Abstract
This randomized controlled study examined neurological changes in socioemotional processing skills through parent training in caregivers of children with attention-deficit/hyperactivity disorder. Thirty mothers of children with attention-deficit/hyperactivity disorder were stratified into parent training and non-parent training groups. Functional magnetic resonance imaging was performed during the “Reading the Mind in the Eyes” test, and parenting difficulties were evaluated using the Parenting Stress Index and the Parenting Scale, twice (before and after parent training). Only mothers in the parent training group showed a significant decrease in Parenting Stress Index and Parenting Scale scores. They also demonstrated increased activity in the left occipital fusiform gyrus during the task of estimating emotions from facial pictures. We presumed that these changes might reflect the potential impact of enrollment in parent training in reducing stress, which might have increased activation of the fusiform gyrus.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Developmental disabilities in children and parenting difficulties are strongly related (Woodman et al., 2015). Attention-deficit/hyperactivity disorder (ADHD), one of the most commonly diagnosed neurodevelopmental disorders in childhood, has a relatively high incidence (approximately 5%) (American Psychiatric Association, 2013). Children with ADHD have functional problems in social and academic areas (Marshall et al., 2014). Additionally, ADHD symptoms in children can negatively affect their caregivers' psychological state, parenting behavior, and parent–child relationships (Hutchison et al., 2016; Pelham et al., 1998). These negative influences could result in inappropriate parental responses, such as maltreatment, creating risk factors for development of later conduct problems in the affected children (Chronis et al., 2007; Chronis-Tuscano et al., 2008).
Parent training (PT) is a psychosocial, support intervention that promotes a nurturing environment for childcare (parental understanding and response), which is widely known for its effectiveness (Daley et al., 2014) and is recommended for parents of children with ADHD (Posner et al., 2020). The PT program for ADHD includes essential information for parents on the characteristics of the disability and treatment options (Iwasaka, 2012; Zwi et al., 2011). Studies of PT in mothers rearing ADHD children have reported that PT significantly decreased their stress and improved their parenting self-efficacy and style (reduced overly harsh responses, lack of consistency, and ineffective limit-setting) (Abikoff et al., 2015; Heath et al., 2015; Shimabukuro et al., 2017). This reduced and prevented behavioral problems in children with ADHD (Smith et al., 2008; Tully et al., 2004). However, these previous studies focused on questionnaire data, which are prone to subjective bias. Few studies have examined the effectiveness of PT using more objective measurements (e.g., neuroimaging data).
A previous neuroimaging study reported that tasks that measure social skills and abilities, such as mind-reading tasks (“Reading the Mind in the Eyes test [RMET] (Baron-Cohen et al., 2001)), are negatively influenced by stress (Nolte et al., 2013) and may, therefore, be useful biomarkers of caregivers’ stress (Shimada et al., 2018). In the RMET, participants evaluate the affective states of others from static eye region images. Previous functional magnetic resonance (fMRI) studies have reported supplemental motor area, and inferior frontal, inferior temporal, fusiform, and occipital gyrus involvement during the RMET (Adams et al., 2010; Baron-Cohen et al., 2006; Castelli et al., 2010; Focquaert et al., 2010; Shimada et al., 2018). However, it is unclear whether improving caregivers’ stress by PT also alters activation of these areas.
Herein, we focused on the effects of PT on parenting stress and parenting practices in mothers of children with ADHD, at a neurological level. We hypothesized that PT would improve qualitative stress and unsound parenting practice indexes, as determined using parenting stress and discipline questionnaires. Moreover, we hypothesized that functional changes in maternal brain could be revealed by blood oxygenation level-dependent (BOLD) fMRI during the RMET. We expected that mothers enrolled in PT may respond more efficiently to others’ emotional status, and that this might be related to activity changes in brain regions involved in judging other’s affective states. Understanding the effects of PT at a functional neural level could potentially aid the development of more precise treatment strategies and biomarkers for evaluating treatment effects.
Methods
Participants
Thirty mothers (mean age 38.72 years; standard deviation [SD] 4.46 years) agreed to participate in this study. The inclusion criteria were as follows: mothers caring for ≥ 1 school-aged child (age 6–12 years) diagnosed with ADHD in a hospital or pediatric clinic. ADHD was diagnosed using the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (American Psychiatric Association, 2013). The exclusion criteria were as follows: mothers who had participated in other PT programs within 2 months prior to enrolment, and changes in the child’s medication status during the study.
All participants had normal or corrected-to-normal vision, with no known medical, neurological, or psychiatric history (based on self-report questionnaires) and met the safety requirements for participation in an MRI study. Participants’ handedness was assessed using the Flinders Handedness Questionnaire (Nicholls et al., 2013; Okubo et al., 2014). The sex, medication status, and intelligence quotient (measured using the Wechsler Intelligence Scale for Children–Fourth Edition) of participants’ children were recorded.
The Ethics Committee of the University of Fukui approved the study protocol; all procedures were conducted in accordance with the Declaration of Helsinki. All participants provided written informed consent.
Study design
Using a permuted-block randomization procedure for computer-based random number generation, mothers were randomly allocated to two groups: those who attended a 13-week PT course (PT group), and those who did not attend the course until after the study ended (non-PT group). In the PT group, mothers were taught and trained on the following contents in four major elements, by two trained clinical psychologists, throughout the 13-week course: 1) psychological/medical knowledge of ADHD and stress management skills, 2) how to observe and appropriately respond to the child’s behaviors and modify parenting skills accordingly, 3) how to provide clear explanation and rules for their children regarding their ADHD symptoms, and 4) effective methods for responding appropriately to the children’s non-adaptive behaviors stemming from their ADHD symptoms (see Supplementary Material 1, Parent training).
