Abstract
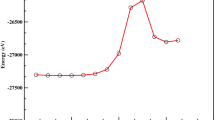
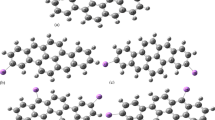
The influence of lithiation process on the HOMO–LUMO gap and the first hyperpolarizability values of corannulene (C20H10) and quadrannulene (C16H8) buckybowls are investigated using density functional theory (DFT) calculations. Lithiation is performed at the bridging position of the C–C bond of benzene rings of quadrannulene and corannulene. The HOMO–LUMO gap of lithiated buckybowls is reduced with respect to pristine ones. The results indicate that the lithiation process causes the remarkable enhancement of the first hyperpolarizability. The time-dependent density functional theory (TD-DFT) calculations are also performed to understand how lithiation affects the first hyperpolarizability values. The present results might be valuable for further theoretical and experimental studies on the electronic and nonlinear optical properties of buckybowl compounds.
Similar content being viewed by others
References
T. Bauert, L. Zoppi, G. Koller, J.S. Siegel, K.K. Baldridge, and K.H. Ernst, J. Am. Chem. Soc. 135, 12857 (2013).
Y.T. Wu and J.S. Siegel, Chem. Rev. 106, 4843 (2006).
P.W. Rabideau and A. Sygula, Acc. Chem. Res. 29, 235 (1996).
L. Wang, W.Y. Wang, Y.Q. Qiu, and H.Z. Lu, J. Phys. Chem. C 119, 24965 (2015).
L. Wang, W.Y. Wang, X.Y. Fang, C.L. Zhu, and Y.Q. Qiu, RSC Adv. 5, 79783 (2015).
V.M. Tsefrikas and L.T. Scott, Chem. Rev. 106, 4868 (2006).
X. Li, F. Kang, and M. Inagaki, Small 12, 3206 (2016).
R. Bharat, T. Bhola, A. Bally, M.K. Valente, Ł. Cyrański, S.M. Dobrzycki, S.M. Spain, P. RempaŁa, P.R. Chin, and B.T. King, Angew. Chem. Int. Ed. 122, 409 (2010).
S. Muhammad, H.L. Xu, R.L. Zhong, Z.M. Su, A.G. Al-Sehemi, and A. Irfan, J. Mater. Chem. C 1, 5439 (2013).
F. Ma, Z.R. Li, Z.J. Zhou, D. Wu, Y. Li, Y.F. Wang, and Z.S. Li, J. Phys. Chem. C 114, 11242 (2010).
E. Tahmasebi, E. Shakerzadeh, and Z. Biglari, Appl. Surf. Sci. 363, 197 (2016).
R.L. Zhong, H.L. Xu, S. Muhammad, J. Zhang, and Z.M. Su, J. Mater. Chem. 22, 2196 (2012).
M. Solimannejad, S. Kamalinahad, and E. Shakerzadeh, J. Electron. Mater. 46, 4420 (2017).
D.S. Chemla, Nonlinear Optical Properties of Organic Molecules and Crystals, 1st edn. (Academic Press, NewYork, 1987).
H.S. Nalwa, Nonlinear Optics of Organic Molecules and Polymers (Boca Raton: CRC Press, 1997).
S.R. Marder, B. Kippelen, A.K.Y. Jen, and N. Peyghambarian, Nature 388, 845 (2007).
A. Streitwieser, S.M. Bachrach, A. Dorigo, and P. von Rague Schleyer, Lithium Chemistry: A Theoretical and Experimental Overview (New York: Wiley, 1995).
H.L. Xu, R.L. Zhong, S.L. Sun, and Z.M. Su, J. Phys. Chem. C 115, 16340 (2011).
C.C. Zhang, H.L. Xu, Y.Y. Hu, S.L. Sun, and Z.M. Su, J. Phys. Chem. A 115, 2035 (2011).
H. Chen, W.Y. Wang, L. Wang, C.L. Zhu, X.Y. Fang, and Y.Q. Qiu, J. Mol. Gr. Modell. 64, 139 (2016).
W.Q. Li, X. Zhou, Y. Changa, J.K. Feng, W.Q. Tian, X.D. Sun, and B. Shao, Chem. Phys. Lett. 588, 131 (2013).
