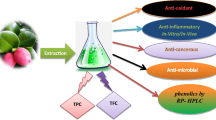
Abstract
Due to the high nutritional value as well as the appreciated sensory characteristic Citrus crop is one of the most popular fruits over the world. Albedo and flavedo are commonly discarded as waste and could represent an important by-product of food industry. In a circular economy contest we carried out a characterization of albedo and flavedo of Citrus limon var pompia (pompia) followed by the evaluation of antioxidant potential and cytotoxic activity. The chemical analysis showed the presence of several functional ingredients such as the flavanones, naringin, neohesperidin, nereriocitrin and hesperidin while quinic acid resulted the main phenolic acid detected. The flavedo extract exerted a complete inhibition of the cholesterol oxidative process from 25 μg of extract. Pompia extracts showed an inhibition of lipid oxidation in the β-carotene-linoleate system with an activity equivalent to about 0.31% (albedo) and 0.34% (flavedo) of the BHT activity, while the antiradical activity resulted equivalent to that of 62.90% (albedo) and 60.72% (flavedo) of the trolox activity. Finally, all extracts did not show any cytotoxic effect on differentiated Caco-2 cells by the AlamarBlue assay while exhibited, by MTT assay, a significant decrease in colon cancer Caco-2 cell viability.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In 2019 the total world’s production of Citrus crop exceeded the 140,000 thousand tonnes of which over 10,000 come from European region. The main product of Citrus transformation industry is over the past years, juice [1]. The processing of fruits for juice production generates a considerable amount of by-products, mainly represented by seeds, pulp residue, albedo and flavedo [2], known to be a good source of bioactive compounds, including polyphenols. In addition to the well documented [3] antioxidant activity, polyphenol compounds have shown protective effects against cancer and, in general cardiovascular diseases [4], anti-obesity effects [5], anti-inflammatory properties [6] and antimicrobial activity [7]. The phenolic compounds of Citrus fruits peel have been previously used as protective agent against oxidation in soybean oil [8] and in sunflower oil [9]. Frydman and coworker [10] used Citrus phenolic compounds as a substrate for the production of a sweetener and flavor enhancer, while Magda et al. [11] reported that the peel powder from Citrus reticulata and Citrus orange acts as an antioxidant in biscuits without affecting the sensory characteristic of food.
Recently some authors [12, 13] reviewed the phytochemical characteristic of Citrus peel highlighting the richness of this substrate of functional ingredients. Several reports in literature [14, 15] show that Citrus derived food products have beneficial characteristic linked to cancer, hypertension or in general to cardio-vascular diesases. Recently Caggia and coworkers [16] showed the potentiality of orange fiber extracted from the waste residue derived from the production of orange juice. The authors used the extracted fiber as ingredient of barkery product with reduced fat content.
Besides several Citrus-based food products are currently available on the market, some example are represented by citrus-peel derived tea or Citrus peel containing chocolate [13]. Among the several examples of utilization of discarded albedo and its components [12, 13] its direct transformation to edible products is maybe its main characteristic. In Sardinia (Italy) the albedo obtained by Citrus limon var. pompia Camarda (pompia) fruits is traditionally transformed, by caramelization with honey, in one of the most ancient confectionery products known as “sa pompia” [17].
Previous phytochemical reports on pompia explored the chemical composition of the volatiles of the flavedo while [18] studied the encapsulation of aqueous albedo extract in novel vesicle and gave preliminary results on the characterization of polar fraction. The volatile fraction extracted from the pompia flavedo is characterized by great amount of limonene [19,20,21] while the essential oil extracted from the leaves, although in less amount, still show limonene as major compound followed by considerable amount of the two isomers of citral [22, 23]. The leaves essential oil has shown good activity against several strains of Listeria monocytogenes and Staphylococcus aureus.
Considering the general interest in the valorisation of the industry by-products like citrus albedo and flavedo and the relatively lack of information related to the pompia, herein we report on the chemical characterization of the polar fraction from albedo and flavedo of pompia by liquid chromatography coupled with mass spectrometry. In addition, the crude extracts were also evaluated for their antioxidant activity (DPPH, cholesterol thermal-degradation, and β-carotene/linoleic acid assays) and toxic effect in human intestinal epithelial cells (Caco-2 cells).
Materials and methods
Chemicals
Solvents used for extraction, HPLC MS grade methanol, acetonitrile, formic acid, cholesterol, 5-cholesten-3β-ol-7-one (7-keto), 5-cholestene-3β,7β-diol (7β-OH), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), were purchased from Sigma-Aldrich Chemical Company (St Louis, MO). HPLC grade water (18 mΩ) was prepared by using a Millipore (Bedford, MA, USA) Milli-Q purification system. Standards reference compounds such as p-coumaric acid, rosmarinic acid, quinic acid, ferulic acid, apigenin, phloridzin, luteolin, sinapic acid, were purchased from Sigma Aldrich; standards of, isoquercetin, rutin, robinin, isorhamnetin, isorhamnetin rutinoside, eriocitrin, myricitrin, quercetin galattoside, myricetin galattoside, naringin, neohesperidin, neoeriocitrin, narirutin, hesperidin were purchased from Extrasynthese (Genay, France). Cell culture materials were purchased from Invitrogen (Milan, Italy). AlamarBlue was obtained from Biosource Europe (Nivelles, Belgium). All the chemicals used in this study were of analytical grade.
