Abstract
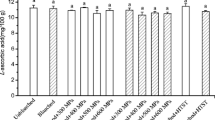
The trapiá, although it is a species present in almost the entire Brazilian territory, has limited consumption due to the absence of commercial plantations and general unawareness of its properties, aggravated by the difficulty of processing due to rapid enzymatic browning that affects the pulp when handled. In this study, the influence of enzymatic inactivation through thermal blanching on the changes in luminosity, bioactive compounds, and antioxidant activity of trapiá pulp was evaluated. Blanching showed optimal effects when the thermal process was applied for a duration of 15 min, as confirmed by the qualitative determination of peroxidase (POD), indicating the interruption of enzymatic activity. The study of instrumental luminosity kinetics (color parameter) of the sample without thermal blanching treatment (control) and with blanching indicated a degradation process over time governed by a first-order model. The thermal process influenced the levels of bioactive compounds, resulting in increased flavonoids, anthocyanins, and β-carotene. Blanching also led to an increase in antioxidant activity measured by ABTS and DPPH methods, with a significant increase in DPPH radical scavenging activity, reaching 96% higher than the unblanched pulp.



Similar content being viewed by others
Data Availability
Not applicable.
References
FGC Pareyn, JL Vieira, MAGariglio. Estatística Florestal da Caatinga. 2. ed. Brasília: Ministério do Meio Ambiente, Associação Plantas do Nordeste, (2015).
M Lacerda, H Lorenzi (2006). Frutas Brasileiras e Exóticas Cultivadas (De Consumo In Natura). Nova Odessa: Plantarum.
R.L. Soares Neto, F.Á.L. Magalhães, F.R.S. Tabosa, M.F. Moro, M.B.C. Silva, M.I.B. Loiola, Flora do Ceará, Brasil: Capparaceae. Rodriguésia 65(3), 671684 (2014)
X Cornejo, JR Maciel, JS Marques, RLS Neto, MB Costaesilva. Capparaceae. Lista de Espécies da Flora do Brasil (2015).
M. Hadidi, A. Ibarz, J. Conde, J. Pagan, Optimisation of steam blanching on enzymatic activity, color and protein degradation of alfalfa (Medicago sativa) to improve some quality characteristics of its edible protein. Food Chem. 276, 591–598 (2019). https://doi.org/10.1016/j.foodchem.2018.10.049
A.A. Escobar-Puentes, S.Y. Reyes-López, Á.D.J.R. Baltazar, V. López-Teros, A. Wall-Medrano, Molecular interaction of β-carotene with sweet potato starch: a bleaching-restitution assay. Food Hydrocoll. 127, 107522 (2022). https://doi.org/10.1016/j.foodhyd.2022.107522
H.W. Xiao, Z. Pan, L.Z. Deng, H.M. El-Mashad, X.H. Yang, A.S. Mujumdar, Q. Zhang, Recent developments and trends in thermal blanching–A comprehensive review. Inf. Process. Agric. 4, 101–127 (2017). https://doi.org/10.1016/j.inpa.2017.02.001
L.Z. Deng, A.S. Mujumdar, Q. Zhang, X.H. Yang, J. Wang, Z.A. Zheng, H.W. Xiao, Chemical and physical pretreatments of fruits and vegetables: effects on drying characteristics and quality attributes–a comprehensive review. Critical Rev. Food Sci. Nutr. 59, 1408–1432 (2019). https://doi.org/10.1080/10408398.2017.1409192
O.Y. Barrón-García, M. Gaytán-Martínez, A.K. Ramírez-Jiménez, I. Luzardo-Ocampo, G. Velazquez, E. Morales-Sánchez, Physicochemical characterization and polyphenol oxidase inactivation of Ataulfo mango pulp pasteurized by conventional and ohmic heating processes. Lwt 143, 111113 (2021). https://doi.org/10.1016/j.lwt.2021.111113
