Abstract
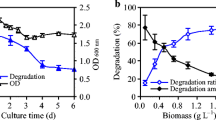
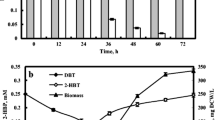
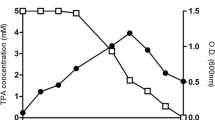
The biodesulfurization of dibenzothiophene (DBT) by growing and resting cells of Rhodococcus erythropolis PTCC 1767 was studied in this work. The effects of Tween 80 on cell growth and its DBT desulfurization ability were investigated in both aqueous and two-phase systems. The growth-supportive behavior of Tween 80 along with its role in increasing the DBT solubility provided an effective biocatalytic activity in the resting cells assay. The desulfurization capability was also dependent on the hydrocarbon fraction phase and the initial concentration of DBT. Three oil phase fractions of 25, 50, and 75%v/v were tested to evaluate the influence of oil phase presence on DBT desulfurization efficiency. A further decrease in desulfurization ability was occurred at higher oil phase ratios mainly due to a higher mass transfer limitation and lower DBT bioavailability. In the biphasic system, the increment of DBT desulfurization yield was followed by increasing DBT concentration up to 3.5 mM, and thereafter, the cessation of increasing trend occurred. Maximum specific production rate of 2-hydroxybiphenyl (2-HBP) with oil phase fraction of 25%v/v was obtained 0.0055 mmol g−1 h−1 at initial DBT and Tween 80 concentrations of 3.5 and 6 mM, respectively. Finally, kinetics of DBT desulfurization by growing cells in aqueous and biphasic systems were best fitted by the Haldane and Michaelis–Menten equations, respectively. The desulfurization activity of R. erythropolis was not repressed in the biphasic system for DBT concentration up to 7.13 mM, while product inhibition was observed at DBT concentration higher than 0.45 mM in the aqueous system.









Similar content being viewed by others
References
Abbasian F, Lockington R, Megharaj M, Naidu R (2016) Identification of a new operon involved in desulfurization of dibenzothiophenes using a metagenomic study and cloning and functional analysis of the genes. Enzyme Microb Technol 77:24–28. https://doi.org/10.1016/j.enzmictec.2016.02.009
Abin-Fuentes A, Leung JC, Mel-S M, Wang DI, Prather KL (2014) Rate-limiting step analysis of the microbial desulfurization of dibenzothiophene in a model oil system. Biotechnol Bioeng 111(5):876–884. https://doi.org/10.1002/bit.25148
Alcon A, Martin AB, Santos VE, Gomez E, Garcia-Ochoa F (2008) Kinetic model for DBT desulphurization by resting whole cells of Pseudomonas putida CECT5279. Biochem Eng J 39:486–495. https://doi.org/10.1016/j.bej.2007.11.005
Boltes K, del Aguila RA, García-Calvo E (2013) Effect of mass transfer on biodesulfurization kinetics of alkylated forms of dibenzothiophene by Pseudomonas putida CECT5279. J Chem Technol Biotechnol 88:422–431. https://doi.org/10.1002/jctb.3877
Boniek D, Figueiredo D, dos Santos AFB, de Resende Stoianoff MA (2015) Biodesulfurization: a mini review about the immediate search for the future technology. Clean Technol Environ Policy 17:29–37. https://doi.org/10.1007/s10098-014-0812-x
Calzada J, Zamarro MT, Alcon A, Santos VE, Diaz E, Garcia JL, Garcia-Ochoa F (2009) Analysis of dibenzothiophene desulfurization in a recombinant Pseudomonas putida strain. Appl Environ Microbiol 75(3):875–877. https://doi.org/10.1128/AEM.01682-08
