Abstract
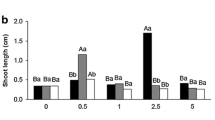
The influence of Putrescine (Put) on the growth and elicitation of anthocyanin in callus cultures of Daucus carota var. Nantes scarlet was investigated through the use of α-DL-difluoromethylarginine (DFMA), the polyamine (PA) biosynthetic inhibitor. It was observed that the addition of Put (0.05 mM) resulted in enhancement of growth and anthocyanin content. The anthocyanin content was found to be enhanced by 1.68 fold on the 21st day as compared to the untreated controls. The PA inhibitor was found to result in lowering of the growth and the anthocyanin accumulation, which could be partially restored by the addition of Put in combination with this inhibitor. The levels of Ca2+ ATPase were also found to be elevated in treatment with Put suggesting the involvement of calcium in the elicitation of anthocyanin. The endogenous titres of PAs and the ethylene production under these treatments were also studied. The treatment with DFMA resulted in lower levels of endogenous PAs and higher levels of ethylene. Lowering of ethylene by putrescine treatment shows that PA treatment also inhibited ethylene formation, which would also imply that endogenous ethylene does not influence anthocyanin production in carrot callus cultures.
Similar content being viewed by others
Abbreviations
- PAs:
-
Polyamines
- Put:
-
Put-rescine
- Spd:
-
Spermidine
- Spm:
-
Spermine
- DFMA:
-
α-DL-difluoromethylarginine
- DFMO:
-
α-DL-difluoromethylornithine
- SAM:
-
S-adenosylmethionine
- ODC:
-
Ornithine decarboxylase
- ACC:
-
aminocyclopropane-carboxylic acid
- 2,4-D:
-
2,4-Dichlorophenoxy acetic acid
- Kn:
-
Kinetin
- IAA:
-
Indole 3- acetic acid
- PCA:
-
perchloric acid
References
Antognoni F.R., Pistocchi P., Casali P., Bagni N. 1995. Does calcium regulate polyamine uptake in carrot protoplasts? Plant Physiol. Biochem., 33: 701–708.
Apelbaum A., Burgoon A.C., Anderson J.D., Lieberman M., Ben-Arie R., Mattoo A.K. 1981. Polyamines inhibit biosynthesis of ethylene in higher-plant tissue and fruit protoplasts. Plant Physiol., 68: 453–456.
Apelbaum A., Vinkler C., Sfakiotakis E., Dilley D.R. 1984. Increased mitochondrial DNA and RNA polymerase activity in ethylene-treated potato tubers. Plant Physiol., 76: 461–464.
Bagni N., Fracassini D.S., Torrigiani P. 1982. Polyamines and cellular growth processes in higher plants. In: Plant Growth Substances, ed. by P.F. Wareing, Academic Press, London: 473–482.
Bais H.P., Sudha G., Ravishanker G.A. 2000. Enhancement of growth and coumarin production in hairy root cultures of witloof chicory (Cichorium intybus L. cv. Lucknow local) under the influence of fungal elicitors. J Biosci. Bioeng., 90: 648–653.
Baykov A.A., Evtushenko O.A., Avaeva S.M. 1988. A malachite green procedure for orthophosphate determination and its use in alkaline phosphatase-based enzyme immunoassay. Anal. Biochem., 171: 266–270.
Ben-Arie R., Lurie S., Mattoo A.K. 1982. Temperature-dependent inhibitory effects of calcium and spermine on ethylene biosynthesis in apple discs correlate with changes in microsomal membrane microviscosity. Plant Sci. Lett., 24: 239–247.
Berlin J., Forche E. 1981. DL-α-difluoromethyl-ornithine causes enlargement of cultured tobacco cells. Z. Pflanzenphysiol., 101: 272–282.
Berta G., Altamura M.M., Fusconi A., Cerruti F., Capitani F., Bagni N. 1997. The plant cell wall is altered by inhibition of polyamine biosynthesis. New Phytol., 137: 569–577.
Biddington N.L. 1992. The influence of ethylene in plant tissue culture. Plant Growth Regul., 11: 173–178.
Chi C.L., Lin W.S., Lee J.E.E., Pua E.C. 1994. Role of polyamines on de novo shoot morphogenesis from cotyledons of B. campestris spp. pekinesis (lour) olsson, in vitro. Plant Cell Rep., 13: 323–329.
Cohen E., Arad S.M., Heimer Y.M., Mizrahi Y. 1982. Participation of ornithine decarboxylase in early stages of tomato fruit development. Plant Physiol., 70: 540–543.
Evans P.T., Malmberg R.L. 1989. Do polyamines have a role in plant development? Annu. Rev. Plant Physiol. Plant Mol. Biol., 40: 235–269.
Even-Chen Z., Mattoo A.K., Goren R. 1982. Inhibition of ethylene biosynthesis by aminoethoxyvinylglycine and by polyamines shunt label from C14-methionine into spermidine in aged orange peel discs. Plant Physiol., 69: 385–388.