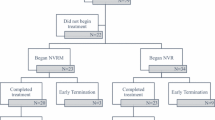
Before and after PT (approximately 90 days apart), mothers in both groups participated in the experiments at baseline (Time 1) and post-baseline (Time 2). Participants’ psychological characteristics were measured using psychological questionnaires, and their brain activities during emotion/gender judgement tasks were examined using fMRI. Of the 30 mother–child dyads (17 dyads in the PT group and 13 dyads in the non-PT group), seven (4 dyads in the PT group, and 3 dyads in the non-PT group) did not participate in the post-baseline session (4 dropped out and 3 were excluded from the analysis). Thus, the efficacy analysis included 13 dyads who received PT and 10 dyads who did not (for detail, see Fig. 1).
Questionnaires for psychological testing
The Interpersonal Reactivity Index (IRI) (Davis, 1983; Sakurai, 1988), which comprises four subscales (empathic concern, personal distress, perspective taking, and fantasy), was used to measure the mothers’ empathic abilities. The State–Trait Anxiety Inventory (STAI) (Hidano et al., 2000; Spielberger et al., 1983) was used to measure their state and trait anxiety. The Beck Depression Inventory-II (BDI-II) (Beck et al., 1996; Kojima et al., 2002) was used to measure the mothers’ current depression. To measure maternal parenting stress, we used the Parent Stress Index (PSI) (Abidin, 1995; Narama et al., 1999), which includes child and parent stressor domains. Higher scores indicated that the rater scored the child’s behavior more negatively or that the rater assessed themselves or the parenting surrounding environment negatively. The Parenting Scale (PS) (Arnold et al., 1993; Itani, 2010) was used to examine the mothers’ parenting approaches in terms of laxness and overreactivity. This refers to the tendency to respond permissively (by passive and inconsistent parenting practices) or harshly (by overly authoritarian parenting practices), respectively, to child misbehavior. During the study, the BDI-II, PSI, and PS scores were measured twice (Time 1 and Time 2) to assess PT effects.
Task and procedures
In the MRI study, mothers completed the revised RMET version (Baron-Cohen et al., 2001; Shimada et al., 2018), which consisted of two conditions. In one condition, the participant was presented 36 black and white photographs showing the eye region of a human face, with two words placed below the photo, one of which correctly identified the person’s emotion (the Theory of Mind [“ToM”] condition). In the other condition, the same 36 photographs were presented with the words “male” or “female” placed beneath, one of which correctly identified the person’s gender (the “GeN” condition). Participants were asked to indicate the word that they felt best described the person in the photograph by pressing one of two buttons in the scanner (Current Design, Philadelphia, PA). The task was run using Presentation software (Neurobehavioral Systems, Albany, CA), and projected using an MRI-compatible LCD display (BOLDscreen, Cambridge Research System, Cambridge, UK) placed on the back of the scanner. The participants viewed the stimuli via a mirror attached to the head coil.
The scanning was divided into two runs, each of which comprised a blocked CACBCA…design, where A: ToM condition, B: GeN condition, and C: resting condition. Both A and B lasted for 30 s, with the instruction word presented for 2.5 s. Then, six photographs were presented for 4.5 s each, followed by a 5-s inter-stimulus interval. C lasted for 15 s, during which only a fixation cross was presented. Each run contained 18 photographs of both A and B. The order of photographs in blocks A and B were counterbalanced across participants. Participants underwent training sessions before fMRI, outside the scanner, using training items (Supplementary Material 2 Figure S1).
fMRI acquisition and analyses
A 3-T MRI scanner (Signa PET/MR, ver. 26, GE Healthcare, Milwaukee, WI) with a standard 8-channel head coil was used. Functional images were corrected with a T2*-weighted gradient-echo echo-planar imaging sequence to produce 40 whole-brain, continuous, transaxial slices (slice thickness, 3.5 mm with a 0.5-mm gap, repetition time [TR], 2500 ms; echo time [TE], 25 ms; flip angle [FA], 80°; field-of-view [FOV], 192 mm; 64 × 64 matrix; voxel dimension, 3.0 × 3.0 mm). A T1-weighted anatomical scan was also acquired using a fast spoiled-gradient recalled imaging sequence (TR, 8.46 ms; TE, 3.24 ms; FA, 11°; FOV, 256 mm; 256 × 256 matrix; 172 slices; voxel dimension, 1.0 × 1.0 × 1.0 mm).
In image data processing, for MRI signal equilibrium, the first four volumes were discarded from the analysis. The remaining 240 images were pre-processed and analyzed using the Statistical Parametric Mapping software (SPM ver.12, Wellcome Trust Center for Neuroimaging, London, UK) implemented in MATLAB R2019b (MathWorks, Natick, USA). Following slice-timing correction and spatial realignment, T1-weighted anatomical images were co-registered to functional images and segmented using a new segmentation algorithm with the DARTEL technique (Ashburner, 2007). Functional images were then spatially normalized into the Montreal Neurological Institute template, re-sampled to a spatial resolution of 3 × 3 × 3 mm3, and spatially smoothed with a 6-mm full-width at half-maximum Gaussian kernel.