W.Q. Li, X. Zhou, Y. Chang, W.Q. Tian, and X.D. Sun, Appl. Phys. Lett. 102, 153307 (2013).
F. Kazemimoghadam and E. Shakerzadeh, Chem. Phys. Lett. 678, 51 (2017).
A.D. Buckingham, Adv. Chem. Phys. 12, 107 (1967).
A.D. Mclean and M. Yoshimine, J. Chem. Phys. 47, 1927 (1967).
R.L. Zhong, H.L. Xu, Z.R. Li, and Z.M. Su, J. Phys. Chem. Lett. 6, 612 (2015).
M. de Wergifosse and B. Champagne, J. Chem. Phys. 134, 074113 (2011).
B. Champagne, E.A. Perpte, D. Jacquenmin, S.J.A. van Gisbergen, E.J. Baerends, C. Soubra-Ghaoui, and K.A. Robins, J. Phys. Chem. A 104, 4755 (2000).
T. Yanai, D.P. Tew, and N.C. Handy, Chem. Phys. Lett. 393, 51 (2004).
A. Alparone, Chem. Phys. Lett. 536, 88 (2013).
M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R.Cheeseman, V.G. Zakrzewski, J.A. Montgomery, Jr., R.E. Stratmann, J.C. Burant,S. Dapprich, J.M. Millam, A.D. Daniels, K.N. Kudin, M.C. Strain, O. Farkas, J.Tomasi, V. Barone, M. Cossi, R. Cammi, B. Mennucci, C. Pomelli, C. Adamo, S.Clifford, J. Ochterski, G.A. Petersson, P.Y. Ayala, Q. Cui, K. Morokuma, D.K.Malick, A.D. Rabuck, K. Raghavachari, J.B. Foresman, J. Cioslowski, J.V. Ortiz,B.B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. Gomperts, R.L.Martin, D.J. Fox, T. Keith, M. A. Al-Laham, C.Y. Peng, A. Nanayakkara, C.Gonzalez, M. Challacombe, P.M.W. Gill, B.G. Johnson, W. Chen, M.W. Wong,J.L. Andres, M. Head-Gordon, E.S. Replogle, and J.A. Pople, Gaussian 09 Inc., Pittsburgh.
V.I. Minkin, R.M. Minyaev, A.G. Starikov, and T.N. Gribanova, Russ. J. Org. Chem. 41, 1289 (2005).
B.J. Smith, Chem. Phys. Lett. 207, 403 (1993).
Y.M. Xie and H.F. Schaefer, Chem. Phys. Lett. 179, 563 (1991).
A. Streitwieser, S.M. Bachrach, A. Dorigo, and P.V.R. Schleyer, Bonding, Structures and Energies in Organolithium Compounds (New York: Wiley, 1995), p. 1.
K. Mierzwicki, S. Berski, and Z. Latajka, Chem. Phys. Lett. 331, 538 (2000).
F.M. Bickelhaupt, N.J.R. van Eikema Hommes, C.F. Guerra, and E.J. Baerends, Organometallics 15, 2923 (1996).
S.G. Raptis, M.G. Papadopoulos, and A.J. Sadlej, Phys. Chem. Chem. Phys. 2, 3393 (2000).
J.L. Oudar and D.S. Chemla, J. Chem. Phys. 66, 2664 (1977).
J.L. Oudar, J. Chem. Phys. 67, 446 (1977).
D.R. Kanis, M.A. Ratner, and T.J. Marks, Chem. Rev. 94, 195 (1994).
D. Ayan and K.P. Swapan, Chem. Soc. Rev. 35, 1305 (2006).
W.Q. Li, X. Zhou, W.Q. Tian, and X.D. Sun, Phys. Chem. Chem. Phys. 15, 1810 (2013).
H. Christoph, J. Grunenberg, H. Hopf, I. Dix, P.G. Jones, M. Scholtissek, and G. Maier, Chem. Eur. J. 14, 5604 (2008).
Acknowledgements
Funding was provided by Shahid Chamran University of Ahvaz (Grant No. 1395).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shakerzadeh, E., Kazemimoghadam, F. & Anota, E.C. How Does Lithiation Affect Electro-Optical Features of Corannulene (C20H10) and Quadrannulene (C16H8) Buckybowls?. J. Electron. Mater. 47, 2348–2358 (2018). https://doi.org/10.1007/s11664-018-6069-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-018-6069-0