O.Y. Barrón-García, E. Morales-Sánchez, M. Gaytán-Martínez, Inactivation kinetics of Agaricus bisporus tyrosinase treated by ohmic heating: Influence of moderate electric field. Innov. Food Sci. Emerg. Technol. 56, 102179 (2019). https://doi.org/10.1016/j.ifset.2019.102179
M.A. Sheikh, C.S. Saini, H.K. Sharma, Synergistic effect of microwave heating and hydrothermal treatment on cyanogenic glycosides and bioactive compounds of plum (Prunus domestica L.) kernels: an analytical approach. Curr. Res. Food Sci. 5, 65–72 (2022). https://doi.org/10.1016/j.crfs.2021.12.007
Z.L. Deng, Z. Pan, A.S. Mujumdar, J.H. Zhao, Z.A. Zheng, Z.J. Gao, H.W. Xiao, High-humidity hot air impingement blanching (HHAIB) enhances drying quality of apricots by inactivating the enzymes, reducing drying time and altering cellular structure. Food Control 96, 104–111 (2019). https://doi.org/10.1016/j.foodcont.2018.09.008
A. Isidoro, V. Battestin, Determinação qualitativa de enzimas deteriorativas catalase e peroxidase em alcachofras provenientes da cidade de São Roque. SP. Revista Scientia Vitae 2, 55–60 (2014)
L.S. Chang, R. Karim, S.M. Abdulkarim, Y.A. Yusof, H.M. Ghazali, Storage stability, color kinetics and morphology of spray-dried soursop (Annona muricata L.) powder: effect of anticaking agents. Int. J. Food Prop. 21, 1937–1954 (2018). https://doi.org/10.1080/10942912.2018.1510836
T.P. Labuza, Shelf-life dating of foods (Food & Nutrition Press Inc., Trumbull, 1982)
HMC Azeredo, JAF Faria, ES Brito. Fundamentos de cinética de degradação e estimativa de vida de prateleira In: HMC Azeredo. Fundamentos de estabilidade de alimentos. Fortaleza: Editora Técnica. (2004).
A. Waterhouse, Folin-ciocalteau micro method for total phenol in wine. Am. J. Enol. Vitic. 48, 357–363 (2006)
AOAC, B. A. M. Association of official analytical chemists. Official methods of analysis, 12 (1990).
MDT Benassi, AJ Antunes. A comparison of metaphosphoric and oxalic acids as extractants solutions for the determination of vitamin C in selected vegetables. Arq. Biol. Tecnol. 507–13 (1988).
F.J. Francis, Analysis of anthocyanins. Anthocyanins Food Colors 1, 280 (1982)
K.C. Santos, J.S. Guedes, M.L. Rojas, G.R. Carvalho, P.E.D. Augusto, Enhancing carrot convective drying by combining ethanol and ultrasound as pre-treatments: effect on product structure, quality, energy consumption, drying and rehydration kinetics. Ultrason. Sonochem. 70, 105304 (2021). https://doi.org/10.1016/j.ultsonch.2020.105304
M.S. Lima, I.D.S.V. Silani, I.M. Toaldo, L.C. Corrêa, A.C.T. Biasoto, G.E. Pereira, J.L. Ninow, Phenolic compounds, organic acids and antioxidant activity of grape juices produced from new Brazilian varieties planted in the Northeast Region of Brazil. Food Chem. 161, 94–103 (2014). https://doi.org/10.1016/j.foodchem.2014.03.109
H.C. Deeth, Heat-induced inactivation of enzymes in milk and dairy products. A review. Int. Dairy J. 121, 105104 (2021). https://doi.org/10.1016/j.idairyj.2021.105104
M.L.S.O. Lima, P.S. Alceu, C.D.C.S. Gonçalves, Investigação qualitativa da biodegradação de corantes têxteis do tipo azo utilizando células de batata doce (Ipomoea batatas) como fonte de biocatalisador. Revista Sítio Novo 4, 30–39 (2020). https://doi.org/10.47236/2594-7036.2020.v4.i2.30-39p
B.E. Halpin, C.Y. Lee, Effect of blanching on enzyme activity and quality changes in green peas. J. Food Sci. 52, 1002–1005 (1987)