After pre-processing, we first performed individual analyses to identify task-related activity. In this analysis, a design matrix containing two regressors of interest (ToM and GeN conditions) was created. Each condition was modelled with a general linear model, by using a 30-s box-car function convolved with the canonical hemodynamic response function, high-pass filtered at 1/128 Hz to minimize low-frequency artefacts. Six motion parameters were also included as nuisance regressors. Then, two contrast images (ToM and GeN) were obtained for each participant.
For group analyses, the two contrast images corresponding to the ToM and GeN conditions generated in the individual analysis were chosen. To investigate the effect of PT, between- and within-group differences (PT/non-PT vs. [Time 1/Time 2]) were analyzed using repeated-measures analysis of variance (ANOVA) within the framework of the SPM flexible factorial design.
Due to the small sample size, the following analyses were conducted, based on previous studies (Baumeister et al., 2018, 2019). To depict regions showing the PT effect in the PT group, as compared to the non-PT group (regions showing a significant decrease and/or increase after the PT intervention period), the contrast of interest was set as follows: PT (Time 1 > Time 2) > non-PT (Time 1 > Time 2) and PT (Time 2 > Time 1) > non-PT (Time 2 > Time 1). These comparisons were performed separately for both ToM and GeN conditions. In addition to whole brain analyses, a region-of-interest (ROI) analysis was conducted using WFU PickAtlas (https://www.fmri.wfubmc.edu/downloads) with the Automated Anatomical Labelling Atlas. Based on the hypothesis, to create the ROI mask, the following regions were selected bilaterally and combined into one mask image: supplemental motor area, inferior frontal, inferior temporal, fusiform, and occipital gyrus. The number of activated voxels were computed in the ROI mask for each condition.
Subsequently, to identify the expected intervention effect in the PT group, we conducted a within-subject SPM one-way ANOVA for separate comparison of the activities within the PT and non-PT groups under both conditions. The statistical threshold was set at p < 0.005, uncorrected at the peak level, and at p < 0.05 at the cluster level, family-wise error-corrected for multiple comparisons over the whole brain. The Neuromorphometrics Atlas (http://www.neuromorphometrics.com/) was used to identify the anatomical localization of significant clusters.
Questionnaires and behavioral task data analyses
Fisher’s exact tests and individual t-tests were performed to compare the clinical and demographic characteristics between groups. Questionnaire scores and behavioral performance during the RMET were compared using mixed-model repeated-measures ANOVAs with time as the within-subject factor (pre- and post-PT period: Time 1 & Time 2), and groups as the between-group factor (PT and non-PT). For the RMET performance, the intervention effects were assessed on reaction times (RTs) and correct answer rate (accuracy) separately for the ToM and GeN conditions. To investigate significant effects further, Bonferroni-corrected pair-wise comparisons were performed. Statistical analyses were performed using IBM SPSS version 24 (IBM Corp., Armonk, NY, USA).
Results
Demographics and assessment outcomes at Time 1
The groups were similar in terms of age; BDI-II, STAI, PSI, PS, and IRI scores; years of education; handedness; and children’s data at Time 1 (Table 1).
Changes in questionnaire scores and RMET performance between Time 1 and Time 2
For the BDI-II scores, 2 × 2 ANOVAs (Time [Time 1, Time 2] × Group [PT, non-PT]) showed no significant main effects or interactions. For the PSI child-domain scores, there was a significant Time × Group interaction, while the post-hoc within-group t-test yielded significantly lower scores at Time 2 than at Time 1 (p = 0.003, 95% confidence interval [CI]: -14.38, -3.47) in the PT, but not in the non-PT group (p = 0.23, 95%CI: -2.52, 9.92). For PSI parent-domain scores, we also found a significant Time × Group interaction, with a significant decrease in the PT group (p = 0.042, 95%CI: -12.68, -0.24), and a trend towards a significant increase in the non-PT group (p = 0.053, 95%CI: -0.09, 14.09). For PS overreactivity scores, there was a significant Time × Group interaction, with a significant decrease in the PT (p = 0.009, 95%CI: -11.415, -1.816), but not in the non-PT group (p = 0.75, 95%CI: -4.57, -6.37). No significant main effects or interactions were found for laxness scores.
Under the ToM condition in the RMET, the RTs showed a trend toward significance for the main effect of Time (p = 0.058) and interaction of Time × Group (p = 0.050), but not for the main effect of Group (p = 0.538). A planned post-hoc t-test showed a significant RT decrease in the PT (p = 0.005, 95%CI: -313.93, -61.64), but not in the non-PT group (p = 0.96, 95%CI: -140.34, 147.31) (Fig. 2). For accuracy, there was a trend toward significance for the main effect of Time (p = 0.063), but not for the main effect of Group (p = 0.801) or interaction of Time × Group (p = 0.682). Other indices (RTs and accuracy under GeN condition) showed no significance or trend toward significance for the main effects or interactions (Table 2).
Box plot showing the reaction times under the ToM condition for both groups and at both time points. Reaction times at Time 1 and Time 2 for (a) PT group, and (b) non-PT group. Abbreviations: ToM, Theory of Mind; PT, mothers enrolled in parent training intervention; non-PT, mothers who did not enroll in parent training intervention until after the end of the study.