Plant material and extraction method
The fruits of pompia were collected in 2020 from Siniscola (NU) Sardinia, Italy (Plants were botanically identified by Dr. M. Chessa). The flavedo and albedo were separated from the fruit by peeling. The collected samples of albedo and flavedo were then extracted with ethanol:water (70:30 v/v; sample to solvent ratio 13:25 w/v) under ultrasound agitation for 1 h and then stored in the dark overnight. Samples were filtered and the solvent was completely removed using a rotary evaporator under vacuum at 30 °C.
Chemical characterization
ESI–MS and ESI–MS/MS analyses
Full scan ESI–MS, MS/MS analyses of standards were performed on an AB Sciex 4000 Q-Trap (Foster City, CA, USA) spectrometer. Standard solutions (1 μg/μL in methanol:water 50:50) were infused at 10 μL/min for tuning, the optimized parameters were: fragmentation reactions selected for each compound, dwell times, and Declustering Potential, Entrance Potential, Collision Energy, Collision Cell Exit Potential values. Data were acquired in the negative ion MS and MS/MS modes.
HPLC–ESI–MS/MS analysis
Qualitative on-line HPLC–ESI–MS/MS analysis of extracts was performed using an UHPLC system interfaced to an ABSciex (Foster City, CA, USA) API4000 Q-Trap instrument in ion trap mode. LC analyses were conducted using a system equipped with a Flexar UHPLC AS system (Perkin-Elmer, USA) consisting of degasser, Flexar FX-10 pump, autosampler and PE 200 column owen. Samples (5 μL) were injected into a XSelect CSH C18 column (Waters, Milford, MA) (100 × 2.1 mm i.d., 2.5 μm d). Mobile phase A was H2O containing 0.1% formic acid while mobile phase B was acetonitrile containing 0.1% formic acid. Elution was carried out at flow of 300 μL/min according to the following gradient: 0–8 min, from 0 to 6% B; 8–25 min, linear gradient 6%–10% B; 25–35 min, linear gradient 10%–40%. The column was kept at 47 °C. The flow from the chromatography was injected directly into the ESI source. Qualitative analysis of the compounds was performed using IDA (information dependent acquisition). The IDA method created included IDA criteria (specifying the charge state, mass range), enhanced MS scan, enhanced resolution, enhanced product ion scan or MS/MS scan. The source temperature was held at 450 °C, and MS parameters were those optimized for the ESI–MS and ESI–MS/MS analyses with ion spray voltage at -4500 V. MS data were acquired using the software provided by the manufacturer (Analyst software 1.6.2) and extracted ion fragmentograms (XIC) were analyzed in order to identify compounds from their deprotonated molecular ions and retention time.
Quantitative on-line HPLC–ESI–MS/MS analyses were performed using the same LC–ESI–MS/MS equipment and the same chromatographic conditions described above, but the mass spectrometer worked with a triple quadrupole analyzer in Multiple Reaction Monitoring (MRM) mode. The API 4000 ES source was tuned by infusing solutions of standards (1 μg/μL in methanol 50%) into the source at a flow rate of 10 μL/min. The voltage applied was -4500 V. Data acquisition and processing were performed using Analyst software 1.6.2.
Preparation of standard solutions
In order to prepare the calibration plot, a sample (1 mg) of each standard was weighed accurately into a 1 mL volumetric flask, dissolved in ethanol 70% (v/v) and the volume made up to the mark with ethanol. The resulting stock solution was diluted with methanol in order to obtain reference solutions containing 0.01, 0.05, 0.1, 1, 5, and 10 μg/mL of external standards.
The calibration curves, for each compound, were made by linear regression by plotting the peak area of external standard against their known concentrations. The result represents the average of curves performed by three injections of each concentration. All quantitative data were elaborated with the aid of Analyst software (AB Sciex).
Total amount of phenolic compounds and flavonoids
Total phenolic content of the pompia extracts was determined according to the Folin–Ciocalteu assay [24]. For this procedure, 100 μL of extract solution were mixed with 100 μL of 50% Folin–Ciocalteu phenol reagent and vortexed. After 3 min, 2 mL aqueous solution of 2% Na2CO3 were added into the test tubes and vortexed again. After 30 min of incubation, the absorbance of the samples was measured at 685 nm. Gallic acid was employed as a reference substance and results were expressed in mg of gallic acid/g of pompia extract dry weight. All measures were repeated three times and averaged.
The total flavonoids content was estimated by the AlCl3 assay [25]. Briefly, one mL of pompia extract solution was added to 1 mL of 2% methanolic AlCl3 6H 2O. The absorbance was measured 10 min later at 430 nm. Rutin was employed as a reference substance and results were expressed as mg rutin/g of pompia extract dry weight. All measures were repeated three times and the results were averaged.
