Abstract
Evaluating the influences of fine-scale habitat heterogeneity on the composition, diversity, structure and functioning of forests is critical to understand how tropical forests will respond to climate change and devise forest management strategies that will enhance biodiversity conservation and aboveground biomass stock. Here, we hypothesized that topographic and soil factors determine fine-scale habitat differentiation, which in turn shape community composition, species richness, structure and aboveground biomass at the local scale in tropical forests. To test this hypothesis, we selected two areas (each 100 × 100 m) with contrasting fine-scale topographic conditions where all trees, palms and lianas with a diameter at breast height ≥ 10 cm were tagged and identified to species. In each selected area, 100 subplots of 10 × 10 m were established. We mainly found that higher topographic variability caused higher habitat differentiation with changes in species composition and community structure, but did not change species richness. Our habitat-scale analyses indicated that, in the less heterogeneous area, the distribution of species was more uniform along a fine-scale topographical gradient with no variation in convexity, which induced changes in structure and aboveground biomass, but not in species richness. The nonsignificant relationship between species richness and aboveground biomass may be attributable to species redundancy or functional dominance. This study suggests that environmental filtering is a fundamental process for shaping community assembly and forest functioning along a local topographical gradient in tropical forests.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding the roles of environmental factors for determining community assembly, ecosystem functioning and biodiversity recovery of tropical forests is a central focus in forest ecology (Ali et al. 2018a; Poorter et al. 2017; Rozendaal et al. 2019). Tropical forests harbor more than half of the global biodiversity and have a major influence on the mitigation of climate change while providing important ecosystem services that humans depend on (Lewis et al. 2015). For instance, aboveground biomass stock, as a key ecosystem property, in tropical forests plays a vital role in the global carbon cycle (Lewis et al. 2015; Anderson-Teixeira et al. 2016) by sequestering carbon dioxide, the main contributor to the greenhouse effect (Anderson-Teixeira et al. 2016). Several studies have shown that topographic factors (e.g., convexity, elevation and slope) can determine plant growth (Moeslund et al. 2013), species distribution (Toledo et al. 2012) and diversity patterns (Ali et al. 2018a; Rodrigues et al. 2019). As such, topography modulates the relationships between species diversity and ecosystem functioning in tropical forests (Ali et al. 2019; Jucker et al. 2018). However, more research is needed to understand the influences of habitat heterogeneity on species diversity, composition, structure and aboveground biomass and to understand the main mechanisms underlying the fine-scale community assembly in species-rich, structurally complex natural tropical forests.
Topography can determine resource availability (e.g., energy, soil nutrients and climatic water), which provide different habitats that favor the differential use of resources by tree species (McEwan and Muller 2006; Brown et al. 2013). The habitat differentiation is important because it can lead to an increased differential number of coexisting species and therefore strongly shapes the community assembly (Brown et al. 2013; Liu et al. 2014; Jucker et al. 2018). It is generally well-understood that topographic or geographical gradients in climatic factors (such as mean annual temperature and precipitation) influence species diversity, structure and function of the forests over a large scale (Jucker et al. 2018; Ali et al. 2019), whereas edaphic factors (such as soil physical and chemical properties) did so at a local or fine scale (Chiang et al. 2016; Ali et al. 2018b). In this context, it is understandable that topographic, climatic and edaphic factors determine the habitats differentiation in forests (i.e., Wang et al. 2016; Guo et al. 2017), and hence may affect community composition, species richness, structure and functioning of the forests due to the heterogeneity of the available resources (Liu et al. 2014; Guo et al. 2017; Ali et al. 2018a, b, 2019; Rodrigues et al. 2019). Determining how habitat differentiation can affect tree communities can also be quite relevant for the management and conservation of forests at a fine and local scale.
The Atlantic Forest in Brazil is a hotspot of vascular plant diversity (Murray-Smith et al. 2009) and has a high capacity for carbon storage in the standing biomass (Magnago et al. 2015). Nevertheless, it is also one of the most threatened tropical forests in the world, mainly due to deforestation and fragmentation (Laurance 2009; Ribeiro et al. 2011). Currently, only around 10% of the mature forests are conserved, while the remnants of native vegetation are restricted to ca. 20% of the original forest cover (Scarano and Ceotto 2015). Hence, evaluating the species diversity and community structure along topographical gradients and across habitat differentiation in these Atlantic forests is crucial for understanding forest functioning.
This study aimed to evaluate whether habitat differentiation affects the community composition, species richness, structure and aboveground biomass along a local topographical gradient in an Atlantic forest in Minas Gerais state, southeastern Brazil. Specifically, we hypothesized that (1) topographical factors and soil properties determine habitat differentiation; (2) tree community composition, species richness, structure and aboveground biomass will change in different habitats; and (3) tree diversity is positively related with aboveground biomass along a fine-scale topographical gradient. This study allowed us to investigate at the community scale whether habitat differentiation increases with increasing variability in topographical and soil factors, thereby leading to increased species richness and enhanced aboveground biomass at a fine scale.
Materials and methods
Study area
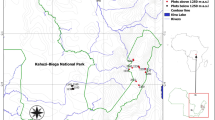
The seasonal semideciduous Atlantic forest fragment studied in Viçosa municipality, Minas Gerais state, southeastern Brazil (Fig. 1) had been used for shade coffee cultivation under natural forest cover until 1926, when it became fully protected, allowing for natural regeneration to occur. Later, land-use has been reserved (Paula et al. 2002). The study area has a moderate humid tropical climate, with a dry season from May to September and a wet season between December and March. The mean annual relative humidity is ca. 80%, mean annual air temperature is 19 °C, and mean annual precipitation is 1340 mm.