Furthermore, additional ANOVA for correct item RTs under the ToM condition showed a similar trend as above; the RTs showed a significant main effect of Time (p = 0.008) and a trend toward a significant interaction of Time × Group (p = 0.052). A post-hoc t-test for each group showed a significant decrease in RTs in the PT (p = 0.001, 95%CI: -323.96, -94.39), but not in the non-PT group (p = 0.56, 95%CI: -93.84, 167.91). However, ANOVA for incorrect items under the ToM condition revealed neither significant interaction (p = 0.074), nor significant within-group changes by post-hoc t-tests in the PT (p = 0.241, 95%CI: -470.33, 125.25) and non-PT groups (p = 0.163, 95%CI: -109.60, 606.19) (see Supplementary Material 3 for full results of 2 × 2 ANOVAs [Time (Time 1, Time 2) × Group (PT, non-PT)] conducted for questionnaire scores and RMET performance between Time 1 and Time 2).
Imaging results
For the whole brain analysis of the repeated-measures ANOVA revealed no significant changes in activity (interaction contrasts) in any region in the PT group as compared to the non-PT group at Time 2, under the ToM or GeN conditions. Likewise, no significant results were shown in the additional ROI analysis.
As few studies have investigated the PT effect on brain function of mothers caring for children with ADHD, and to balance Type I and II error risks, we explored the potential PT effects by using more lenient threshold (p < 0.005, uncorrected, with a cluster size of k ≥ 40 voxels) (Lieberman & Cunningham, 2009; Nishiyama et al., 2015) in repeated-measures ANOVA. We then observed bilateral clusters of increased activation in the calcarine cortex, left inferior temporal gyrus, and right inferior occipital gyrus (Supplementary Material 4, Table S1). The ROI analysis revealed activity changes in the left calcarine cortex (including occipital fusiform gyrus) in the PT group as compared to the non-PT group at Time 2 (Supplementary Material 5, Table S2).
Furthermore, planned within-group comparisons showed significantly increased activation in the left occipital fusiform gyrus in the PT group during the ToM condition at Time 2 as compared to Time 1 (Fig. 3, Table 3). The non-PT group showed no such difference. Other tests did not show any significant differences.
Regions showing significant differences between Time 1 and Time 2 in the PT group during the RMET. The colored regions show significantly increased activation after PT intervention in the PT group. The statistical threshold was set at p < 0.005, uncorrected at the peak level, and p < 0.05, family-wise error-corrected for multiple comparisons at the cluster level. Locations were defined using the Neuromorphometrics Atlas implemented in SPM. The color gradient bar on the right side of the figure indicates t-value, ranging from red (minimum) to yellow (maximum). Abbreviations: PT group, mothers enrolled in the parent training intervention. PT, parent training; RMET, Reading the Mind in the Eyes Test. X Y Z, Montreal Neurologic Institute coordinates.
Additionally, associations between increased signal changes of the left occipital fusiform gyrus, and degree of reduction of RTs under the ToM condition and reduced PSI and PS (Time 1 vs Time 2) scores were investigated using the MarsBar Toolbox (http://www.sourceforge.net/projects/marsbar). However, there were no significant correlations of brain activity with RTs or questionnaire scores (Supplementary Material 6, Table S3).
Discussion
In our study, mothers enrolled in the PT program exhibited less parenting stress, as measured in both the child- and parent-domains of the PSI. PS scores revealed that mothers in the PT group adopted fewer inappropriate overreactive parenting practices.
Our findings were consistent with those of previous studies using PT for mothers with ADHD children, showing decreased maternal stress and enhanced positive parenting practices after PT (Chronis-Tuscano et al., 2013; Heath et al., 2015; Shimabukuro et al., 2017; Treacy et al., 2005). Presumably, these results indicate that PT promotes mothers’ understanding of the behavior of their children with ADHD, thus reducing their stress and improving parenting disciplines. Considering the content of PT, reduced stress might suggest that, by participating in PT, mothers increased their ADHD knowledge and their understanding of the cause-and-effect relationships between their own emotional responses to the child’s misbehaviors, which changed their perception of these behaviors, and thus reduced stress. For improved parenting practices, mothers might well learn the content and purpose of our PT; particularly training on how to provide clear explanations and rules for their children, and to respond appropriately to the children’s ADHD-related non-adaptive behaviors.
Additionally, we conducted PT as a group-based program. Previous studies have shown that group-based programs improved parenting stress and practices in mothers of children with ADHD (Shimabukuro et al., 2017; Treacy et al., 2005). The group-based program provides opportunities for mothers to discuss and share their concerns with peers having similar difficulties (in particular, ADHD), which may further motivate them to participate in the program and enhance their mental health. However, since the non-PT group was not given this opportunity, the effects may include not only those of PT itself, but also the effects of the group meetings.
For the behavioral performance of RTs in the RMET under the ToM condition, the main effect of Time and interaction of Time × Group showed a trend toward significance. The post-hoc test showed significantly reduced RTs at Time 2 only for the PT group. The results of the interaction of Time × Group and the post-hoc test suggest that the PT group tends to improve more with respect to RT compared to non-PT group. However, this should be interpreted with caution, as the main effect of Time showed that improvement tends to be time dependent regardless of the group. In additional analyses, the interaction effect on correct responses remained a trend toward significance, but the main effect of Time was (more strongly) significant, suggesting that the effect of time might be stronger than the effect of PT on the decrease in RTs to correct responses. For the behavioral performance of accuracy in the RMET, a trend toward significance was observed for the main effect of Time, showing somewhat worse performance at Time 2 regardless of the group. It is unclear why RTs and accuracy tended to decrease regardless of the group, but one possible mechanism may be reduced engagement with the task at Time 2 due to boredom and/or habituation (Orr & Stern, 1970). However, this study did not assess the degree of engagement with the task, and this interpretation remains a speculation. Regarding RMET scores, another possibility is the ceiling effect in neurotypical adults, and thus, this tool may be insensitive to slight differences (Black, 2019). Figure 4 shows the distribution of accuracy on ToM condition.
