen CL, Jensen JL, Orntoft TF (2004) Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res 64:5245–5250
Artico S, Nardeli SM, Brilhante O, Fátima MG, Alves-Ferreira M (2010) Identification and evaluation of new reference genes in Gossypium hirsutum for accurate normalization of real-time quantitative RT-PCR data. BMC Plant Biol 10:49
Ashraf M (2010) Inducing drought tolerance in plants: recent advances. Biotechnol Adv 28:169–183
Babitha KC, Ramu SV, Nataraja KN et al (2015) EcbZIP60, a basic leucine zipper transcription factor from Eleusine coracana L. improves abiotic stress tolerance in tobacco by activating unfolded protein response pathway. Mol Breed 35:181
Bajji M, Kinet JM, Lutts S (2001) The use of the electrolyte leakage method for assessing cell membrane stability as a water stress tolerance test in durum wheat. Plant Growth Regul 36:61–70
Baldoni E, Genga A, Cominelli E (2015) Plant MYB transcription factors: their role in drought response mechanisms. Int J Mol Sci 16(7):15811–15851
Bartels D, Sunkars R (2005) Drought and salt tolerance in plants. CRC Crit Rev Plant Sci 24:23–58
Barthakur S, Babu V, Bansal KC (2001) Over expression of osmotin induces proline accumulation and confers tolerance to osmotic stress in transgenic tobacco. J Plant Biochem Biotechnol 10:31–37
Battaglia M, Olvera-Carrillo Y, Garciarrubio A, Campos F, Covarrubias AA (2008) The enigmatic LEA proteins and other hydrophilins. Plant Physiol 148:6–24
Cellier F, Conejero G, Breitler JC, Casse F (1998) Molecular and physiological responses to water deficit in drought-tolerant and drought-sensitive lines of sunflower. Plant Physiol 116:319–328
Chen H, Chen W, Zhou J, He H, Chen L, Chen H et al (2012) Basic leucine zipper transcription factor OsbZIP16 positively regulates drought resistance in rice. Plant Sci 193–194:8–17
Close TJ (1996) Dehydrins: emergence of a biochemical role of a family of plant dehydration proteins. Plant Physiol 97:795–803
Colom MR, Vazzana C (2003) Photosynthesis and PSII functionality of drought-resistant and droughtsensitive weeping lovegrass plants. Environ Exp Bot 49:135–144
Fleige S, Pfaffl MW (2006) RNA intergrity and the effect on the real-time qRT-PCR performance. Mol Aspects Med 27:126–139
George JK, Kumar VIP, Anandaraj M (2012) Transcriptomics approaches for gene discovery in plants—a case study in Piper. Agrotechnology 1:2
Goel D, Singh AK, Yadav V, Babbar SB, Bansal KC (2010) Overexpression of osmotin gene confers tolerance to salt and drought stresses in transgenic tomato (Solanum lycopersicum L). Protoplasma 245:133–141
Hanin M, Brini F, Ebel C, Toda Y, Takeda S, Masmoudi K (2011) Plant dehydrins and stress tolerance. Plant Signal Behav 6:1503–1509
Hong Y, Zhang H, Huang L, Li D, Song F (2016) Overexpression of a stress-responsive NAC transcription factor gene ONAC022 improves drought and salt tolerance in rice. Front Plant Sci 7:4
Jamaux I, Steinmertz A, Belhassen E (1997) Looking for molecular and physiological markers of osmotic adjustment in sunflower. New Phytol 137:117–127
Kandiannan K, Krishnamurthy KS, Anke Gowda SJ, Anandaraj M (2014) Climate change and black pepper production. Indian J Arecanut Spices Med Plants 16:31–37
Kang J, Choi H, Im M, Kim SY (2002) Arabidopsis basic leucine zipper proteins that mediate stress-responsive abscisic acid signaling. Plant Cell 14:343–357
Krishnamurthy KS, Saji KV (2000) Response of piper species to water stress. Indian J Hortic 63:433–438
Krishnamurthy KS, Ankegowda SJ, George JK (1998) Impact of water stress on some physiological parameters in black pepper. In: Sadanandan AK, Krishnamurthy KS, Kandiannan K, Korikanthimath VS (eds) Water and nutrient management for sustainable production and quality of spices. Indian Society for Spices, Calicut, pp 153–157
Kumar SA, Kumari PH, Kumar GS, Mohanalatha C, Kishor PBK (2015) Osmotin: a plant sentinel and a possible agonist of mammalian adiponectin. Front Plant Sci 6:163
Lata C, Prasad M (2011) Role of DREBs in regulation of abiotic stress responses in plants. J Exp Bot 62:4731–4748
Li Y, Feng D, Zhang D, Su J, Zhang Y, Li Z, Mu P, Liu B, Wang H, Wang J (2012) Rice MAPK phosphatase IBR5 negatively regulates drought stress tolerance in transgenic Nicotiana tabacum. Plant Sci 10:188–189
Li J, Ban L, Wen H, Wang Z, Dzyubenko N, Chapurin V, Gao H, Wang X (2015) An aquaporin protein is associated with drought stress tolerance. Biochem Biophys Res Commun 459:208–213
Liu C, Wu Y, Wang X (2012) bZIP transcription factor OsbZIP52/RISBZ5: a potential negative regulator of cold and drought stress response in rice. Planta 235:1157–1169
Mawlong I, Ali K, Srinivasan R et al (2015) Functional validation of a drought-responsive AP2/ERF family transcription factor-encoding gene from rice in Arabidopsis. Mol Breed 35:163
