Abstract
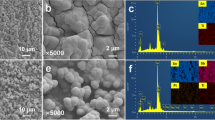
SnO2-Sb and α-PbO2 have been successively deposited onto the surface of a titanium substrate, followed by fabrication of β-PbO2 doped with Fe element and polytetrafluoroethylene (PTFE) thereon. Due to the collaborative contribution of α-PbO2 and PTFE, the stability of the Ti/SnO2-Sb/α-PbO2/Fe-β-PbO2-PTFE electrode was found to be significantly improved. The oxygen evolution overpotential of the electrode was measured to be 1.95 V versus saturated calomel electrode (SCE). Also, the contact angle of the optimized superhydrophobic electrode reached 156.8°. The optimized superhydrophobic electrode modified with PTFE exhibited lower charge-transfer resistance and good oxidative ability towards organics. The electrocatalytic activity of the devised electrodes was studied using methyl orange degradation. Factors affecting the decolorization of methyl orange were optimized. After 30 min of electrolysis, a maximum removal efficiency of 83% was achieved at a current density of 30 mA cm−2 for an initial methyl orange concentration of 40 mg L−1 at pH 5. The results confirmed that the decolorization followed a first-order kinetics model. The color of the methyl orange solution changed from orange to colorless upon completion of the degradation reaction. Such superhydrophobic Ti/SnO2-Sb/α-PbO2/Fe-β-PbO2-PTFE electrodes could effectively degrade organic pollutants under low voltages, which is of great significance for reducing energy consumption.
Similar content being viewed by others
References
E. Tsantaki, T. Velegraki, A. Katsaounis, and D. Mantzavinos, J. Hazard. Mater. 207, 91 (2012).
M. Zhou and J. He, J. Hazard. Mater. 153, 357 (2008).
C.A. Martínez-Huitle, M.A. Rodrigo, I. Sirés, and O. Scialdone, Chem. Rev. 115, 13362 (2015).
C.A. Martinez-Huitle and E. Brillas, Appl. Catal. B-Environ. 87, 105 (2009).
E. Brillas and C.A. Martínez-Huitle, Appl. Catal. B-Environ. 166, 603 (2015).
H. Lin, J. Niu, J. Xu, Y. Li, and Y. Pan, Electrochim. Acta 97, 167 (2013).
Á. Anglada, A. Urtiaga, and I. Ortiz, J. Chem. Technol. Biotechnol. 84, 1747 (2009).
Z. Xu, H. Liu, J. Niu, Y. Zhou, C. Wang, and Y. Wang, J. Hazard. Mater. 327, 144 (2017).
P. Duan, X. Yang, G. Huang, J. Wei, Z. Sun, and X. Hu, Colloids Surf. A 569, 119 (2019).
X. Duan, C. Zhao, W. Liu, X. Zhao, and L. Chang, Electrochim. Acta 240, 424 (2017).
X. Li, H. Xu, and W. Yan, Appl. Surf. Sci. 389, 278 (2016).
M. Xu, Z. Wang, F. Wang, P. Hong, C. Wang, X. Ouyang, C. Zhu, Y. Wei, Y. Hun, and W. Fang, Electrochim. Acta 201, 240 (2016).
Z. Xu, Y. Yu, H. Liu, and J. Niu, Sci. Total Environ. 579, 1600 (2017).
Q. Zhang, X. Guo, X. Cao, D. Wang, and J. Wei, Chin. J. Catal. 36, 975 (2015).
X. Li, D. Pletcher, and F.C. Walsh, Chem. Soc. Rev. 40, 3879 (2011).
F. Fu, W. Yang, and C. Ke, Mater. Chem. Phys. 220, 155 (2018).
S. Abaci, U. Tamer, K. Pekmez, and A. Yildiz, Electrochim. Acta 50, 3655 (2005).
J.M. Aquino, R.C. Rocha-Filho, L.A.M. Ruotolo, N. Bocchi, and S.R. Biaggio, Chem. Eng. J. 251, 138 (2014).
J. Wu, H. Xu, and W. Yan, RSC Adv. 5, 19284 (2015).
J. Niu, H. Lin, J. Xu, H. Wu, and Y. Li, Environ. Sci. Technol. 46, 10191 (2012).
R. Inguanta, E. Rinaldo, S. Piazza, and C. Sunseri, J. Solid State Electrochem. 16, 3939 (2012).
A. Moncada, M.C. Mistretta, S. Randazzo, S. Piazza, C. Sunseri, and R. Inguanta, J. Power Sources 256, 72 (2014).
P.N. Bartlett, T. Dunford, and M.A. Ghanem, J. Mater. Chem. 12, 3130 (2002).
Z. Wang, Y. Mao, M. Xu, Y. Wei, Y. Hu, C. Zhu, W. Fang, and F. Wang, J. Electrochem. Soc. 164, H981 (2017).
T. Chen, X. Li, C. Qiu, W. Zhu, H. Ma, S. Chen, and O. Meng, Biosens. Bioelectron. 53, 200 (2014).
M. Panizza and G. Cerisola, Chem. Rev. 109, 6541 (2009).
J.P. Carr and N.A. Hampson, Chem. Rev. 72, 679 (1972).
G. Zhao, Y. Zhang, Y. Lei, B. Lv, J. Gao, Y. Zhang, and D. Li, Environ. Sci. Technol. 44, 1754 (2010).
R. Xie, X. Meng, P. Sun, J. Niu, W. Jiang, L. Bottomley, D. Li, Y. Chen, and J. Crittenden, Appl. Catal. B-Environ. 203, 515 (2017).
Y. Jin, F. Wang, M. Xu, Y. Hun, W. Fang, Y. Wei, and C. Zhu, J. Taiwan Inst. Chem. E 51, 135 (2015).
C. Yang, Y. Wang, B. Hu, H. Zhang, Y. Lv, and X. Zhou, J. Electron. Mater. 47, 5965 (2018).
T. Darmanin, E.T. de Givenchy, S. Amigoni, and F. Guittard, Adv. Mater. 25, 1378 (2013).
E. Vazirinasab, R. Jafari, and G. Momen, Surf. Coat. Technol. 341, 40 (2018).
G. He, S. Lu, W. Xu, P. Ye, G. Liu, H. Wang, and T. Dai, J. Alloys Compd. 747, 772 (2018).
X. Xing, J. Ni, X. Zhu, Y. Jiang, and J. Xia, Chemosphere 205, 361 (2018).
Y. Jiang, Z. Hu, M. Zhou, L. Zhou, and B. Xi, Sep. Purif. Technol. 128, 67 (2014).
R. Kotz, S. Stucki, and B. Carcer, J. Appl. Electrochem. 21, 14 (1991).
Z. Wang, M. Xu, F. Wang, X. Liang, Y. Wei, Y. Hu, C.G. Zhu, and W. Fang, Electrochim. Acta 247, 535 (2017).
Acknowledgments
This research was supported by the Natural Science Foundation of China (No. 21676146) and the Financial Foundation of State Key Laboratory of Materials-Oriented Chemical Engineering.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jin, Y., Lv, Y., Yang, C. et al. Fabrication of Superhydrophobic Ti/SnO2-Sb/α-PbO2/Fe-β-PbO2-PTFE Electrode and Application in Wastewater Treatment. J. Electron. Mater. 49, 2411–2418 (2020). https://doi.org/10.1007/s11664-019-07936-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-019-07936-7