Histograms of accuracy (correct %) under the ToM condition at Time 1 and Time 2. Histograms of accuracy for ToM condition. Upper panel denotes scores in Time 1, and lower panel denotes scores in Time 2. Abbreviations: ToM, Theory of Mind; PT, mothers enrolled in parent training intervention; non-PT, mothers who did not enroll in the parent training intervention
No significant changes in regional brain activation was found in the PT group as compared to the non-PT group under either condition. The current results may underestimate relevant differential effects of PT due to the lack of power caused by the small sample size. However, using a more lenient threshold, we did observe increased activation in the calcarine cortex, inferior temporal gyrus, and inferior occipital gyrus at Time 2 in the PT group, as compared with the non-PT group. These regions are known to be involved in processing facial information, including emotion recognition (Collins et al., 2012; Pitcher et al., 2011). Indeed, previous neuroimaging studies using RMET also reported involvement of these regions during this task (Adams et al., 2010; Castelli et al., 2010; Schmidt et al., 2020), which agrees with our findings. Furthermore, additional ROI analysis showed activation changes in overlapping region in the left visual cortex (including occipital fusiform gyrus). However, these regions did not survive correction for multiple testing, and the findings should be interpreted cautiously. Planned within-group comparisons showed significantly increased activation in the occipital part of the fusiform gyrus while judging others’ affective states from facial features only in the PT group under the ToM condition. The fusiform gyrus has been reported to be involved in face and object perception (Fusar-Poli et al., 2009), understanding of socioemotional meanings (Gallagher et al., 2000), and in the perception of emotions in facial expressions (Baron-Cohen et al., 1999; Greimel et al., 2010; Li et al., 2019). In one study, using the RMET, Batista et al. (2017) found a positive correlation between ToM scores and cortical thickness in the fusiform gyrus. In another study, Li et al. (2019) used intracranial electrodes and suggested that the fusiform gyrus participates in a relatively early stage of facial expression processing. Thus, we presumed that the increased brain activity and reduced reaction times seen in the PT group might reflect their improved sensitivity/efficiency to others’ affective states.
Taken together, we presumed that these changes might reflect that PT could reduce stress, and involvement of the fusiform gyrus. However, the causal relationship was not clarified in this study since this could not be proven by correlation analysis.
This study has some limitations. First, as mentioned above, it included a relatively small sample size and lacked a control group comprising mothers of typically developed children. Second, as all psychometric measures were self-reported, subjective bias might be present. Lastly, because of the single-center study design, the participants’ demographic variability might be limited. Future research should address these limitations.
Conclusions
In summary, we examined the neural and behavioral effects of PT on mothers rearing children with ADHD. Mothers who participated in PT showed significantly reduced stress and improved parenting practices, and elevated activity in the fusiform gyrus during emotion recognition. This suggests that participation in PT can reduce stress, which may consequently increase activation of the fusiform gyrus.
Data availability
The dataset used for the current study is not publicly available due to the Privacy Act of Japan. Regarding questions about this data, readers may contact Professor Akemi Tomoda (atomoda@u-fukui.ac.jp).
References
Abidin, R. R. (1995). Parenting stress index: Professional manual. Psychological Assessment Resources.
Abikoff, H. B., Thompson, M., Laver-Bradbury, C., Long, N., Forehand, R. L., Miller Brotman, L., Klein, R. G., Reiss, P., Huo, L., & Sonuga-Barke, E. (2015). Parent training for preschool ADHD: A randomized controlled trial of specialized and generic programs. Journal of Child Psychology and Psychiatry, and Allied Disciplines, 56(6), 618–631. https://doi.org/10.1111/jcpp.12346
Adams, R. B., Rule, N. O., Franklin, R. G., Wang, E., Stevenson, M. T., Yoshikawa, S., Nomura, M., Sato, W., Kveraga, K., & Ambady, N. (2010). Cross-cultural reading the mind in the eyes: An fMRI investigation. Journal of Cognitive Neuroscience, 22(1), 97–108. https://doi.org/10.1162/jocn.2009.21187
American Psychiatric Association. (2013). Diagnostic and statistical manual of mental disorders (DSM-5®). American Psychiatric Association.