Fienberg A.A., Choi J.H., Lubich W.P., Sung Z.R. 1984. Developmental regulation of polyamine metabolism in growth and differentiation of carrot culture. Planta, 162: 532–539.
Feirer R.P., Mignon G., Litvay J.D. 1984. Arginine decarboxylase and polyamines required for embryogenesis in the wild carrot. Science., 223: 1433–1435.
Flores H.E., Galston A.W. 1982. Analysis of polyamines in higher plants. Plant Physiol., 69: 701–706.
Freund J.E., Perles B.M. 1999. Sampling and sampling distributions. In: Statistics. A first course. Prentice Hall International, Inc. pp. 261–288.
Fuhrer J., Kaur-Sawhney R., Shih L.M., Galston A.W. 1982. Effects of exogenous 1,3-diaminopropane and spermidine on senescence of oat leaves. Plant Physiol., 70: 1597–1600.
Galston A.W., Kaur-Sawhney R.K. 1990. Polyamines in plant physiology. Plant Physiol., 94: 406–410.
Heimer Y.H., Mizrahi Y., Bachrach U. 1979. Ornithine decarboxylase activity in rapidly proliferating plant cells. FEBS Lett., 104: 146–149.
Kallio A., McCann P., Bey P. 1981. DL-α (difluoromethyl)arginine: a potent enzyme-activated inhibitor of bacterial arginine decarboxylase. Biochemistry., 20: 3163–3166.
Kurosaki, F., Matsushita M., Nishi, A. 1992. Essential role of polyamines in growth of cultured carrot cells. Phytochem., 31: 3889–3892.
Lowry O.H., Rosebrough N.J., Farr A.L., Randall R.J. 1951. Protein measurement with the folin phenol reagent. J Biol. Chem., 193: 265–275.
Mader J.C., Hanke D.E. 1997. Polyamine sparing may be involved in the prolongation of cell division due to inhibition of phenylpropanoid synthesis in cytokinin-starved soybean cells. J. Plant Growth Regul., 16: 89–93.
Mattoo A.K., Baker J.E., Chalutz E., Lieberman M. 1977. Effect of temperature on the ethylene-synthesising systems in apple, tomato and Penicillum. Plant Cell Physiol., 18: 715–719.
Meijer E.G.M., Simmonds J. 1988. Polyamine levels in relation to growth and somatic embryogenesis in tissue cultures of Medicago sativa L. J. Exp. Bot., 203: 787–794.
Metcalf B.W., Bey P., Danzin C., Jung M.J., Casara P., Ververt J.P. 1978. Catalytic irreversible inhibition of mammalian ornithine decarboxylase (E.C.4.1.1.17) by substrate and product analogs. J Amer. Chem. Soc., 100: 2551–2553.
Mizusaki S., Tanabe Y., Noguchi M., Tamaki E. 1973. Phytochemical studies on tobacco alkaloids. XVI. Changes in the activities of ornithine decarboxylase, putrescine N-methyltransferase, and N-methylputrescine oxidase in tobacco roots in relation to nicotine biosynthesis. Plant Cell Physiol., 14: 103–110.
Murashige M., and Skoog T. 1962. A revised medium for rapid growth and bioassay of tobacco tissue cultures. Physiol. Plant., 15: 473–497.
Palavan N., Galston A.W. 1982. Polyamine biosynthesis and titer during various developmental stages of Phaseolus vulgaris. Physiol. Plant., 55: 438–444.
Phillips R., Press M.C., Bingham L., Grimmer C. 1988. Polyamines in cultured artichoke explants: effects are primarily on xylogenesis rather than cell division. J Exp. Bot. 39: 473–480.
Slocum R.D., Kaur-Sawhney R., Galston A.W. 1984. The physiology and biochemistry of polyamines in plants. Arch. Biochem. Biophys., 235: 283–303.
Smith T.A., Best G.R., Abbott J., Clements E.D. 1978. Polyamines in Paul’s scarlet rose suspension cultures. Planta, 144: 63–68.
Stickland G.G., Sundarland N. 1972. Production of anthocyanins, flavonols and chlorogenic acid by cultured callus tissues of Happlopappus gracilis. Ann. Bot., 36: 443–457.
Stroinski A., Szczotka Z. 1989. Effect of cadmium and Phytophthora infestans on polyamine levels in potato leaves. Physiol. Plant., 77: 244–246.
Suttle J.C. 1981. Effect of polyamines on ethylene production. Phytochem., 20: 1477–1480.
Vambutas V.K., Racker E. 1965. Partial resolution of the enzymes catalyzing photophosphorylation. I. Stimulation of photophosphorylation by a preparation of latent, Ca++ dependent adenosine triphosphatase from chloroplasts. J Biol. Chem., 240: 2660–2667.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sudha, G., Ravishankar, G.A. Influence of putrescine on anthocyanin production in callus cultures of Daucus carota mediated through calcium ATPase. Acta Physiol Plant 25, 69–75 (2003). https://doi.org/10.1007/s11738-003-0038-8
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11738-003-0038-8