Location of the study area in relation to South America (a), the Minas Gerais State, Brazil (b), and the forest fragment within the campus of the Federal University of Viçosa (UFV), Viçosa municipality, Minas Gerais state, southeastern Brazil (c). Location of the two sample plots within the forest fragment (d)
The study area is located between 620 and 820 m a.s.l., and the relief varies from strongly undulating to mountainous. A Dystric Red-Yellow Latosol soil dominates in hilltops and mountainsides, and a Cambic Red-Yellow Podzolic dominates in the upper fluvial terraces (EMBRAPA 1997).
Forest inventory and data collection
We selected two sampling areas with contrasting topographic conditions: a southeastern area and a northeastern area. Each area (100 × 100 m) was subdivided into 100 plots of 10 × 10 m (Fig. 1). A total of 200 plots (2 ha) from the two areas were sampled from December 2016 to January 2017. Within each plot, all trees having a diameter at breast height (DBH) 10 cm were identified to the species level and tagged for the measurement of tree height. All individuals were identified using specialized literature, through consultation with the Herbarium of Universidade Federal de Viçosa, or by taxonomists. The Angiosperm Phylogeny Group IV (APG IV 2016) was used for taxon classification.
Estimation of aboveground biomass
The aboveground biomass of individual stems was calculated using the general allometric equation (Eq. 1) proposed by Chave et al. (2014), based on tree DBH (cm), height (H, m) and species wood density (ρ, g cm−3). We used data from the Global Wood Density Database (Zanne et al. 2009; Chave et al. 2009) to obtain the wood density of each species, using genus or family averages whenever species-level information was not available (e.g., Jucker et al. 2018; Ali et al. 2019).
The total aboveground biomass per plot was the sum of the aboveground biomass of all trees having DBH ≥ 10 cm, which was then converted to megagrams per hectare (Mg ha−1) (Ali et al. 2018b). Species-level biomass was calculated as the sum of the biomass of all individuals from a given species. Estimation of aboveground biomass was performed using the R package BIOMASS (Réjou-Méchain et al. 2017).
Measurements of topographic variables
We measured three topographic variables (i.e., slope, elevation, and convexity) within each plot. Topographic variables were measured using a total station, which measures vertical and horizontal angles and linear distances and is positioned at an obstacle-free location and aimed at a prism. The prism sits on a metal stick and placed over the point to be measured. The total station then emits a laser beam that reflects in the prism and returns to the equipment. Using the response time of of the laser beam to the equipment and the angle of rotation of the station’s bezel, the internal computer calculates the angles and distances and stores the data in its internal memory (Kahmen and Faig 1988). The data was then transferred to a computer and analyzed with AutoCAD software (Autodesk Inc., San Rafael, CA, USA).
Elevation was calculated using the mean elevation at each of the four corners of the plot. The slope (measured in degrees) was the mean angular deviation of the horizontal of each of the four triangular planes formed by the connection of three of its edges (Guo et al. 2017). Convexity was determined by subtracting the elevation at the centre of the quadrat from the mean elevation of the eight surrounding plots. On edge plots, convexity was calculated as the altitude of the plot of interest minus the mean altitude of the surrounding plots (Lan et al. 2011).
Measurements of soil properties
Within each plot, a composite sample of the surface soil (0–10 cm depth) was collected. Soil properties of the samples were measured in the Soil Analysis Laboratory of the Federal University of Viçosa, following standard protocols (EMBRAPA 1997). The following soil properties were assessed: soil organic carbon (C); total N; available P, K, Ca, Mg, Fe, Zn; effective cation exchange capacity (CEC); exchangeable acidity (H + Al); sum of bases (SB); base saturation (V); aluminum saturation (m); pH and organic matter (OM).
Data and statistical analyses
All data and statistical analyses were conducted in R.3.2.2 (R-Core-Team 2017). Soil properties were summarized through principal components analysis (PCA) on the correlation matrix, using the FactoMineR package (Husson et al. 2018). For this purpose, all variables were centered and standardized to meet the assumptions of linearity and normality. After that, we performed multivariate regression tree (MRT) analysis (De’ath 2002; Larsen and Speckman 2004) to group habitats with similar species composition (i.e., species assemblages) according to topographic variables (Punchi-Manage et al. 2013; Wang et al. 2016; Guo et al. 2017). MRT is a method of constrained clustering that identifies clusters (a group of plots) that are most similar to each other based on a set of predefined values (De’ath 2002). We then analyzed species dissimilarity (Euclidian distance) between each cluster as being defined by threshold values of topographic variables (De’ath 2002). In this study, the root node consisted of all 100 plots (10 × 10 m) from each area. Each cluster defines a species assemblage, and the threshold values of environmental variables define an associated habitat type (Larsen and Speckman 2004; Punchi-Manage et al. 2013; Rodrigues et al. 2019). MRT analysis was performed using the rpart package (Therneau et al. 2017). We represented the spatial distribution of topographic variables and species composition from each area using Kriging maps with the Field package (Nychka et al. 2019).
Species richness in the two sampled areas was evaluated using both individual-based and sampled-based rarefaction and extrapolation curves, which were constructed using the first (species richness, q = 0) Hill number (Chao et al. 2014). Extrapolations were made from the abundance data considering between two and three times the total sample size by habitat type (Colwell et al., 2012). Sample and individual-based rarefaction/extrapolations were computed using the iNEXT package (Hsieh et al. 2016). Rarefaction was estimated as the mean of 100 replicate bootstrapping runs to estimate 95% confidence intervals. Whenever the 95% confidence intervals did not overlap, species numbers differed significantly at p < 0.05 (Colwell et al. 2012).