Arnold, D. S., O’Leary, S. G., Wolff, L. S., & Acker, M. M. (1993). The Parenting Scale: A measure of dysfunctional parenting in discipline situations. Psychological Assessment, 5(2), 137–144. https://doi.org/10.1037/1040-3590.5.2.137
Ashburner, J. (2007). A fast diffeomorphic image registration algorithm. NeuroImage, 38(1), 95–113. https://doi.org/10.1016/j.neuroimage.2007.07.007
Baron-Cohen, S., Ring, H. A., Wheelwright, S., Bullmore, E. T., Brammer, M. J., Simmons, A., & Williams, S. C. (1999). Social intelligence in the normal and autistic brain: An fMRI study. The European Journal of Neuroscience, 11(6), 1891–1898. https://doi.org/10.1046/j.1460-9568.1999.00621.x
Baron-Cohen, S., Wheelwright, S., Hill, J., Raste, Y., & Plumb, I. (2001). The “Reading the Mind in the Eyes” Test revised version: A study with normal adults, and adults with Asperger syndrome or high-functioning autism. Journal of Child Psychology and Psychiatry, and Allied Disciplines, 42(2), 241–251. https://doi.org/10.1111/1469-7610.00715
Baron-Cohen, S., Ring, H., Chitnis, X., Wheelwright, S., Gregory, L., Williams, S., Brammer, M., & Bullmore, E. (2006). fMRI of parents of children with Asperger Syndrome: A pilot study. Brain and Cognition, 61(1), 122–130. https://doi.org/10.1016/j.bandc.2005.12.011
Batista, S., d’Almeida, O. C., Afonso, A., Freitas, S., Macário, C., Sousa, L., Castelo-Branco, M., Santana, I., & Cunha, L. (2017). Impairment of social cognition in multiple sclerosis: Amygdala atrophy is the main predictor. Multiple Sclerosis (houndmills, Basingstoke, England), 23(10), 1358–1366. https://doi.org/10.1177/1352458516680750
Baumeister, S., Wolf, I., Holz, N., Boecker-Schlier, R., Adamo, N., Holtmann, M., Ruf, M., Banaschewski, T., Hohmann, S., & Brandeis, D. (2018). Neurofeedback Training Effects on Inhibitory Brain Activation in ADHD: A Matter of Learning? Neuroscience, 378, 89–99. https://doi.org/10.1016/j.neuroscience.2016.09.025
Baumeister, S., Wolf, I., Hohmann, S., Holz, N., Boecker-Schlier, R., Banaschewski, T., & Brandeis, D. (2019). The impact of successful learning of self-regulation on reward processing in children with ADHD using fMRI. Attention Deficit and Hyperactivity Disorders, 11(1), 31–45. https://doi.org/10.1007/s12402-018-0269-6
Beck, A. T., Steer, R. A., & Brown, G. K. (1996). Manual for the beck depression inventory-II. Psychological Corporation.
Black, J. E. (2019). An IRT Analysis of the Reading the Mind in the Eyes Test. Journal of Personality Assessment, 101(4), 425–433. https://doi.org/10.1080/00223891.2018.1447946
Castelli, I., Baglio, F., Blasi, V., Alberoni, M., Falini, A., Liverta-Sempio, O., Nemni, R., & Marchetti, A. (2010). Effects of aging on mindreading ability through the eyes: An fMRI study. Neuropsychologia, 48(9), 2586–2594. https://doi.org/10.1016/j.neuropsychologia.2010.05.005
Chronis, A. M., Lahey, B. B., Pelham, W. E., Williams, S. H., Baumann, B. L., Kipp, H., Jones, H. A., & Rathouz, P. J. (2007). Maternal depression and early positive parenting predict future conduct problems in young children with attention-deficit/hyperactivity disorder. Developmental Psychology, 43(1), 70–82. https://doi.org/10.1037/0012-1649.43.1.70
Chronis-Tuscano, A., Raggi, V. L., Clarke, T. L., Rooney, M. E., Diaz, Y., & Pian, J. (2008). Associations between maternal attention-deficit/hyperactivity disorder symptoms and parenting. Journal of Abnormal Child Psychology, 36(8), 1237–1250. https://doi.org/10.1007/s10802-008-9246-4
Chronis-Tuscano, A., Clarke, T. L., O’Brien, K. A., Raggi, V. L., Diaz, Y., Mintz, A. D., Rooney, M. E., Knight, L. A., Seymour, K. E., Thomas, S. R., Seeley, J., Kosty, D., & Lewinsohn, P. (2013). Development and preliminary evaluation of an integrated treatment targeting parenting and depressive symptoms in mothers of children with attention-deficit/hyperactivity disorder. Journal of Consulting and Clinical Psychology, 81(5), 918–925. https://doi.org/10.1037/a0032112
Collins, H. R., Zhu, X., Bhatt, R. S., Clark, J. D., & Joseph, J. E. (2012). Process and domain specificity in regions engaged for face processing: An fMRI study of perceptual differentiation. Journal of Cognitive Neuroscience, 24(12), 2428–2444. https://doi.org/10.1162/jocn_a_00273
Daley, D., van der Oord, S., Ferrin, M., Danckaerts, M., Doepfner, M., Cortese, S., Sonuga-Barke, E. J. S., & European ADHD Guidelines Group. (2014). Behavioral interventions in attention-deficit/hyperactivity disorder: a meta-analysis of randomized controlled trials across multiple outcome domains. Journal of the American Academy of Child and Adolescent Psychiatry, 53(8), 835–847, 847.e1–5 https://doi.org/10.1016/j.jaac.2014.05.013
Davis, M. H. (1983). Measuring individual differences in empathy: Evidence for a multidimensional approach. Journal of Personality and Social Psychology, 44(1), 113.
Focquaert, F., Steven-Wheeler, M. S., Vanneste, S., Doron, K. W., & Platek, S. M. (2010). Mindreading in individuals with an empathizing versus systemizing cognitive style: An fMRI study. Brain Research Bulletin, 83(5), 214–222. https://doi.org/10.1016/j.brainresbull.2010.08.008
Fusar-Poli, P., Placentino, A., Carletti, F., Landi, P., Allen, P., Surguladze, S., Benedetti, F., Abbamonte, M., Gasparotti, R., Barale, F., Perez, J., McGuire, P., & Politi, P. (2009). Functional atlas of emotional faces processing: A voxel-based meta-analysis of 105 functional magnetic resonance imaging studies. Journal of Psychiatry & Neuroscience : JPN, 34(6), 418–432.