Calzada J, Alcon A, Santos VE, Garcia-Ochoa F (2011) Mixtures of Pseudomonas putida CECT 5279 cells of different ages: optimization as biodesulfurization catalyst. Process Biochem 46:1323–1328. https://doi.org/10.1016/j.procbio.2011.02.025
Caro A, Boltes K, Leton P, Garcia-Calvo E (2008a) Description of by-product inhibition effects on biodesulfurization of dibenzothiophene in biphasic media. Biodegradation 19:599–611. https://doi.org/10.1007/s10532-007-9165-z
Caro A, Boltes K, Leton P, Garcia-Calvo E (2008b) Biodesulfurization of dibenzothiophene by growing cells of Pseudomonas putida CECT 5279 in biphasic media. Chemosphere 73:663–669. https://doi.org/10.1016/j.chemosphere.2008.07.031
Chen H, Zhang WJ, Cai YB, Zhang Y, Li W (2008) Elucidation of 2-hydroxybiphenyl effect on dibenzothiophene desulfurization by Microbacterium sp. strain ZD-M2. Bioresour Technol 99:6928–6933. https://doi.org/10.1016/j.biortech.2008.01.033
Chen S, Zhao C, Liu Q, Zhang X, Sun S, Zang M (2019) Biodesulfurization of diesel oil in oil–water two phase reaction system by Gordonia sp. SC-10. Biotechnol Lett 41:547–554. https://doi.org/10.1007/s10529-019-02663-9
Dejaloud A, Vahabzadeh F, Habibi A (2017) Ralstonia eutropha as a biocatalyst for desulfurization of dibenzothiophene. Bioprocess Biosyst Eng 40:969–980. https://doi.org/10.1007/s00449-017-1760-8
Derikvand P, Etemadifar Z, Biria D (2014) Taguchi optimization of dibenzothiophene biodesulfurization by Rhodococcus erythropolis R1 immobilized cells in a biphasic system. Int Biodeterior Biodegrad 86:343–348. https://doi.org/10.1016/j.ibiod.2013.10.006
Dinamarca MA, Rojas A, Baeza P, Espinoza G, Ibacache-Quiroga C, Ojeda J (2014) Optimizing the biodesulfurization of gas oil by adding surfactants to immobilized cell systems. Fuel 116:237–241. https://doi.org/10.1016/j.fuel.2013.07.108
Feng J, Zeng Y, Ma C, Cai X, Zhang Q, Tong M, Yu Bo X (2006) The surfactant tween 80 enhances biodesulfurization. Appl Environ Microbiol 72:7390–7393. https://doi.org/10.1128/AEM.01474-06
Guchhait S, Biswas D, Bhattacharya P, Chowdhury R (2005) Bio-desulfurization of model organo-sulfur compounds and hydrotreated diesel—experiments and modeling. Chem Eng J 112:145–151. https://doi.org/10.1016/j.cej.2005.05.006
Jia X, Wen J, Sun Z, Caiyin Q, Xie S (2006) Modeling of DBT biodegradation behaviors by resting cells of Gordonia sp. WQ-01 and its mutant in oil–water dispersions. Chem Eng Sci 61:1987–2000. https://doi.org/10.1016/j.ces.2005.10.045
Kazemi M, Raheb J (2004) Isolation and identification of a sulfide/sulfoxide monooxygenase gene from a newly isolated Rhodococcus Sp, strain FMF. J Sci Islam Repub Iran 15(2):119–126
Kilbane JJ II, Stark B (2016) Biodesulfurization: a model system for microbial physiology. World J Microbiol Biotechnol 32:137. https://doi.org/10.1007/s11274-016-2084-6
Kim YJ, Chang JH, Cho KS, Ryu HW, Chang YK (2004) A physiological study growth and dibenzothiophene (DBT) desulfurization characteristics of Gordonia sp. CYKS1. Korean J Chem Eng 21:436–441. https://doi.org/10.1007/BF02705433
Li J-L, Chen B-H (2009) Surfactant-mediated biodegradation of polycyclic aromatic hydrocarbons. Mater 2:76–94. https://doi.org/10.3390/ma2010076
Luo MF, Xing JM, Gou ZX, Li S, Liu HZ, Chen JY (2003) Desulfurization of dibenzothiophene by lyophilized cells of Pseudomonas delafieldii R-8 in the presence of dodecane. Biochem Eng J 13:1–6. https://doi.org/10.1016/S1369-703X(02)00078-5