P. Singhal, S. Satya, S.N. Naik, Blanching: a sustainable and effective treatment for extending shelf life of bamboo shoots. Food Chem. Adv. 2, 100179 (2023). https://doi.org/10.1016/j.focha.2022.100179
H.W. Xiao, X.D. Yao, H. Lin, W.X. Yang, J.S. Meng, Z.J. Gao, Effect of SSB (superheated steam blanching) time and drying temperature on hot air impingement drying kinetics and quality attributes of yam slices. J. Food Process Eng 35, 370–390 (2012). https://doi.org/10.1111/j.1745-4530.2010.00594.x
J. Zhu, X. Xia, F. Zhang, S. Song, H. Cui, K. Hayat, C.T. Ho, Taste characteristic and the mechanism of light-colored Maillard reaction products derived from gluten hydrolysate. Food Biosci. 52, 102394 (2023). https://doi.org/10.1016/j.fbio.2023.102394
T. Henle, Protein-bound advanced glycation endproducts (AGEs) as bioactive amino acid derivatives in foods. Amino Acids 29, 313–322 (2005)
J.M. Silván, J. Lagemaat, A. Olano, M.D.D. Castillo, Analysis and biological properties of amino acid derivates formed by Maillard reaction in foods. J. Pharm. Biomed. Anal. 41, 1543–1551 (2006). https://doi.org/10.1016/j.jpba.2006.04.004
Z. Zhang, B. Wang, B. Adhikari, Maillard reaction between pea protein isolate and maltodextrin via wet-heating route for emulsion stabilisation. Future Foods 6, 100193 (2022). https://doi.org/10.1016/j.fufo.2022.100193
H. Zhao, Endogenous antioxidants and antioxidant activities of beers, in Processing and impact on antioxidants in beverages. (Academic Press, Cambridge, 2014), pp.15–24. https://doi.org/10.1016/B978-0-12-404738-9.00002-7
A. Garvín, R. Ibarz, A. Ibarz, Photodegradation Foods (2021). https://doi.org/10.1016/B978-0-08-100596-5.22950-8
B. Ling, J. Tang, F. Kong, E.J. Mitcham, S. Wang, Kinetics of food quality changes during thermal processing: a review. Food Bioprocess Technol. 8, 343–358 (2015). https://doi.org/10.1007/s11947-014-1398-3
G. Akar, I.B. Mazı, Color change, ascorbic acid degradation kinetics, and rehydration behavior of kiwifruit as affected by different drying methods. J. Food Process Eng 42, e13011 (2019). https://doi.org/10.1111/jfpe.13011
E.E. Badin, Y.E. Rossi, M.A. Montenegro, A. Ibarz, P.D. Ribotta, A.R. Lespinard, Thermal processing of raspberry pulp: Effect on the color and bioactive compounds. Food Bioprod. Process. 124, 469–477 (2020). https://doi.org/10.1016/j.fbp.2020.08.016
F. Salehi, Color changes kinetics during deep fat frying of kohlrabi (Brassica oleracea var. gongylodes) slice. Int. J. Food Prop. 22, 511–519 (2019). https://doi.org/10.1080/10942912.2019.1593616
B. Nayak, R.H. Liu, J. Tang, Effect of processing on phenolic antioxidants of fruits, vegetables, and grains—a review. Crit. Rev. Food Sci. Nutr. 55, 887–918 (2015). https://doi.org/10.1080/10408398.2011.654142
E. Mecha, S.T. Leitão, B. Carbas, A.T. Serra, P.M. Moreira, M.M. Veloso, M.R. Bronze, Characterization of soaking process’ impact in common beans phenolic composition: contribute from the unexplored Portuguese germplasm. Foods 8, 296 (2019). https://doi.org/10.3390/foods8080296
R.F. Dibanda, E.P. Akdowa, Q.M. Tongwa, Effect of microwave blanching on antioxidant activity, phenolic compounds and browning behaviour of some fruit peelings. Food Chem. 302, 125308 (2020). https://doi.org/10.1016/j.foodchem.2019.125308
J. Mandha, H. Shumoy, A.O. Matemu, K. Raes, Characterization of fruit juices and effect of pasteurization and storage conditions on their microbial, physicochemical, and nutritional quality. Food Biosci. 51, 102335 (2023). https://doi.org/10.1016/j.fbio.2022.102335
