Abstract
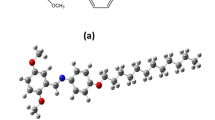
Herein, we demonstrate effect of substituents on optoelectronic properties of discotic liquid crystals (DLCs) by using density functional theory (DFT) calculations at B3LYP/Lanl2Z level of theory. Three parent DLCs, namely, (1) benzene-1,3,5-triyl tris(3,5-dialkoxybenzoate), (2) N1, N3, N5-tris(3-alkoxyphenyl)benzene-1,3,5-tricarboxamide, and (3) trialkyl 4, 4′, 4″-(benzenetricarbonyltris (azanediyl)) tribenzoate benzoate and their –N and –S group derivatives of 1, 2, and 3, were investigated to observe the change in optoelectronic response of these systems. The frontier molecular orbital studies and electron affinity values indicate that the studied compounds are stable against the oxygen and moisture present in air. The calculated charge transfer integrals, electron, and hole mobility values revealed that parent DLCs and their derivatives can be employed as an effective n-type material for OLEDs; however, derivatives have enhanced charge transfer values compared with their parents. For better understanding of the thermochemistry and effect of substituents, frequency calculations were carried out. P1-D4 derivative having R = -NH-CO-CH3 terminal group came out to be theoretically the most favored having the lowest ΔG value. Computed UV/visible spectroscopic analysis showed minimum absorbance and maximum transmittance for derivative P2-D1 having –S-NH2 substituent. Molecular electrostatic potential surfaces mapped at potential range, i.e., − 8.531e-3esu to + 8.531e-3esu, describe electrophilic and nucleophilic characteristics. Introduction of electron donor groups enhanced electrical conductivity, excitation energy, and charge transfer integral, thus increasing optoelectronic properties of DLCs. However, these claims require further experimental verification.





Similar content being viewed by others
References
Tant J, Geerts YH, De Lehmann CV, Zucchi G, Laursen BW, Bjornholm T, Lemaur V, Marcq V, Burquel A, Hennebicq E, Gardebien F, Viville P, Beljonne D, Lazzaroni R, Cornil J (2005) Liquid crystalline metal-free phthalocyanines designed for charge and exciton transport. J. Phys. Chem. B 10:20315–20323
Laschat S, Baro A, Steinke N (2007) Discotic liquid crystals:from tailor made synthesis to plastic electronics. Angew. Chem. 46:4832–4887. https://doi.org/10.1002/anie.200604203
Wohrle T, Wurzbach I, Kirres J (2016) Discotic liquid crystals. Chem. Rev. 116:1139–1241. https://doi.org/10.1021/acs.chemrev.5b00190
Mikolajczyk MM, Zaleśny R, Czyżnikowska Z, Toman P, Leszczynski J, Bartkowiak W (2011) Long-range corrected DFT calculations of charge-transfer integrals in model metalfree phthalocyanine complexes. J. Mol. Model. 17:2143–2214. https://doi.org/10.1007/s00894-010-0865-7
Sista P, Nguyen H, Murphy JW, Hao J, Dei DK, Palaniappan K, Servello J, Kularatne RS, Gnade BE, Xue BF (2010) Synthesis and electronic properties of semiconducting polymers containing benzodithiophene with alkyl phenylethynyl substituents. Macromolecules 43:8063–8070 https://www.cheric.org/research/tech/periodicals/doi.php?art_seq=891095
Song Y, Wang Q, Ni Q (2020) Designing a novel material with considerable nonlinear optical responses based on the bicorannulenyl molecule. J. Mol. Model. 26:201. https://doi.org/10.1007/s00894-020-04450-0
Liu B, Giordano F, Pei K, Decoppet JD, Zhu WH, Zakeeruddin SM, Gratzel M (2015) Molecular engineering of pyrido[3,4-b]pyrazine-based donor-acceptor-π-acceptor organic sensitizers: effect of auxiliary acceptor in cobalt- and iodine-based electrolytes. Chem Eur J 21:18654–18661. https://doi.org/10.1002/chem.201503514
Pei K, Wu YZ, Wu WJ, Zhang Q, Chen BQ, Tian H, Zhu WH (2012) Constructing organic D-A-π-A-featured sensitizers with a quinoxaline unit for high-efficiency solar cells: the effect of an auxiliary acceptor on the absorption and the energy level alignment. Chem Eur J 18:8190–8200. https://doi.org/10.1002/chem.201103542
Liu YX, Wan M, Zhao JS, Cui CS, Liu JF (2014) Effects of alkyl or alkoxy side chains on the electrochromic properties of four ambipolar donor-acceptor type polymers. RSC Adv. 4:52712–52726. https://doi.org/10.1039/C4RA08664C