Antiradical activity (DPPH assay)
The antiradical activity was determined by the 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical-scavenging method [26]. Each sample was mixed with 900 µL of 100 mM Tris–HCl buffer, pH 7.4 and the resulting solution was then added to 1 mL of 0.5 mM DPPH in methanol (250 µM in the reaction mixture). The control sample was prepared using methanol. Trolox was employed as a reference antioxidant substance. Absorbances of the mixtures were measured at 517 nm. The activity was calculated as IC50 and Trolox equivalent. All tests and analyses were run in triplicate and averaged.
β-Carotene/linoleic acid assay
A stock solution of β-carotene/linoleic acid mixture was prepared as follows: 0.5 mg β-carotene were dissolved in 1 mL of chloroform, then 25 µL linoleic acid and 200 mg Tween 40 were added. Then, 100 mL distilled water saturated with oxygen (30 min, 100 mL/min) were added with vigorous shaking; 2.5 mL of this reaction mixture were dispensed into test tubes and 10 µL portions of the pompia extracts were added; the emulsion system was incubated for up to 48 h at room temperature. The same procedure was repeated with the synthetic antioxidant butylated hydroxytoluene (BHT) as positive control. After this incubation period, absorbance of the mixtures was measured at 490 nm. The activity was calculated as BHT equivalents and as IC50 values. All tests and analyses were run in triplicate and averaged. [27]
Cholesterol assay
The cholesterol oxidation assay was conducted in dry state as previously described [28, 29]. Aliquots of 0.5 mL (2586 nmol) of cholesterol solution (2 mg/mL of MeOH) were dried in a round-bottom test tube under vacuum, and then incubated in a bath at 140 °C for 1 h (oxidized controls, 0) under artificial light exposure. Controls (non-oxidized cholesterol, Ctrl) were kept at 0 °C in the dark. In a different set of experiments, aliquots (0.5–50 µg) of albedo and flavedo extracts in solution (1 mg/mL and 100 µg/mL) were added to 0.5 mL of cholesterol solution (2 mg/mL MeOH). The mixtures cholesterol/extract were dried under vacuum, and then incubated in dry state in a bath at 140 °C for 1 h. The oxidation was stopped by immersing the test tubes in an ice bath for 10 min and adding 1 mL of MeOH. Quantification of cholesterol, 7-ketocholesterol (7-keto), and 7β-hydroxycholesterol (7β-OH) were carried out with an Agilent Technologies 1100 liquid chromatograph equipped with a diode array detector (HPLC–DAD). Cholesterol and oxysterols were analyzed using an Inertsil ODS-2 column, 150 × 4.6 mm, 5 µm particle size (Superchrom), and MeOH as the mobile phase, at a flow rate of 0.7 mL/min. Cholesterol and 7β-OH were detected at 203 nm and 7-keto at 245 nm [29]. Recording and integration of the chromatogram data was carried out through an Agilent OpenLAB Chromatography data system. The identification of cell lipid components was made using standard compounds and conventional UV spectra. Calibration curves each compound was constructed using reference standards and were found to be linear, with correlation coefficients > 0.995.
Cell cultures
The Caco-2 cell line was obtained from the European Collection of Cell Cultures (ECACC) (Salisbury, Wiltshire U.K.). Caco-2 cells have been obtained from a human colon adenocarcinoma (undifferentiated cancer Caco-2 cells); after confluence (approximately 9 to 21 days of growth), these cells spontaneously differentiate into enterocytes (differentiated Caco-2 cells) [30]. Subcultures of the Caco-2 cells were grown in T-75 culture flasks and passaged with a trypsin–EDTA solution. Cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal calf serum (FCS), 2 mM l-glutamine, and penicillin (100 units/mL)–streptomycin (100 µg/mL), at 37 °C in 5% CO2.
Cytotoxic activity of pompia extracts in differentiated Caco-2 cells: AlamarBlue assay
The cytotoxic effect of pompia extracts was evaluated in small intestine enterocytes (differentiated Caco-2 cells) by the AlamarBlue test [31, 32]. Caco-2 were seeded in 24-well plates at a density of 5 × 104 cells/mL, and culture medium was replaced 3 times a week for 21 days until complete differentiation of the cells. Prior to the addition of pompia extracts, cell culture medium was removed, differentiated Caco-2 cells were washed with phosphate-buffered saline (PBS) containing Ca2+ and Mg2+, and then fresh medium was added. Cells were subsequently exposed to various concentrations of albedo and flavedo extracts (25–500 µg/mL, in water solution) in complete culture medium and incubated for 24 h. The cell medium was removed and replaced with 1 mL of AlamarBlue solution (10% v/v in culture medium without FCS) in each well. Cells were incubated overnight at 37 °C, in order to allow viable cells to convert AlamarBlue oxidize blue form to the reduced pink form. After incubation, the absorbance was measured at 570 and 600 nm with an Infinite 200 auto microplate reader (Tecan, Austria). The percentage of AlamarBlue reduction was calculated and compared to the controls. The evaluation of the morphology of differentiated Caco-2 cells after 24 h of incubation with various amounts of pompia extracts was performed by microscopic analysis with a ZOE™ Fluorescent Cell Imager (Bio-Rad Laboratories, Inc., California, USA).