Maghsoudi S, Vossoughi M, Kheirolomoom A, Tanaka E, Katoh S (2001) Biodesulfurization of hydrocarbons and diesel fuels by Rhodococcus sp. strain P32C1. Biochem Eng J 8:151–156. https://doi.org/10.1016/S1369-703X(01)00097-3
Martin AB, Alcon A, Santos VE, Garcia-Ochoa F (2005) Production of a biocatalyst of Pseudomonas putida CECT5279 for DBT biodesulfurization: influence of the operational condition. Energy Fuels 19:775–782. https://doi.org/10.1021/ef0400417
Martinez I, Santos VE, Alcon A, Garcia-Ochoa F (2015) Enhancement of the biodesulfurization capacity of Pseudomonas putida CECT5279 by co-substrate addition. Process Biochem 50:119–124. https://doi.org/10.1016/j.procbio.2014.11.001
Miao-dong W, Wei L, Yao S, Da-hui W, Hai F (2006) Effects of surfactant on biodesulfurization by Corynebacterium sp. ZD-1 in the presence of organic phase. J Zhejiang Univ Sci A 7:371–375. https://doi.org/10.1631/jzus.2006.AS0371
Mohebali G, Ball AS (2016) Biodesulfurization of diesel fuels-Past, present and future perspectives. Int Biodeterior Biodegrad 110:163–180. https://doi.org/10.1016/j.ibiod.2016.03.011
Raheb J, Naghdi S, Karkhane AA, Yakhchali B, Flint KP (2004) Nucleotide sequence of a desulfurization operon from a newly isolated Rhodococcus FMF. Shiraz E Med J 5(2)
Rhee SK, Chang JH, Chang YK, Chang HN (1998) Desulfurization of dibenzothiophene and diesel oils by a newly isolated Gordona strain, CYKS1. Appl Environ Microbiol 64(6):2327–2331
Schmidts T, Dobler D, Nissing C, Runkel F (2009) Influence of hydrophilic surfactants on the properties of multiple W/O/W emulsions. J Colloid Interface Sci 338:184–192. https://doi.org/10.1155/2017/5471376
Srinivasan K, Mahadevan R (2010) Characterization of proton production and consumption associated with microbial metabolism. BMC Biotechnol 10(2):1–10. https://doi.org/10.1186/1472-6750-10-2
Su T, Su J, Liu S, Zhang C, He J, Huang Y, Xu S, Gu L (2018) Structural and biochemical characterization of BdsA from Bacillus subtilis WU-S2B, a key enzyme in the “4S” desulfurization pathway. Front Microbiol 9:231. https://doi.org/10.3389/fmicb.2018.00231
Xia W (2018) A novel and effective method for removing organic sulfur from low rank coal. J Clean Prod 172:2708–2710. https://doi.org/10.1016/j.jclepro.2017.11.141
Yang J, Marison IW (2005) Two-stage process design for the biodesulphurisation of a model diesel by a newly isolated Rhodococcus globerulus DAQ3. Biochem Eng J 27:77–82. https://doi.org/10.1016/j.bej.2005.08.012
Ye JP, Zhang PY, Zhang GM, Wang SQ, Nabi M, Zhang Q, Zhang HB (2018) Biodesulfurization of high sulfur fat coal with indigenous and exotic microorganisms. J Clean Prod 197:562–570. https://doi.org/10.1016/j.jclepro.2018.06.223
Zhang SH, Chen H, Wei L (2013) Kinetic analysis of biodesulfurization of model oil containing multiple alkyl dibenzothiophenes. Appl Microbiol Biotechnol 97:2193–2200. https://doi.org/10.1007/s00253-012-4048-6
Zhao C, Luo KL (2018) Household consumption of coal and related sulfur, arsenic, fluorine and mercury emissions in China. Energy Policy 112:221–232. https://doi.org/10.1016/j.enpol.2017.10.021
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dejaloud, A., Habibi, A. & Vahabzadeh, F. DBT desulfurization by Rhodococcus erythropolis PTCC 1767 in aqueous and biphasic systems. Chem. Pap. 74, 3605–3615 (2020). https://doi.org/10.1007/s11696-020-01191-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01191-5