Differences in community composition among habitats in each of the two areas was evaluated using nonmetric multidimensional scaling (NMDS) and Jaccard similarity (Clarke 1993) in the R package vegan (Oksanen et al. 2018). We used permutation multivariate analysis of variance (PERMANOVA, 9999 permutations) to determine differences in species composition, also using vegan (Oksanen et al. 2018). We used axis 1 of the NMDS (Euclidian distance) as a proxy for community composition, which explains the highest amount of variance with absence/presence data (Oksanen et al. 2018; Villa et al. 2018).
Variables were tested for normal distribution by evaluating the Q–Q plot, and homogeneity of variances by Bartlett’s test using the dplyr package (Crawley 2012; Wickham et al. 2018). To compare means of the variables (i.e., topography and soil factors, community composition, species richness, abundance, tree height, and aboveground biomass) among habitats in each of the two areas, we used a one-way ANOVA (for normally distributed data) followed by a post hoc Tukey’s test (p < 0.05), and PERMANOVA (for non-normally distributed data) followed by a posterior pairwise Adonis test (Martinez-Arbizu 2019). All these analyses were performed with the packages stats and pairwiseadonis (Martinez-Arbizu 2019).
We constructed a series of linear models to find the most parsimonious models explaining the main effects of predictor variables (i.e., topographic and soil factors) on the response variables (i.e., community composition, species richness, abundance, tree height, aboveground biomass) across the local scale topographical gradient. We also tested the main effects of species richness on community structural attributes (i.e., abundance, tree height, aboveground biomass). We used a generalized linear mixed effects model (GLMMs) with Poisson error distribution to investigate the effects of multiple predictors on species richness. Generalized linear models (GLMs) with negative binomial distributions to assess the effects of predictors on abundance. The effects of topographic factors, soil factors and species richness on aboveground biomass and community composition were calculated using linear mixed effects models (LMMs) with Gaussian distributions. We assessed collinearity between selected predictor variables using Pearson correlation analysis, and when two variables were strongly correlated (r ≥ 0.7) they were then included into the separate models (Fig. S1. Appendix/from Electronic Supplement Material, ESM hereafter). Predictor variables were grouped into three categories, i.e., topographic variables, soil properties and species richness. The topographic variables included elevation, slope and convexity, whereas soil properties included pH and exchangeable acidity (H + AL). In these models, the identity of the subplots as pseudoreplication in each area was included as a random factor to avoid autocorrelation.
To select the best model, we applied a multi-model inference approach (Burnham and Anderson 2002) with the MuMIn package (Barton 2017), which allows all possible combinations of the explanatory variables included in the global model (Barton 2017). To determine which of these variables were the most decisive in explaining changes in community composition, species richness, abundance, community structural and aboveground biomass, we used an information theoretical approach based on the Akaike information criterion with a correction for small sample sizes (AICc) and AICc weights (Burnham and Anderson 2002). We selected the best model with the lowest AICc and all models whose difference in AICc with the best model was less than four units (Burnham et al. 2011). All models were calculated in R using the packages lme4, nlme, and MASS (Pinheiro and Bates 2017; Ripley 2017; Bates et al. 2019).
Results
We observed an evident differentiation between areas in relation to topographic and soil variables. The first two axes of the PCA explained 66.8% of the commulative variation in the topographical and soil factors (Fig. 2). The first axis explained 49.6% of variance and was positively correlated with cation exchange capacity (r = 0.55, p < 0.001) and pH (r = 0.42, p < 0.001). The second axis explained 17.2% of the variation and was positively correlated with elevation but negatively with a slope (Fig. 2).
Biplot of the principal component analysis (PCA) for the topographic and soil factors of the study area within the campus of the Federal University of Viçosa, Viçosa municipality, Minas Gerais state, southeastern Brazil. For analysis, elevation (elev), slope, convexity (convex), pH, effective cation exchange capacity (t), exchangeable acidity (HAl), sum of bases (SB), base saturation (V), organic matter (mo) were included
The southeastern area was topographically less heterogeneous, with the habitat types determined by the two topographic factors, i.e., elevation and slope, and hence can be divided into five habitats, i.e., (1) high plateau, (2) intermediate plateau, (3) low plateau, (4) high valley, and (5) low valley (Table 1). The northeastern area was topographically more heterogeneous, where habitat types were determined by all three topographic variables (elevation, slope, and convexity), and hence can be divided into seven habitats. The northeastern area (NE) consisted of the same types of habitats as the southeastern area (NE), but had two additional habitat types, i.e., (1) intermediate low valley and (2) a transition area between the high valley and low plateau (Table 1). The soil properties were not important in the habitats differentiation. Elevation determined the first split of MRT for habitat types in both areas (breakpoint: SE = 711, NE = 716), accounted for 21–34% of species variance, and the second split accounted lower values (< 10%) of species variance. In the SE, elevation was also the main factor for the second (breakpoint = 718) and third (breakpoint = 705) split of habitat types, followed by slope, which determined fourth split (breakpoint = 29). Convexity was not included in the MRT for the SE but it was a main factor for the second split (breakpoint = 0.37) in the NE. Elevation determined the third (breakpoint = 707) and fifth (breakpoint = 703) split and slope determined the fourth (breakpoint = 28) and sixth (breakpoint = 19) splits of habitat types in the NE (Fig. S2. from ESM).