Cytotoxic activity of pompia extracts in cancer Caco-2 cells: MTT assay
The cytotoxic effect of pompia extracts was also preliminary evaluated in cancer (undifferentiated) Caco-2 cells by the MTT assay [32, 33]. Caco-2 cells were seeded in 96-well plates at a density of 5 × 104 cells/mL in 100 μL of medium and cultured overnight. Prior to the addition of extracts, the cell culture medium was removed, Caco-2 cells were washed with PBS containing Ca2+ and Mg2+, and then fresh medium was added. Cells were subsequently exposed to various amounts of albedo and flavedo extracts (25–500 µg/mL, in solution) in complete culture medium and incubated for 24 h. The cell culture medium was then removed from each well of the 96-well plates; an 8 μL portion of MTT solution (5 mg/mL of H2O) was added to cells in fresh medium and left for 4 h at 37 °C. The medium was aspirated, 100 μL of DMSO was added to the wells, and color development was measured at 570 nm with an Infinite 200 auto microplate reader (Infinite 200, Tecan, Austria). The absorbance is proportional to the number of viable cells.
Statistical analyses
Graph Pad INSTAT software (GraphPad software, San Diego, CA, USA) was used to calculate the means and standard deviations (SD) of two or three independent experiments involving duplicate or triplicate analyses for each sample/condition. Evaluation of statistical significance of differences was performed by one-way analysis of variance (One-way ANOVA), using Bonferroni Multiple Comparisons Test.
Results and discussion
IDA method with EMS survey scans, ER and EPI scans was developed and used with the aim to establish a qualitative composition of the phenolics occurring in albedo and flavedo of pompia. The MS2 mode is a useful tool to provide information on the nature of metabolites analyzed. In Table 1 are reported the chemical compounds detected in flavedo and albedo of pompia. The chemical composition of the two fractions was qualitative similar but with differences in the amounts of the different components. The identification of compounds was firstly performed on the base of the MS/MS fragmentation pattern and then the chemical structures were confirmed by injection of standard solutions.
Flavanones, characterized by the absence of any π-bond between the carbons 2- and 3- and by the presence of a chiral carbon at the 2-position, are the most abundant polyphenols in Citrus fruits Flavanones represent the main class of polyphenols also in Citrus peel (albedo and flavedo) where are mainly found in glycosylated form. [34]. The identified flavanones in our samples were eriocitrin, naringin, myricitrin, neohesperidin, neoeriocitrin, narirutin and hesperidin all of them quite common phenols of Citrus fruit [18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35]. Great attention has been given to Citrus flavanones, among the biological activities, hesperidin and eriocitrin were identified as potent antioxidant agents while neohesperidin and naringin showed hypoglycenimc effect related to their capacity to inhibit starch digestion [12].
Other flavonoids such as isoquercetin, luteolin, apigenin and isorhamnetin (aglicone form) and phlordzin, robinin, rutin, quercetin galactoside and isorhamnetin rutinoside (glyosidic form) were detected in pompia samples. Glycosylated flavones and flavanols are in general detected in small amount in Citrus peel [36, 37]. As reported by several authors [38] the presence of sugar molecule in the structure could affect the antioxidant potential of Citrus flavonoid. Citrus peel is reported to be also a good source of phenolic acids; among this class of compounds the hydroxycinnamic acids are overall present in higher concentration than the hydroxybenzoic acids [2, 39]. The chemical analysis of both pompia albedo and flavedo shows the presence of ferulic, p-coumaric and sinapic acids which are considered the most representative hydroxycinnamic acids for Citrus species [12, 37].
On the base of the fragmentation data of each standard, a multiple reaction monitoring (MRM) method was developed in order to obtain a quantitative evaluation. The UHPLC-ESI–MS/MS (MRM) method was then applied to the albedo and flavedo extracts and a simultaneous determination of 21 compounds was obtained (Table 1). The main flavanones of albedo and flavedo were naringin, neohesperidin, nereriocitrin and hesperidin and their concentration ranged from 0.24 µg/mL of hesperidin in flavedo extract to 16.7 µg/mL of neohesperidin in albedo extract confirming previous literature data [18]. Among organic acids, quinic acid was the main compound in both albedo and flavedo in line with previous results of Manconi et al. [18]. The LC-DAD and LC-ESI-(HR)MSn profiles previously conducted on the hydroethanolic extract of “pompia intrea”, the product obtained by the caramelization of pompia albedo with honey, showed high levels neohesperidin, neoeriocitrin and naringin [17]. In general, our results showed that flavedo is richest, with respect to the albedo, in flavonoid compounds, as confirmed also by spectrophotometric analysis (Table 2), by contrast the flavanones were found in higher concentration in albedo extracts (Table1). The same trend was also found in the total phenolic compounds determined by spectrophotometric Folin-Ciocalteu method (Table 2), with values of 2.42 ± 0.26 and 2.89 ± 0.27 mg of gallic acid/g of extract measured in albedo and flavedo extracts, respectively.