Gallagher, H. L., Happé, F., Brunswick, N., Fletcher, P. C., Frith, U., & Frith, C. D. (2000). Reading the mind in cartoons and stories: An fMRI study of “theory of mind” in verbal and nonverbal tasks. Neuropsychologia, 38(1), 11–21. https://doi.org/10.1016/s0028-3932(99)00053-6
Greimel, E., Schulte-Rüther, M., Kircher, T., Kamp-Becker, I., Remschmidt, H., Fink, G. R., Herpertz-Dahlmann, B., & Konrad, K. (2010). Neural mechanisms of empathy in adolescents with autism spectrum disorder and their fathers. NeuroImage, 49(1), 1055–1065. https://doi.org/10.1016/j.neuroimage.2009.07.057
Heath, C. L., Curtis, D. F., Fan, W., & McPherson, R. (2015). The association between parenting stress, parenting self-efficacy, and the clinical significance of child ADHD symptom change following behavior therapy. Child Psychiatry and Human Development, 46(1), 118–129. https://doi.org/10.1007/s10578-014-0458-2
Hidano, N., Fukuhara, M., Iwawaki, M., Soga, S., & Spielberger, C. D. (2000). State-trait anxiety inventory-form JYZ. Tokyo: Japan UNI Agency (in Japanese)
Hutchison, L., Feder, M., Abar, B., & Winsler, A. (2016). Relations between Parenting Stress, Parenting Style, and Child Executive Functioning for Children with ADHD or Autism. Journal of Child and Family Studies, 25(12), 3644–3656. https://doi.org/10.1007/s10826-016-0518-2
Itani, T. (2010). The Japanese version of the Parenting Scale: Factor structure and psychometric properties. The Japanese Journal of Psychology, 81(5), 446–452.
Iwasaka, H. (2012). Parent Training Guidebook. Jihou
Kojima, M., Furukawa, T. A., Takahashi, H., Kawai, M., Nagaya, T., & Tokudome, S. (2002). Cross-cultural validation of the Beck Depression Inventory-II in Japan. Psychiatry Research, 110(3), 291–299. https://doi.org/10.1016/s0165-1781(02)00106-3
Li, Y., Richardson, R. M., & Ghuman, A. S. (2019). Posterior Fusiform and Midfusiform Contribute to Distinct Stages of Facial Expression Processing. Cerebral Cortex (New York, N.Y. : 1991), 29(7), 3209–3219 https://doi.org/10.1093/cercor/bhy186
Lieberman, M. D., & Cunningham, W. A. (2009). Type I and Type II error concerns in fMRI research: Re-balancing the scale. Social Cognitive and Affective Neuroscience, 4(4), 423–428. https://doi.org/10.1093/scan/nsp052
Marshall, S. A., Evans, S. W., Eiraldi, R. B., Becker, S. P., & Power, T. J. (2014). Social and academic impairment in youth with ADHD, predominately inattentive type and sluggish cognitive tempo. Journal of Abnormal Child Psychology, 42(1), 77–90. https://doi.org/10.1007/s10802-013-9758-4
Narama, M., Kanematsu, Y., Araki, A., Maru, M., Nakamura, N., Takeda, J., Shirahata, N., & Kudo, Y. (1999). Validity and Reliability of the Japanese Version of the Parenting Stress Index. Journal of Child Health, 58(5), 610–616.
Nicholls, M., Thomas, N., Loetscher, T., & Grimshaw, G. (2013). The Flinders Handedness survey (FLANDERS): A brief measure of skilled hand preference. Cortex, 49(10), 2914–2926. https://doi.org/10.1016/j.cortex.2013.02.002
Nishiyama, Y., Okamoto, Y., Kunisato, Y., Okada, G., Yoshimura, S., Kanai, Y., Yamamura, T., Yoshino, A., Jinnin, R., Takagaki, K., Onoda, K., & Yamawaki, S. (2015). fMRI Study of Social Anxiety during Social Ostracism with and without Emotional Support. PloS One, 10(5), e0127426. https://doi.org/10.1371/journal.pone.0127426
Nolte, T., Bolling, D. Z., Hudac, C. M., Fonagy, P., Mayes, L., & Pelphrey, K. A. (2013). Brain mechanisms underlying the impact of attachment-related stress on social cognition. Frontiers in Human Neuroscience, 7, 816. https://doi.org/10.3389/fnhum.2013.00816
Okubo, M., Suzuki, H., & Nicholls, M. (2014). A Japanese version of the FLANDERS handedness questionnaire. The Japanese Journal of Psychology, 85(5), 474–481. https://doi.org/10.4992/jjpsy.85.13235
Orr, W. C., & Stern, J. A. (1970). The relationship between stimulus information, reaction time, and cortical habituation. Psychophysiology, 7(3), 475–484. https://doi.org/10.1111/j.1469-8986.1970.tb01773.x
Pelham, W. E., Lang, A. R., Atkeson, B., Murphy, D. A., Gnagy, E. M., Greiner, A. R., Vodde-Hamilton, M., & Greenslade, K. E. (1998). Effects of deviant child behavior on parental ADHD alcohol consumption. Stress-induced drinking in parents of children. The American Journal on Addictions, 7(2) 103–114
Pitcher, D., Walsh, V., & Duchaine, B. (2011). The role of the occipital face area in the cortical face perception network. Experimental Brain Research, 209(4), 481–493. https://doi.org/10.1007/s00221-011-2579-1
Posner, J., Polanczyk, G. V., & Sonuga-Barke, E. (2020). Attention-deficit hyperactivity disorder. Lancet (london, England), 395(10222), 450–462. https://doi.org/10.1016/S0140-6736(19)33004-1
Sakurai, S. (1988). The relationship between empathy and helping behavior in college students. Bulletin of Nara University of Education (cultural and Social Science), 37, 149–154.