The NMDS showed that tree species composition varied considerably among habitats along the topographical gradient (Fig. 3). We found no significant difference between the species richness patterns of the different habitats using both individual-based and sampled-based rarefaction and extrapolation curves (Fig. 4). Likewise, species richness at the plot level did not differ among habitats (Table 1).
Nonmetric multidimensional scaling based on species composition according to habitats (point colors) by study area, southeastern (a) and northeastern (b) within the campus of the Federal University of Viçosa, Viçosa municipality, Minas Gerais state, southeastern Brazil. According to the MRT, the areas were divided into the following habitats: (high plateau (Hp); intermediate plateau (Ip); low plateau (Lp); high valley (Hv); low valley (Lv); intermediate low valley (Iv), and transition area between the high valley and low plateau (Tpv)
Sample-based and individual-based rarefaction (solid line) and extrapolation curves (dashed lines) of species richness for different habitats by northeastern (a, c) and southeastern (b, d) areas within the campus of the Federal University of Viçosa, Viçosa municipality, Minas Gerais state, southeastern Brazil. According to the MRT, the areas were divided into the following habitats: high plateau (Hp); intermediate plateau (Ip); low plateau (Lp); high valley (Hv); low valley (Lv); intermediate low valley (Iv), and transition area between the high valley and low plateau (Tpv). Rarefaction and extrapolation curves present the lines that represent the mean values and the bands the standard deviation with 95% confidence intervals
Community structural attributes and aboveground biomass did not show differences among habitats in the NE (Table 1). However, when analyzed at the area scale, basal area, tree height and aboveground biomass differed significantly between low valleys and high plateaus in the SE. Subplots in depressions were dominated by species that had high tree maximum height and aboveground biomass, but low basal area (Table 1). Finally, soil factors differed significantly among habitats only in the SE (Table 1).
The multi-model comparison among habitats within areas showed that models including a single topographical factor (i.e., elevation) consistently explained more variation in community composition than those with multiple effects of topographic and soil factors (Table 2; Table S1. from ESM). At local-scale analysis, topographic factors were better predictors than soil properties. However, species richness, abundance, community structural and aboveground biomass was not related to different predictors. There was no significant relationship between species richness and aboveground biomass at the local scale. Species composition, elevation and slope showed a non-uniform spatial distribution in each area (Fig. 5).
Kriging maps of topographic variables (elevation and slope) and species composition along the environmental gradient in each area (100 × 100 m), northeastern (figures on the left) and southeastern (figures on the right) areas within the campus of the Federal University of Viçosa, Viçosa municipality, Minas Gerais state, southeastern Brazil. Each grid represents a community weighted variable value of a 10 × 10 m subplot. Elevational contours are indicated by black lines
Discussion
Our results showed that topography is an important driver for habitat differentiation, thereby determining community composition and structure, but not species richness, at the local scale in tropical forests. More specifically, we found that elevation and slope were the main factors explaining habitat differentiation in studied areas, and hence determining variation in community composition. Our results corroborate the hypothesis that topographic factors determine community composition, structure and aboveground biomass in tropical forests (Guo et al. 2017; Ali et al. 2018a; Jucker et al. 2018; Ali et al. 2019). The observed habitat structuring along topographical gradients is probably linked to the underlying spatial variation of light, soil nutrients and climatic water availability, which are strongly influenced by topographic factors (John et al. 2007; Moeslund et al. 2013; Ali et al. 2019). This study shows that environmental filtering is a fundamental process for shaping community assembly in tropical forests (e.g., Liu et al. 2014), even at a local-scale indicating that species composition changes along a topographical gradient.
Our results indicated that species richness, using both individual-based and sampled-based rarefaction and extrapolation curves, maintains a similar pattern between different habitats. Although the studied northeastern area had a species richness higher than the southeastern area at the plot scale, the observed pattern is consistent with the niche complementarity hypothesis, i.e., the occurrence of a positive diversity effect at the more heterogeneous area due to an increased resource use via niche differentiation (Tilman 1999; Pausas and Austin 2001; Poorter et al. 2017) caused by the higher topographic variability (Liu et al. 2014; Ali, et al. 2018a). Such variation among areas is probably due to the heterogeneous distribution of resources such as light, water and nutrients (e.g., Katabuchi et al. 2012). We presume that the nonsignificant differences for species richness among habitat types in the studied areas may be due to the marked difference in elevation of the topographic gradient.
Community structural attributes including maximum tree height and basal area as well as aboveground biomass differed among habitats in the studied southeastern area only. In our analyses, maximum tree height decreased from valleys to plateaus. Valley areas have a higher availability of soil water and nutrients as compared to plateau areas (Moeslund et al. 2013; Liu et al. 2014). The increased tree height observed in valleys is a typical feature of trees growing in areas with high resource availability, whereas the decreased height of trees from plateau habitats is characteristic of plants in resource-poor areas (Poorter 2009; Reich 2014). These results are consistent with the findings of a previous study that species distribution along the topographical gradients can be strongly influenced by habitat filtering, which selects attributes such as tree height, being related to resource use, such as the light niche and colonizing strategy (Liu et al. 2014; Ali et al. 2018a). While these results indicate that habitat differentiation influences the distributions of many individual species, the community-level effects of soil resource variation have not been examined extensively, which can be analyzing using soil nutrients and topographic data to examine their relative contributions to diversity and aboveground biomass (e.g., Chiang et al. 2016; Ali et al. 2018b).