Pompia extracts were assayed for their antioxidant activity during cholesterol oxidation and linoleic acid/β-carotene oxidation as well as for antiradical activity in terms of ‘radical scavenging ability’, towards the stable radical 2,2-diphenyl-1-picrylhydrazyl (DPPH).
The oxidative degradation of cholesterol, essential component of biological membranes and lipoproteins, is well known to play a role in the development of tissue damage and in a wide range of other pathological events [40]. The consumption of cholesterol in dry state at 1 h and the formation of its mayor oxidation products, the 7-keto and 7β-OH derivatives, were measured as markers of oxidative process. Figure 1 shows the antioxidant activity measured during cholesterol oxidation in the presence of different amounts (0.5–50 µg) of pompia extracts (flavedo and albedo). Antioxidant activity is reported as percentage of cholesterol protection, calculated considering the percent of sterol consumption in the presence of the antioxidant with respect to total cholesterol consumption without antioxidant (100% of consumption or 0% of protection). Flavedo extract exerted a significant protection (45%) of cholesterol from degradation at 10 µg, showing a complete inhibition of the oxidative process from 25 µg. In this system, albedo extract, although less effective, showed a protection > 50% at 50 µg. Figure 2 shows the values (expressed as μg) of the oxysterols 7β-OH and 7-keto measured during cholesterol oxidation in the controls (Ctrl) and in oxidized samples in the absence (0) or in the presence of different amounts (0.5–50 µg) of pompia extracts. Flavedo extract could significantly prevent the formation of both oxysterols from 10 µg, while albedo extract significantly reduced 7-keto formation from 25 µg. This model of lipid oxidation has been amply used to assess the scavenging ability against peroxyl radicals LOO· of natural extracts and pure phenolic compounds [29, 41]. The antioxidant potency of an extract in this system strictly depends on the amount of total phenols and the type of phenolic compounds [21, 29, 41]. Flavedo extract showed a greater ability than albedo extract to protect sterol against free radical attack and to inhibit oxysterol formation, maybe correlated to its richness in total phenols and flavonoid compounds. The high LOO· scavenging ability of pompia flavedo extract was comparable to the previously observed scavenging ability of Myrtus communis L. berries ethanol extract which showed a complete inhibition of the cholesterol oxidative process at 25 and 50 µg at the 1 h time point [42]. Our previous study evidenced the inefficacy of the essential oil obtained from pompia flavedo against cholesterol degradation, correlated to its low content of phenols [21].
Antioxidant activity (% protection) of different amounts (0.5–50 μg) of flavedo and albedo extracts measured during cholesterol oxidation at 140 °C for 1 h. Three independent experiments are performed and data are presented as mean ± SD (n = 6); a = p < 0.001; b = p < 0.01 versus oxidised control (0% protection)
Values of oxysterols 7β-OH and 7-keto (expressed as μg) measured in the controls (Ctrl) and in oxidized samples in the absence (0) or in the presence of different amounts (0.5–50 μg) of flavedo (A) and albedo (B) extracts, during the oxidation of cholesterol at 140 °C for 1 h. Three independent experiments are performed and data are presented as mean ± SD (n = 6); a = p < 0.001; c = p < 0.05 versus oxidized sample (0). Values of 7β-OH and 7-keto in the controls were 0.01 ± 0.01 μg and 0.08 ± 0.04 μg, respectively
The inhibition of lipid oxidation in the β-carotene-linoleate system showed an IC50 = 7.92 ± 1.7 (albedo extract) and 7.16 ± 1.6 mg/mL (flavedo extract). This activity is equivalent to about 0.31 and 0.34% of the BHT activity. The pompia extracts showed a mean ability for scavenging free radical species (DPPH test systems) with an IC50 = 0.44 ± 0.02 mg/mL and 0.46 ± 0.03 mg/mL for albedo and flavedo respectively. This activity is equivalent to about 62.90 and 60.72% of the trolox activity (Table 2). Both pompia extracts showed a certain degree of antioxidant activity in the β-carotene-bleaching test and a radical scavenging activity in the DPPH assay, as previously observed for the essential oils obtained from pompia flavedo and leaves [21, 22]. Our results are in line with those previously observed in the DPPH assay applied to the hydroalcolic extract obtained from the peel of C. limon, that showed an IC50 value of about 434.50 ± 5.9 µg/mL [43]. Edible portions of Citrus fruits but also its by-products have been reported to possess marked antioxidant activity [12]. In particular, the antioxidant properties of Citrus peel extracts has been correlared to the presence of phenolic compounds like hesperidin, hesperetin, eriocitrin, naringenin, nobiletin, and tangeretin, luteolin, and quercetin [12]. Our data substantiated the antiradical properties and lipid peroxidation inhibition properties of both pompia flavedo and albedo extracts, maybe correlated to the high amount of antioxidant flavones and hydroxycinnamic acids.