Molaei P, Ebadi A, Namvar A, Khandan Bejandi T (2012) Water relation, solute accumulation and cell membrane injury in sesame (Sesamum indicum L.) cultivars subjected to water stress. Ann Biol Res 3:1833–1838
Omar SA, Elsheery NI, Kalaji HM, Ebrahim MKH, Pietkiewicz S, Lee CH, Allakhverdiev SI, Xu ZF (2013) Identification and differential expression of two dehydrin cDNAs during maturation of Jatropha curcas seeds. Biochemistry (Moscow) 78(5):485–495
Park W, Scheffler BE, Bauer PJ, Campbell BT (2012) Genome-wide identification of differentially expressed genes under water deficit stress in upland cotton (Gossypium hirsutum L.). BMC Plant Biol 12:90
Parkhi V, Kumar V, Sunilkumar G, Campbell LM, Singh NK (2009) Expression of apoplastically secreted tobacco Osmotin in cotton confers drought tolerance. Mol Breed 23:625–639
Pfaffl MW, Tichopad A, Prgomet C, Neuvians TP (2004) Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper-Excel-based tool using pair-wise correlations. Biotechnol Lett 26:509–515
Pillay VS, Sasikumaran S, Ibrahim KK (1988) Effect of rainfall pattern on the yield of black pepper. In: Agrometeorology of plantation crops (Pl. 152–159). Kerala Agricultural University, Trichur
Raghothama KG, Liu D, Nelson DE, Hasegawa PM, Bressan RA (1993) Analysis of an osmotically regulated pathogenesis-related osmotin gene promoter. Plant Mol Biol 23:1117–1128
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3:1101–1108
Shao HB, Chu LY, Jaleel CA, Manivannan P, Panneerselvam R, Shao MA (2009) Understanding water deficit stress-induced changes in the basic metabolism of higher plants-biotechnologically and sustainably improving agriculture and the eco-environment in arid regions of the globe. Crit Rev Biotechnol 29:131–151
Shinozaki K, Yamaguchi-Shinozaki K (2007) Gene networks involved in drought stress response and tolerance. J Exp Bot 58:221–227
Shinozaki K, Yamaguchi-Shinozaki K, Seki M (2003) Regulatory network of gene expression in the drought and cold stress responses. Curr Opin Plant Biol 6:410–417
Silver N, Best S, Jiang J, Thein SL (2006) Selection of housekeeping genes for gene expression studies in human reticulocytes using real-time PCR. BMC Mol Biol 7:33
Singh NK, Bracker CA, Hasegawa PM, Handa AK, Buckel S, Hermodson MA, Pfankoch E, Regnier FE, Bressan RA (1987) Characterization of osmotin: a thaumatin-like protein associated with osmotic adaptation in plant cells. Plant Physiol 85:529–536
Subramanyam K, Sailaja KV, Subramanyam K, Rao DM, Lakshmidevi K (2011) Ectopic expression of an Osmotin gene leads to enhanced salt tolerance in transgenic chilli pepper (Capsicum annum L). Plant Cell Tiss Organ Cult 105:181–192
Tunnacliffe A, Wise MJ (2007) The continuing conundrum of the LEA proteins. Naturwissenschaften 94:791–812
Vandesompele J, De Preter K, Pattyn F et al (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:RESEARCH0034
Xie C, Zhang R, Qu Y, Miao Z, Zhang Y, Shen X, Wang T, Dong J (2012a) Overexpression of MtCAS31 enhances drought tolerance in transgenic Arabidopsis by reducing stomatal density. New Phytol 195:124–135
Xie F, Xiao P, Chen D, Xu L, Zhang B (2012b) miRDeepFinder: a miRNA analysis tool for deep sequencing of plant small RNAs. Plant Mol Biol 80:75–84
Yamaguchi-Shinozaki K, Shinozaki K (2005) Organization of cis-acting regulatory elements in osmotic-and cold-stress-responsive promoters. Trends Plant Sci 10:88–94
Yang Y, He M, Zhu Z et al (2012) Identification of the dehydrin gene family from grapevine species and analysis of their responsiveness to various forms of abiotic and biotic stress. BMC Plant Biol 12:140
Zhang Y, Shih DS (2007) Isolation of an osmotin-like protein gene from strawberry and analysis of the response of this gene to abiotic stresses. J Plant Physiol 164:68–77
Zhang J, Nguyen HT, Blum A (1999) Genetic analysis of osmotic adjustment in crop plants. J Exp Bot 50(332):291–302
Zhang N, Si H, Wen G, Du H, Liu B, Wang D (2011) Enhanced drought and salinity tolerance in transgenic potato plants with a BADH gene from spinach. Plant Biotechnol Rep 5:71
Zhu B, Chen TH, Li PH (1995) Activation of two osmotin-like protein genes by abiotic stimuli and fungal pathogen in transgenic potato plants. Plant Physiol 108:929–937
Acknowledgements
The authors acknowledge the funding from the Indian Council of Agricultural Research through Centre for Agricultural Bioinformatics (CABin scheme, operating through ICAR, Indian Agricultural Statistics Research Institute, New Delhi, India).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Y Wang.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
George, K.J., Malik, N., Vijesh Kumar, I.P. et al. Gene expression analysis in drought tolerant and susceptible black pepper (Piper nigrum L.) in response to water deficit stress. Acta Physiol Plant 39, 104 (2017). https://doi.org/10.1007/s11738-017-2398-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-017-2398-5