The difference in tree height observed among habitats in the studied southeastern area correlated with differences in basal area, whereas habitats with lower tree heights had higher basal areas, but the habitat with the tallest tree, i.e., the low valley, had the lowest basal areas. Our data support the results of a previous report that the conditions at high-elevation habitats are apparently well suited for slow-growing canopy tree species and have higher tree densities and basal areas per unit area, compared to low-elevation habitats (Punchi-Manage et al. 2013). Furthermore, there is a strong correlation between community structure attributes and abiotic factors (Cornwell and Ackerly 2010) as a trade-off to better withstand conditions of strong winds, nutrient-poor soils or low soil water availability (Poorter 2009). We also found a significant decrease in aboveground biomass among subplots in the studied southeastern area, from valleys to plateaus, probably due to the differences in tree height and basal area among habitats (e.g., Leuschner et al. 2007). Thus, the local topography is related to community structural attributes, with flatter habitats harboring larger trees with higher biomass and lower basal area than steeper areas. This fact may be correlated with the existence of a marked small-scale edaphic gradient since we detected significant differences in soil properties among habitats in the southeastern area.
Previous studies on forests have reported positive relationships between species richness and biomass (Paquette and Messier 2011; Poorter et al. 2017). However, our results show a nonsignificant relationship between species richness and aboveground biomass, which might be attributable to functional redundancy or functional dominance (Ali et al. 2019). The positive relationship between species richness and biomass reported in the aforementioned studies is consistent with the sampling, niche complementarity and facilitation effects (e.g., Poorter et al. 2015; Tilman 1999). On the other hand, the negative relationship between species richness and aboveground biomass indicates that communities having high aboveground biomass may exclude weak competitors (Ali et al. 2016), as predicted by competitive exclusion hypothesis (Grime 1973). Moreover, the direct and indirect effects of environmental factors such as soil and topographic factors could also explain such weak or negative relationship between species richness and aboveground biomass (Jucker et al. 2018; Ali et al. 2019).
In this study, we found nonsignificant relationship between species richness and aboveground biomass, which may be attributable to the species redundancy or niche overlap in the studied northeastern area, which means that despite an increase in species richness, biomass remains relatively constant without significant variation (Williams and Houseman 2014; Muledi et al. 2017). For instance, in the northeastern area, there were fewer stems and more species, probably due to the higher topographic heterogeneity, which increases species coexistence. In the southeastern area, on the other hand, the inverse situation was observed, with the presence of a higher number of stems and lower number of species, which could analogously be caused by the lower topographic heterogeneity (Ali et al. 2018a; Jucker et al. 2018). As such, a recent study has found a weak positive relationship between species diversity and aboveground biomass at larger spatial scales due to species redundancy (Poorter et al. 2015). Furthermore, other studies in tropical forests have reported that functionally dissimilar species may be playing equivalent roles in the ecosystem processes, e.g., biomass production (Lohbeck et al. 2016; Poorter et al. 2017). Presumably, the higher species richness in the northeastern area compensates for the aboveground biomass produced by the higher number of stems in the southeastern area. This result indicated that stocks of aboveground biomass were equivalent between the two studied areas, despite the northeastern area having 48% more species and 20% fewer stems than in the southeastern area.
Conclusions
This study reveals that higher variability of topographic factors determines habitat differentiation with changes in tree community composition, structure and aboveground biomass, but not in species richness. Our local-scale analyses indicate that in the less heterogeneous area there is a more uniform distribution pattern of elevation along with no variation in convexity across the gradients, which induces changes in community structural attributes. On the other hand, the more heterogeneous area shows no difference among habitats along the topographical gradients. We provide the first indication of the relative importance of topographic factors to community composition, structure and aboveground biomass along topographical gradients in a tropical Atlantic forest.
References
Ali A, Yan ER, Chen HYH, Chang SX, Zhao YT, Yang XD, Xu MS (2016) Stand structural diversity rather than species diversity enhances aboveground carbon storage in secondary subtropical forests in Eastern China. Biogeosciences 13:4627–4635