Pompia extracts was then tested for cytotoxicity in differentiated Caco-2 cell monolayers as an intestinal epithelial cell model [32], in order to estimate their potential applicability in cosmeceutical, nutraceutical, and pharmaceutical products. The differentiated Caco-2 cells, retaining many of the morphological and functional characteristics (such as brush border microvilli, tight junctions, and dome formation) of normal small intestine polarized enterocytes, are extensively used in the prediction of intestinal drug absorption and in studies of toxicity of natural extracts [30, 32]. Figure 3A shows the viability (expressed as % control) measured in differentiated Caco-2 cells after 24 h-incubation in the presence of different concentrations (10–500 μg/mL) of pompia extracts by the AlamarBlue assay. Both the extracts did not show a toxic effect on intestinal epithelial cells in the tested concentration range, with treated cells showing viability values similar to control cells. Similar results were were previously observed for the hydroethanolic extracts obtained from “pompia intrea”, that did not show toxic effect by MTT assay in differentiated Caco-2 cell cultures at the concentration range 5–50 μg/mL [17].
Viability, expressed as % of the control cells (Ctrl), induced by incubation for 24 h with different concentrations (10–500 μg/mL) of albedo and flavedo extracts in differentiated Caco-2 cells by AlamarBlue assay (A). The panel shows representative images of phase contrast of control differentiated Caco-2 cells and cells treated for 24 h with the two pompia extracts at 500 µg/ml; bar = 100 µm (B). Three independent experiments are performed and data are presented as mean ± SD; no significant differences were observed versus Ctrl
A microscopic observation was also performed in order to evidence the effect of pompia extracts on the morphology of differentiated Caco-2 cells. The panel of Fig. 3B shows representative images of phase contrast of control differentiated Caco-2 cells and cells treated for 24 h with the highest tested dose (500 µg/mL) of the two pompia extracts. Differentiated Caco-2 cells were small and closely linked to each other. The cell treatment with pompia extracts did not trigger noticeable changes in the morphology of treated cells and in the distance between cells. We presented evidence that both pompia extracts did not affect cell morphology and viability in differentiated Caco-2 cell monolayers, a cell model that mimics the normal intestinal absorptive epithelium [30, 32]. The absence of toxicity in this cell model makes these extracts suitable for potential pharmaceutical, nutraceutical, and cosmeceutical applications.
Pompia extracts were also preliminarly tested and compared for cytotoxicity in colon cancer Caco-2 cell cultures (undifferentiated Caco-2 cells) by MTT assay. Cytotoxic MTT assay is normally used to assess the cell metabolic activity linked to mitochondrial functional state [44]. Figure 1S shows the viability, expressed as % of the control, induced in cancer (undifferentiated) Caco-2 cells after 24 h incubation in the presence of different concentrations of flavedo and albedo extracts (10–500 μg/mL). The treatment with both the extracts induced a significant reduction in cell viability (40%), in comparison with control, at the concentration of 500 μg/mL. A 22% significant decrease in Caco-2 cell viability was observed for flavedo extract at 250 μg/mL. Several investigations have shown the growth inhibitory effect and antiproliferative properties of Citrus extracts [12, 45] and the essential oil obtained from the pompia flavedo [21] in cancer cells. However, further studies are needed to evaluate the cellular effects of pompia flavedo and albedo extracts on cancer cells.
Conclusion
Liquid chromatography coupled with mass spectrometry analysis was successfully applied to the extracts of albedo and flavedo pompia fruits. Qualitative UHPLC-MS/MS analyses show the presence, among flavanone compounds, of eriocitrin, naringin, myricitrin, neohesperidin, neoeriocitrin, narirutin and hesperidin. Quantitative determinations show that the flavedo was the richest in flavonoid compounds while the flavanones were found in higher concentration in albedo extracts. In addition to several hydroxycinnamic acids the chemical analyses also show the presence in both albedo and flavedo of quinic acid, which was the most representative detected organic acid.
The overall data suggest that pompia extracts exhibited antioxidant protection against cholesterol degradation and a DPPH radical scavenging activity. Flavedo extract showed the highest antioxidant ability in protecting sterol against free radical attack and inhibiting oxysterol formation. Moreover, the extracts did not show any toxicity on intestinal epithelial cells in the tested concentration range, without affecting cell viability and morphology.
Our data qualify hydroalcoholic extracts obtained from pompia flavedo and albedo as a good source of biologically active components with considerable antioxidant activity, useful for pharmaceutical, nutraceutical, and cosmeceutical applications.