J.D.P. Matos, R.M.F. Figueirêdo, A.J.M. Queiroz, L.P.F.R. Silva, M.S. Moraes, S.N. Silva, Z.R.T. Costa, L.M.S. Rodrigues, A.F. Vieira, D.D.F. Leite, P.B. Silva, J.A.D. Esmero, F.S. Santos, M.G. Gonçalves, Physical-chemical and bioactive characterization of jambolan and acerola mixed pulp. Sylwan 165(1), 45–61 (2021)
J. Sung, J.H. Suh, Y. Wang, Effects of heat treatment of mandarin peel on flavonoid profiles and lipid accumulation in 3T3-L1 adipocytes. J. Food Drug Anal. 27, 729–735 (2019). https://doi.org/10.1016/j.jfda.2019.05.002
S. Oancea, A review of the current knowledge of thermal stability of anthocyanins and approaches to their stabilization to heat. Antioxidants 10, 1337 (2021). https://doi.org/10.3390/antiox10091337
S. Xie, Y. Liu, H. Chen, Z. Zhang, M. Ge, Anthocyanin degradation and the underlying molecular mechanism in a red-fleshed grape variety. Lwt 151, 112198 (2021). https://doi.org/10.1016/j.lwt.2021.112198
S. Deepika, P.P. Sutar, Combining osmotic–steam blanching with infrared–microwave–hot air drying: production of dried lemon (Citrus limon L.) slices and enzyme inactivation. Drying Technol. 36, 1719–1737 (2018). https://doi.org/10.1080/07373937.2017.1422744
L. Yuan, F. Cheng, J. Yi, S. Cai, X. Liao, F. Lao, L. Zhou, Effect of high-pressure processing and thermal treatments on color and in vitro bioaccessibility of anthocyanin and antioxidants in cloudy pomegranate juice. Food Chem. 373, 131397 (2022). https://doi.org/10.1016/j.foodchem.2021.131397
D. Yıldız, D.B. Gürel, Ö. Çağındı, S. Kayaardı, Heat treatment and microwave applications on homemade sour cherry juice: the effect on anthocyanin content and some physicochemical properties. Curr. Plant Biol. 29, 100242 (2022). https://doi.org/10.1016/j.cpb.2022.100242
B. Hiranvarachat, S. Devahastin, N. Chiewchan, G.V. Raghavan, Structural modification by different pretreatment methods to enhance microwave-assisted extraction of β-carotene from carrots. J. Food Eng. 115, 190–197 (2013). https://doi.org/10.1016/j.jfoodeng.2012.10.012
O.J. Jesús, L.A. Cira-Chávez, A.A. Gardea-Béjar, J.C. Guevara-Arauza, D.R. Sepúlveda, J. Reyes-Hernández, S. Ruiz-Cruz, Effect of heat treatment on the content of some bioactive compounds and free radical-scavenging activity in pungent and non-pungent peppers. Food Res. Int. 50, 519–525 (2013). https://doi.org/10.1016/j.foodres.2011.01.006
P. Schober, C. Boer, L.A. Schwarte, Correlation coefficients: appropriate use and interpretation. Anesth. Analg. 126, 1763–1768 (2018). https://doi.org/10.1213/ANE.0000000000002864
R. Indiarto, R. Reni, G.L. Utama, E. Subroto, A.D. Pangawikan, M. Djali, The physicochemical, antioxidant, and sensory properties of chocolate biscuits incorporated with encapsulated mangosteen (Garcinia mangostana L.) peel extract. Int. J. Food Prop. 26, 122–138 (2023). https://doi.org/10.1080/10942912.2022.2159429
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Moura, H.V., de Figueirêdo, R.M.F., Queiroz, A.J.d. et al. Blanching applied in enzymatic inactivation and its influence on browning, bioactive compounds, and antioxidant activity of trapiá (Crataeva tapia L.) pulp. Food Measure 18, 2894–2902 (2024). https://doi.org/10.1007/s11694-024-02367-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-024-02367-3