Ali A, Lin SL, He JK, Kong FM, Yu JH, Jiang HS (2018a) Climatic water availability is the main limiting factor of biotic attributes across large-scale elevational gradients in tropical forests. Sci Total Environ 647:1211–1221
Ali A, Lohbeck M, Yan E-R (2018b) Forest strata-dependent functional evenness explains whole-community aboveground biomass through opposing mechanisms. For Ecol Manag 424:439–447
Ali A, Lin SL, He JK, Kong FM, Yu JH, Jiang HS (2019) Elucidating space, climate, edaphic and biodiversity effects on aboveground biomass in tropical forests. Land Degrad Dev. https://doi.org/10.1002/ldr.3278
Anderson-Teixeira KJ, Wang MMH, Mcgarvey JC, LeBauer DS (2016) Carbon dynamics of mature and regrowth tropical forests derived from a pantropical database (TropForC-db). Glob Chang Biol 22:1690–1709
Apg IV (2016) An update of the Angiosperm Group classification for the orders and families of flowering plants: APG IV. Biol J Linnean Soc 141:399–436
Barton K (2017) ‘MuMIn’: multi-model inference. R package version 1.40.0. https://cran.r-project.org/web/packages/MuMIn/MuMIn.pdf. Accessed 15 Feb 2018
Bates D, Maechler M, Ben Bolker B, Walker S, Christensen RHB, Singmann H, Dai B, Scheipl F, Grothendieck G, Green P, Fox J (2019) ‘lme4’: linear mixed-effects models using ‘Eigen’ and S4. R package version 1.1-21. https://cran.r-project.org/web/packages/lme4/lme4.pdf. Accessed 31 May 2019
Brown C, Burslem DFRP, Illian JB, Bao L, Brockelman W, Cao M, Chang LW, Dattaraja HS, Davies S, Gunatilleke CVS, Gunatilleke IAUN, Huang J, Kassim AR, LaFrankie JV, Lian J, Lin L, Ma K, Mi X, Nathalang A, Noor S, Ong P, Sukumar R, Su SH, Sun IF, Suresh HS, Tan S, Thompson J, Uriarte M, Valencia R, Yap SL, Ye W, Law R (2013) Multispecies coexistence of trees in tropical forests: spatial signals of topographic niche differentiation increase with environmental heterogeneity. Proc R Soc Lond Ser B Biol sci 280:1764
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer, New York
Burnham KP, Anderson DR, Huyvaert KP (2011) AIC model selection and multimodel inference in behavioral ecology: some background, observations, and comparisons. Behav Ecol Sociobiol 65:23–35
Chao A, Gotelli NJ, Hsieh TC, Sander EL, Ma KH, Colwell RK, Ellison AM (2014) Rarefaction and extrapolation with Hill numbers: a framework for sampling and estimation in species diversity studies. Ecol Monogr 84:45–67
Chave J, Coomes D, Jansen S, Lewis SL, Swenson NG, Zanne AE (2009) Towards a worldwide wood economics spectrum. Ecol Lett 12:351–366
Chave J, Réjou-Méchain M, Búrquez A et al (2014) Improved allometric models to estimate the aboveground biomass of tropical trees. Glob Chang Biol 20:3177–3190
Chiang J-M, Spasojevic MJ, Muller-Landau HC, Sun I-F, Lin Y, Su S-H, Chen Z-S, Chen C-T, Swenson NG, McEwan RW (2016) Functional composition drives ecosystem function through multiple mechanisms in a broadleaved subtropical forest. Oecologia 182:829–840
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Aust J Ecol 18:117–143
Colwell RK, Chao A, Gotelli NJ, Lin SY, Mao CH, Chazdon RL, Longino JT (2012) Models and estimators linking individual-based and sample based rarefaction, extrapolation and comparison of assemblages. J Plant Ecol 5:3–21
Cornwell WK, Ackerly DD (2010) A link between plant traits and abundance: evidence from coastal California woody plants. J Ecol 98:814–821
Crawley MJ (2012) The R book, 2nd edn. Wiley, London
De’ath G (2002) Multivariate regression trees: a new technique for modeling species-environment relationships. Ecology 83:1105–1117
EMBRAPA (1997) Manual de métodos de análises de solo, 2nd edn. Centro Nacional de Pesquisa de Solos, Empresa Brasileira de Pesquisa Agropecuária, Rio de Janeiro
Grime JP (1973) Competitive exclusion in herbaceous vegetation. Nature 242:344–347
Guo Y, Wang B, Mallik AU, Huang F, Xiang W, Ding T, Wen S, Lu S, Li D, He Y, Li X (2017) Topographic species-habitat associations of tree species in a heterogeneous tropical forest. J Plant Ecol 57:1–10
Hsieh TC, Ma KH, Chao A. (2016) ‘iNEXT’: iNterpolation and EXTrapolation for species diversity. R package version 2.0.12.. https://cran.r-project.org/web/packages/iNEXT/iNEXT.pdf Accessed 10 Dec 2017
Husson F, Josse J, Le S, Mazet J (2018) “FactoMineR” package multivariate: exploratory data analysis and data mining. RStudio package version 1.0.14. https://cran.r-project.org/web/packages/FactoMineR/FactoMineR.pdf
John R, Dalling JW, Harms KE, Yavitt JB, Stallard RF, Mirabello M, Hubbell SP, Valencia R, Navarrete H, Vallejo M, Foster RB (2007) Soil nutrients influence spatial distributions of tropical tree species. Proc Natl Acad Sci USA 104:864–869
Jucker T, Bongalov B, Burslem DFRP, Nilus R, Dalponte M, Lewis SL, Phillips OL, Qie L, Coomes DA (2018) Topography shapes the structure, composition and function of tropical forest landscapes. Ecol Lett 21:989–1000
Kahmen H, Faig W (1988) Surveying. Walter Gruyter e Co, Berlin, p 578