References
FAO 2020, Available at: http://www.fao.org/economic/est/est-commodities/citrus-fruit/en/
F.A. Marin, C. Soler-Rivas, O. Benavente-Garcio, J. Castillo, J.A. Perez-Alvarez, Food Chem. 100, 736–741 (2007). https://doi.org/10.1016/j.foodchem.2005.04.040
F. Shahidi, P. Ambigaipalan, J. Funct. Foods 18, 820–897 (2015). https://doi.org/10.1016/j.jff.2015.06.018
A.M. Boudet, Phytochem. 68, 2722–2735 (2007). https://doi.org/10.1016/j.phytochem.2007.06.012
Y.W. Huang, Y. Liu, S. Dushenkov, C.T. Ho, M.T. Huang, J. Funct. Foods 1, 304–310 (2009). https://doi.org/10.1016/j.jff.2009.06.002
E. Middleton, C. Kandaswami, T.C. Theoharides, Pharmacol. Rev. 52, 673–751 (2000)
R. Puupponen-Pimiä, L. Nohynek, C. Meier, M. Kahkonen, M. Heinonen, A. Hopia, K.M. Oksman-Caldentey, J. App. Microbiol. 90, 494–507 (2001). https://doi.org/10.1046/j.1365-2672.2001.01271.x
H.A. Abd El-aal, F.T. Halaweish, Lucrări Ştiinţifice 53, 457–464 (2009)
V.T. Tumbas, G.S. Ćetkovic, S.M. Djilas, J.M. Canadanovic-Brunet, J.J. Vulic, Z. Knez, Acta Periodica Technologica 40, 195–203 (2010). https://doi.org/10.2298/APT1041195T
A. Frydman, O. Weisshaus, D.V. Huhman, L.W. Sumner, M. Bar-Peled, E. Lewinsohn, R. Fluhr, J. Gressel, Y. Eyal, J. Agric. Food Chem. 53, 9708–9712 (2005). https://doi.org/10.1021/jf051509m
R.A. Magda, A.M. Awad, K.A. Selim, Alex. J. Fd. Sci. Technol. 45, 75–82 (2008)
M. Leporini, R. Tundis, V. Sicari, M.R. Loizzo, Ital. J. Food Sci. 33, 63–107 (2021). https://doi.org/10.15586/ijfs.v33i2.200913
N.E. Wedamulla, M. Fan, Y.-J. Choi, E.-K. Kim, J. Func. Food. 95, 105163 (2022). https://doi.org/10.1016/j.jff.2022.105163
A.K. Gupta, P. Mishra, M. Senapati, P.P. Sahu, J. Food Eng 306, 110637 (2021). https://doi.org/10.1016/j.jfoodeng.2021.110637
H.R. Park, K.S. Shin, Food Biosci. 43, 101301 (2021). https://doi.org/10.1016/j.fbio.2021.101301
C. Cagga, R. Palmeri, N. Russo, R. Timpone, C.L. Randazzo, A. Todaro, S. Barbagallo, Front. Nutr. 7, 46 (2020). https://doi.org/10.3389/fnut.2020.00046
M. Deiana, P. Montoro, I. Jerkovic, A. Atzeri, Z. Marijanavoic, G. Serreli, S. Piacente, C.I.G. Tuberoso, Food Res. Int. 120, 620–230 (2019). https://doi.org/10.1016/j.foodres.2018.11.016
M. Manconi, M.L. Manca, F. Marongiu, C. Caddeo, I. Castangia, G.L. Petretto, G. Pintore, G. Sarais, D’hallewin, G., Zaru, M., Bacchetta, G., Fadda, A.M., Int. J. Pharm. 506, 449–457 (2016). https://doi.org/10.1016/j.ijpharm.2016.04.014
G. Fenu, A. Carai, M. Foddai, E. Aazara, S. Careddu, M. Usai, Int. J Essent. Oil Therapy 4, 23–25 (2010)
G.L. Petretto, G. Sarais, M.T. Maldini, M. Foddai, B. Tirillni, J.P. Rourke, M. Chessa, G. Pintore, J. Food Process. Pres. 40, 950–957 (2016). https://doi.org/10.1111/jfpp.12674
A. Rosa, M. Nieddu, G.L. Petretto, G. Sarais, J. Ess. Oil Res. 31, 390–399 (2019). https://doi.org/10.1080/10412905.2019.1606740
F. Fancello, G.L. Petretto, S. Zara, M.L. Sanna, R. Addis, M. Maldini, M. Foddai, J.P. Rourke, M. Chessa, G. Pintore, LWT-Food Sci Tech. 69, 579–585 (2016). https://doi.org/10.1016/j.lwt.2016.02.018
F. Fancello, G.L. Petretto, S. Marceddu, T. Venditti, G. Pintore, G. Zara, I. Mannazzu, M. Budroni, S. Zara, Food Microbiol. 87, 103386 (2020). https://doi.org/10.1016/j.fm.2019.103386
V.L. Singleton, J.A.J. Rossi, Am. J. Enol. Vit. 16, 144–153 (1965)
J.L. Lamaison, A. Carnat, Pharm. Acta Helv. 65, 315–320 (1990)
D.M. Peterson, M.J. Hahn, C.L. Emmons, Food Chem. 79, 473–478 (2002). https://doi.org/10.1016/S0308-8146(02)00219-4
A. Dapkevicius, R. Venskutonis, T.A. Van Beek, P.H. Linssen, J. Sci. Food Agri. 77, 140–146 (1998). https://doi.org/10.1002/(SICI)1097-0010(199805)77:1%3C140::AID-JSFA18%3E3.0.CO;2-K