Schmidt, A., Davies, C., Paloyelis, Y., Meyer, N., De Micheli, A., Ramella-Cravaro, V., Provenzani, U., Aoki, Y., Rutigliano, G., Cappucciati, M., Oliver, D., Murguia, S., Zelaya, F., Allen, P., Shergill, S., Morrison, P., Williams, S., Taylor, D., Borgwardt, S., & Fusar-Poli, P. (2020). Acute oxytocin effects in inferring others’ beliefs and social emotions in people at clinical high risk for psychosis. Translational Psychiatry, 10(1), 203. https://doi.org/10.1038/s41398-020-00885-4
Shimabukuro, S., Daley, D., Thompson, M., Laver-Bradbury, C., Nakanishi, E., & Tripp, G. (2017). Supporting Japanese Mothers of Children with ADHD: Cultural Adaptation of the New Forest Parent Training Programme. Japanese Psychological Research, 59(1), 35–48. https://doi.org/10.1111/jpr.12140
Shimada, K., Kasaba, R., Fujisawa, T. X., Sakakibara, N., Takiguchi, S., & Tomoda, A. (2018). Subclinical maternal depressive symptoms modulate right inferior frontal response to inferring affective mental states of adults but not of infants. Journal of Affective Disorders, 229, 32–40. https://doi.org/10.1016/j.jad.2017.12.031
Smith, L. E., Greenberg, J. S., Seltzer, M. M., & Hong, J. (2008). Symptoms and behavior problems of adolescents and adults with autism: effects of mother-child relationship quality, warmth, and praise. American Journal of Mental Retardation : AJMR, 113(5), 387–402. https://doi.org/10.1352/2008.113:387-402
Spielberger, C. D., Gorsuch, R. L., Luschene, R. E., Vagg, P. R., & Jacobs, G. A. (1983). Manual for the State-Trait Anxiety Inventory (Form Y). Menlo Park. CA: Mind Garden
Treacy, L., Tripp, G., & Baird, A. (2005). Parent Stress Management Training for Attention-Deficit/Hyperactivity Disorder
Tully, L. A., Arseneault, L., Caspi, A., Moffitt, T. E., & Morgan, J. (2004). Does maternal warmth moderate the effects of birth weight on twins’ attention-deficit/hyperactivity disorder (ADHD) symptoms and low IQ? Journal of Consulting and Clinical Psychology, 72(2), 218–226. https://doi.org/10.1037/0022-006X.72.2.218
Woodman, A. C., Mawdsley, H. P., & Hauser-Cram, P. (2015). Parenting stress and child behavior problems within families of children with developmental disabilities: Transactional relations across 15 years. Research in Developmental Disabilities, 36C, 264–276. https://doi.org/10.1016/j.ridd.2014.10.011
Zwi, M., Jones, H., Thorgaard, C., York, A., & Dennis, J. A. (2011). Parent training interventions for Attention Deficit Hyperactivity Disorder (ADHD) in children aged 5 to 18 years. The Cochrane Database of Systematic Reviews, 12, CD003018. https://doi.org/10.1002/14651858.CD003018.pub3
Acknowledgements
The authors would like to thank all the mothers and their children who generously and courageously participated in this research. We also would like to express our sincere appreciation to the Okinawa Institute of Science and Technology Graduate University (OIST) team, especially Prof. Gail Tripp and Dr. Shizuka Shimabukuro who helped us to improve the skills of our trainers. In addition, we also thank Editage [http://www.editage.com] for editing and reviewing this manuscript.
Funding
Open access funding provided by Kobe University. This research was supported by funds from a Grant-in-Aid for Young Scientists (JP18K13107 to KM), Grant-in-Aid for Scientific Research C (JP21K02352 to KM), Challenging Exploratory Research (JP19K21755 to AT), and Grant-in-Aid for “Creating a Safe and Secure Living Environment in the Changing Public and Private Spheres” from the Japan Science and Technology Corporation (JST)/Research Institute of Science and Technology for Society (RISTEX) to AT.
Author information
Authors and Affiliations
Contributions
AT conceived the project. KM, AY, KS, RK, and AT designed the experiments. KM, AY, RK, KS, TF, and AT performed the experiments, collected the data, and analyzed the data. KM, YM, and AT wrote the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
The research protocol was approved by the Ethics Committee of the University of Fukui.
Consent to participate
Written informed consent was obtained from all participants.
Consent for publication
Not applicable.
Code availability
Not applicable.
Conflicts of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Makita, K., Yao, A., Shimada, K. et al. Neural and behavioral effects of parent training on emotion recognition in mothers rearing children with attention-deficit/hyperactivity disorder. Brain Imaging and Behavior 17, 436–449 (2023). https://doi.org/10.1007/s11682-023-00771-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-023-00771-9