Katabuchi M, Kurokawa H, Davies SJ, Tan S, Nakashizuka T (2012) Soil resource availability shapes community trait structure in a species-rich dipterocarp forest. J Ecol 100:643–665
Lan GY, Hu YH, Cao M, Zhu H (2011) Topography related spatial distribution of dominant tree species in a tropical seasonal rain forest in China. For Ecol Manag 262:1507–1513
Larsen DR, Speckman PL (2004) Multivariate regression trees for analysis of abundance data. Biometrics 60:543–549
Laurance WF (2009) Conserving the hottest of the hotspots. Biol Conserv 142:1137
Leuschner C, Moser G, Bertsch C, Röderstein M, Hertel D (2007) Large altitudinal increase in tree root/shoot ratio in tropical mountain forests of Ecuador. Basic Appl Ecol 8:219–230
Lewis SL, Edwards DP, Galbraith D (2015) Increasing human dominance of tropical forests. Science 349:827–832
Liu J, Yunhong T, Slik JWF (2014) Topography related habitat associations of tree species traits, composition and diversity in a Chinese tropical forest. For Ecol Manag 330:75–81
Lohbeck M, Bongers F, Martinez-Ramos M, Poorter L (2016) The importance of biodiversity and dominance for multiple ecosystem functions in a human-modified tropical landscape. Ecology 97:2772–2779
Magnago LFS, Magrach A, Laurance WF, Martins SV, Meira-Neto JA, Simonelli M, Edwards DP (2015) Would protecting tropical forest fragments provide carbon and biodiversity cobenefits under REDD+? Glob Chang Biol 21:3455–3468
Martinez-Arbizu P (2019) pairwiseAdonis: pairwise multilevel comparison using adonis. R package version 2.4-5. https://github.com/pmartinezarbizu/pairwiseAdonis
McEwan RW, Muller RN (2006) Spatial and temporal dynamics in canopy dominance of an old-growth mixed mesophytic forest. Can J For Res 36:1536–1550
Moeslund JE, Arge L, Bøcher PK, Dalgaard T, Svenning J-C (2013) Topography as a driver of local terrestrial vascular plant diversity patterns. Nord J Bot 31:129–144
Muledi JI, Bauman D, Drouet T, Vleminckx J, Jacobs A, Lejoly J, Meerts P, Shutcha MN (2017) Fine-scale habitats influence tree species assemblage in a Miombo forest. J Plant Ecol 10:958–969
Murray-Smith C, Brummitt NA, Oliveira-Filho AT, Bachman S, Moat J, Lughadha EM, Lucas EJ (2009) Plant diversity hotspots in the Atlantic Coastal Forests of Brazil. Conserv Biol 23:151–163
Nychka D, Furrer R, Paige J, Sain S (2019). ‘fields’ package: tools for spatial data. R package version 2.4-5. https://cran.r-project.org/web/packages/fields/fields.pdf. Accessed 10 Feb 2018
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner E (2018) ‘Vegan’: community ecology package. R package version 2.4-6. https://cran.r-project.org/web/packages/vegan/vegan.pdf. Accessed 5 Feb 2018
Paquette A, Messier C (2011) The effect of biodiversity on tree productivity: from temperate to boreal forests. Glob Ecol Biog 20:170–180
Paula A, Silva AF, Souza AL, Santos FAM (2002) Alterações florísticas ocorridas num período de quatorze anos na vegetação arbórea de uma floresta estacional semidecidual em Viçosa-MG. Rev Árvore 26:743–749
Pausas JG, Austin MP (2001) Patterns of plant species richness in relation to different environments: an appraisal. J Veg Sci 12:153–166
Pinheiro J, Bates D (2017) ‘nlme’: linear and nonlinear mixed effects models. R package version 3.1-131. https://cran.r-project.org/web/packages/nlme/nlme.pdf. Accessed 20 Jan 2018
Poorter L (2009) Leaf traits show different relationships with shade tolerance in moist versus dry tropical forests. New Phytol 181:890–900
Poorter L, van der Sande MT, Thompson J, Arets EJMM, Alarcón A, Álvarez-Sánchez J, Ascarrunz N, Balvanera P, Barajas-Guzmán G, Boit A, Bongers F, Carvalho FA, Casanoves F, Cornejo-Tenorio G, Costa FRC, de Castilho CV, Duivenvoorden JF, Dutrieux LP, Enquist BJ, Fernández-Méndez F, Finegan B, Gormley LHL, Healey JR, Hoosbeek MR, Ibarra-Manríquez G, Junqueira AB, Levis C, Licona JC, Lisboa LS, Magnusson WE, Martínez-Ramos M, Martínez-Yrizar A, Martorano LG, Maskell LC, Mazzei L, Meave JA, Mora F, Muñoz R, Nytch C, Pansonato MP, Parr TW, Paz H, Pérez-García EA, Rentería LY, Rodríguez-Velazquez J, Rozendaal DMA, Ruschel AR, Sakschewski B, Salgado-Negret B, Schietti J, Simões M, Sinclair FL, Souza PF, Souza FC, Stropp J, ter Steege H, Swenson NG, Thonicke K, Toledo M, Uriarte M, van der Hout P, Walker P, Zamora N, Peña-Claros M (2015) Diversity enhances carbon storage in tropical forests. Glob Ecol Biogeog 24:1314–1328
Poorter L, van der Sande MT, Arets EJMM, Ascarrunz N, Enquist BJ, Finegan B, Licona JC, Martínez-Ramos M, Mazzei L, Meave JA, Muñoz R, Nytch CJ, de Oliveira AA, Pérez-García EA, Prado-Junior J, Rodríguez-Velázques J, Ruschel AR, Salgado-Negret B, Schiavini I, Swenson NG, Tenorio EA, Thompson J, Toledo M, Uriarte M, van der Hout P, Zimmerman JK, Peña-Claros M (2017) Biodiversity and climate determine the functioning of Neotropical forests. Glob Ecol Biogeogr 26:1423–1434
Punchi-Manage R, Getzin S, Wiegand T, Kanagaraj R, Savitri Gunatilleke CV, Nimal Gunatilleke IA, Wiegand K, Huth A (2013) Effects of topography on structuring local species assemblages in a Sri Lankan mixed dipterocarp forest. J Ecol 101:149–160