S.K. Kim, W.W. Nawar, Lipids 28, 917–922 (1993). https://doi.org/10.1007/BF02537501
A. Rosa, A. Atzeri, M. Deiana, P. Scano, A. Incani, C. Piras, F. Cesare Marincola, Food Res. Int. 69, 322–330 (2015). https://doi.org/10.1016/j.foodres.2015.01.001
S. Travelin, J. Gråsjö, J. Taipalensuu, G. Ocklind, P. Artursson, Application of epithelial cell culture in studies of drug transport, in Epithelial cell culture protocols. ed. by C. Wise (Humana Press, Totowa, 2002), pp.233–272
J. O’Brien, I. Wilson, T. Orton, F. Pognan, Eur. J. Biochem. 267, 5421–5426 (2000). https://doi.org/10.1046/j.1432-1327.2000.01606.x
A. Rosa, A. Rescigno, A. Piras, A. Atzeri, P. Scano, S. Porcedda, P. Zucca, A. Dessì, Food Chem. Toxicol. 50, 3799–3807 (2012). https://doi.org/10.1016/j.fct.2012.07.003
C.J. Schiller, A. Klainz, K. Mynett, A. Gescher, Toxicol. In Vitro 6, 575–578 (1992). https://doi.org/10.1016/0887-2333(92)90070-8
Y. Nogata, K. Sakamoto, H. Shiratsuchi, T. Ishii, M. Yano, H. Ohto, Biosci Biotechnol Biochem. 70, 178–192 (2006). https://doi.org/10.1271/bbb.70.178
M.K. Khan, D.O. Zill-E-Huma, J. Food Comp. Anal. 33, 85–104 (2014). https://doi.org/10.1016/j.jfca.2013.11.004
D. Danzi, G. Ladu, C.V. Prieto, A.G. Bullon, G.L. Petretto, F. Fancello, T. Venditti, J. Sci. Food Agri. (2020). https://doi.org/10.1002/jsfa.9025
J.W. Erdman, D. Balentine, L. Arab, G. Beecher, J.T. Dwyer, J. Folts, J. Burrowes, J. Nutr. 137, 718S-737S (2007). https://doi.org/10.1093/jn/137.3.718S
S. Rafiq, R. Kaul, S.A. Sofi, N. Bashir, F. Nazir, G. Ahmad Nayik, J. Saudi Soc. Agric. Sci. 17, 351–358 (2018). https://doi.org/10.1016/j.jssas.2016.07.006
N. Balasundram, K. Sundram, S. Samman, Food Chem. 99, 191–220 (2006). https://doi.org/10.1016/j.foodchem.2005.07.042
C. Garenc, P. Julien, E. Levy, Free Radic Res 44, 47–73 (2010). https://doi.org/10.3109/10715760903321804
A. Rosa, C.I.G. Tuberoso, A. Atzeri, E. Bifulco, M.P. Melis, M.A. Dessì, Food Chem. 129, 1045–1053 (2011). https://doi.org/10.1016/j.foodchem.2011.05.072
C.I.G. Tuberoso, A. Rosa, E. Bifulco, M.P. Melis, A. Atzeri, F.M. Pirisi, M.A. Dessì, Food Chem. 123(4), 1242–1251 (2010). https://doi.org/10.1016/j.foodchem.2010.05.094
M. Makni, R. Jemai, W. Kriaa, Y. Chtourou, H. Fetoui, Biomed. Res. Int. 2018, 6251546 (2018). https://doi.org/10.1155/2018/6251546
M. Ghasemi, T. Turnbull, S. Sebastian, I. Kempson, Int. J. Mol. Sci. 22, 12827 (2021). https://doi.org/10.3390/ijms222312827
E. Palazzolo, V.A. Laudicina, M.A. Germanà, Curr. Org. Chem. 17, 3042–3049 (2013). https://doi.org/10.2174/13852728113179990122
Funding
Open access funding provided by Università degli Studi di Sassari within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
GLP: Conceptualization, Methodology, formal analysis, writing original draft preparation. GS: Conceptualization, review-editing. MM, formal analysis, review-editing. BT, formal analysis. MC: review-editing. GP: review-editing and AR: Methodology, formal analysis, writing original draft preparation.
Corresponding author
Ethics declarations
Conflict of interest
The authors confirm that they have no conflicts of interest with respect to the work described in this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rosa, A., Petretto, G.L., Maldini, M. et al. Chemical characterization, antioxidant and cytotoxic activity of hydroalcoholic extract from the albedo and flavedo of Citrus limon var. pompia Camarda. Food Measure 17, 627–635 (2023). https://doi.org/10.1007/s11694-022-01659-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-022-01659-w