R Core Team (2017) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed 15 Feb 2018
Reich PB (2014) The world-wide ‘fast-slow’ plant economics spectrum: a traits manifesto. J Ecol 102:275–301
Réjou-Méchain M, Tanguy A, Piponiot C, Chave J, Hérault B (2017) biomass: an r package for estimating above-ground biomass and its uncertainty in tropical forests. Methods Ecol Evol 8:1163–1167
Ribeiro MC, Martensen AC, Metzger JP, Tabarelli M, Scarano F, Fortin M-J (2011) The Brazilian Atlantic Forest: a shrinking biodiversity hotspot. In: Zachos F, Habel J (eds) Biodiversity hotspots. Springer, Berlin
Ripley B (2017) ‘MASS’: support functions and datasets for venables and RIPLEY’s MASS. R package version 7.3-48. https://cran.r-project.org/web/packages/MASS/MASS.pdf. Accessed 1 Feb 2018
Rodrigues AC, Villa PM, Viana NA (2019) Fine-scale topography shape richness, community composition, stem and biomass hyperdominant species in Brazilian Atlantic forest. Ecol Indic 102:208–217
Rozendaal DMA, Bongers F, Aide TM, Alvarez-Dávila E, Ascarrunz N, Balvanera P, Becknell JM, Bentos TV, Brancalion PHS, Cabral GAL, Calvo-Rodriguez S, Chave J, César RG, Chazdon RL, Condit R, Dallinga JS, de Almeida-Cortez JS, Jong B, Oliveira A, Denslow JS, Dent DH, DeWalt SJ, Dupuy JM, Durán SM, Dutrieux LP, Espírito-Santo MM, Fandino MC, Fernandes GW, Finegan B, García H, Gonzalez N, Moser VG, Hall JS, Hernández-Stefanoni JL, Hubbell S, Jakovac CC, Hernández AJ, Junqueira AB, Kennard D, Larpin D, Letcher SG, Licona J-C, Lebrija-Trejos E, Marín-Spiotta E, Martínez-Ramos M, Massoca PES, Meave JA, Mesquita RCG, Mora F, Müller SC, Muñoz R, de Oliveira Neto SN, Norden N, Nunes YRF, Ochoa-Gaona S, Ortiz-Malavassi E, Ostertag R, Peña-Claros M, Pérez-García EA, Piotto D, Powers JS, Aguilar-Cano J, Rodriguez-Buritica S, Rodríguez-Velázquez J, Romero-Romero MA, Ruíz Jorge, Sanchez-Azofeifa A, de Almeida AS, Silver WL, Schwartz NB, Thomas WW, Toledo M, Uriarte M, Sampaio EVS, van Breugel M, van der Wal H, Martins SV, Veloso MDM, Vester HFM, Vicentini A, Vieira ICG, Villa PM, Williamson GB, Zanini KJ, Zimmerman J, Poorter L (2019) Biodiversity recovery of Neotropical secondary forests. Sci Adv 5:3114
Scarano FR, Ceotto P (2015) Brazilian Atlantic forest: impact, vulnerability, and adaptation to climate change. Biodivers Conserv 24:2319
Therneau T, Atkinson B, Ripley B (2017) ‘rpart’: recursive partitioning and regression trees. R package version 4.1-11. https://CRAN.R-project.org/package=rpart. Accessed 4 March 2018
Tilman D (1999) The ecological consequences of changes in biodiversity: a search for general principles. Ecology 80:1455–1474
Toledo M, Peña-Claros M, Bongers F, Alarcón A, Balcázar J, Chuviña J, Leaño C, Licona JC, Poorter L (2012) Distribution patterns of tropical woody species in response to climatic and edaphic gradients. J Ecol 100:253–263
Villa PM, Martins SV, Oliveira Neto SN, Rodrigues AC, Martorano L, Delgado L, Cancio NM, Gastauerg M (2018) Intensification of shifting cultivation reduces forest resilience in the northern Amazon. For Ecol Manag 430:312–320
Wang Q, Punchi-Manage R, Lu Z, Franklin SB, Wang Z, Li Y, Chi X, Bao D, Guo Y, Xu JLY, Qiao X, Jiang M (2016) Effects of topography on structuring species assemblages in a subtropical forest. J Plant Ecol 10:440–449
Wickham H, François R, Henry L, Müller K (2018). ‘dplyr’ package: a grammar of data manipulation. R package version 7.3-48. https://cran.r-project.org/web/packages/dplyr/dplyr.pdf. Accessed 15 Feb 2018
Williams BM, Houseman GR (2014) Experimental evidence that soil heterogeneity enhances plant diversity during community assembly. J Plant Ecol 7:461–469
Zanne AE, Lopez-Gonzalez G, Coomes DA, Ilic J, Jansen S, Lewis SL, Miller RB, Swenson NG, Wiemann MC, Chave J (2009) Data from: towards a worldwide wood economics spectrum. Dryad digital repository. https://doi.org/10.5061/dryad.234. Accessed 3 Dec 2017
Acknowledgements
We acknowledge the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for concession the scholarship of the first and, PDJ to the second author. We also thank Dr. Ferry Slik for his valuable comments on the early version of this article. AA is financially supported by China Postdoctoral Science Foundation (Grant No. 2018M643117) for ecological research at South China Normal University. We acknowledge the support of Northeast Forestry University for this publication.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: This research received no external funding.
The online version is available at http://www.springerlink.com.
Corresponding editor: Chai Ruihai.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Rodrigues, A.C., Villa, P.M., Ali, A. et al. Fine-scale habitat differentiation shapes the composition, structure and aboveground biomass but not species richness of a tropical Atlantic forest. J. For. Res. 31, 1599–1611 (2020). https://doi.org/10.1007/s11676-019-00994-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-019-00994-x