Lee PL, Hsu HLC, Lee JF, Chuang HY, Lin PY (2011) New conjugated copolymers based on benzo[1,2-b; 3,4-b’]dithiophene and derivatives of benzo[g]quinoxaline for bulk heterojunction solar cells. J Polym Sci Part A Pol Chem 49:662–670. https://doi.org/10.3390/ma11102063
Ying WJ, Yang JB, Wielopolski M, Moehl T, Moser JE, Comte P, Hua JL, Zakeeruddin SM, Tian H, Grazel M (2014) New pyrido[3,4-b]pyrazine-based sensitizers for efficient and stable dye-sensitized solar cells. Chem. Sci. 5:206–214. https://doi.org/10.1039/C3SC51844B
Lu XF, Feng QY, Lan T, Zhou G, Wang ZS (2012) Molecular engineering of quinoxaline-based organic sensitizers for highly efficient and stable dye-sensitized solar cells. Chem. Mater. 24:3179–3187. https://doi.org/10.1021/cm301520z
Lu XF, Fan SH, Wu JH, Jia XW, Wang ZS, Zhou G (2014) Controlling the charge transfer in D-A-D chromophores based on pyrazine derivatives. J Org Chem 79:6480–6489. https://doi.org/10.1021/jo500856k
Chang DW, Lee HJ, Kim JH, Park SY, Park SM, Dai L, Baek JB (2011) Novel quinoxaline-based organic sensitizers for dye-sensitized solar cells. Org. Lett. 13:3880–3883. https://doi.org/10.1021/ol2012378
Kong LQ, Wang M, Ju XP, Zhao JS, Zhang Y, Xie Y (2017) The availability of neutral cyan, green, blue and purple colors from simple D–A type polymers with commercially available thiophene derivatives as the donor units. Polymers 9:656. https://doi.org/10.3390/polym9120656
M.J.G. 09 R.D. 0. Frisch, Trucks GW, Schlegel HB, Scuseria GE, 2009. Robb MA, CJ R., S. G., B. V., M. B., P. G. A., N. H., C. M., L. X., H. H. P., I. A. F., B. J., Z. G., S. J. L., H. M., E. M., T. K., F. R., Hasegaw, Gaussian 09, Revision D.01, (2009)
Boukhvalov DW, Katsnelson MI, Lichtenstein AI (2008) Hydrogen on graphene: electronic structure, total energy, structural distortions and magnetism from first-principles calculations. Phys. Rev. B 77:035427. https://doi.org/10.1103/PhysRevB.77.03542
Hlel A, Mabrouk A, Chemek M, Ben Khalifa I, Alimi K (2015) A DFT study of charge-transfer and opto-electronic properties of some new materials involving carbazole units. Computational Condensed Matter 3:30–40 https://www.researchgate.net/deref/http/3A/2F/2Fdx.doi.org/2F10.1016/2Fj.cocom.2015.02.001
S. Li, Semiconductor physical electronics, 2nd ed. (Springer, Berlin, 2006
Manimekalai A, Vijayalakshmi N (2015) Computational and spectral studies of 6-phenylazo-3-(ptolyl)-2H-chromen-2-one. Spectrochim Acta Part A Mol Biomol Spectros 136:388–396. https://doi.org/10.1016/j.saa.2014.09.045
Cheng YC, Silbey RJ, da Siva Filho DA, Calbert JP, Cornil J, Bre’das JL (2003) Three-dimensional band structure and bandlike mobility in oligoacene single crystals: a theoretical investigation. J. Chem. Phys. 118:3764. https://doi.org/10.1063/1.1539090
Lin BC, Cheng CP, Lao ZP (2003) Reorganization energies in the transports of holes and electrons in organic amines in organic electroluminescence studied by density functional theory. J. Phys. Chem. A 107:5241–5251. https://doi.org/10.1021/jp0304529
Liu CC, Mao SW, Kuo MY (2010) Cyanated pentaceno[2,3-c]chalcogenophenes for potential application in air-stable ambipolar organic thin-film transistors. J. Phys. Chem. C 114:22316–22321. https://doi.org/10.1021/jp1099464
Newman CR, Frisbie CD, da Silva Filho DA, Brédas JL, Ewbank PC, Mann KR (2004) Introduction to organic thin film transistors and design of n-channel organic semiconductors. Chem. Mater. 16:4436–4451. https://doi.org/10.1021/cm049391x
Chang YC, Kuo MY, Chen CP, Lu HF, Chao I (2010) On the air stability of n-channel organic field-effect transistors: a theoretical study of adiabatic electron affinities of organic semiconductors. J. Phys. Chem. C 114:11595–11061. https://doi.org/10.1021/jp1025625
Hu Y, Yin J, Chaitanya K, Xue-Hai J (2016) Theoretical investigation on charge transfer properties of 1,3,5-tripyrrolebenzene (TPB) and its derivatives with electron-withdrawing substituents. Croat Chem Acta 89:81–90. https://doi.org/10.5562/cca2787
Perrot, Pierre (1998). A to Z of thermodynamics. Oxford University Press.
W. Demtröder (2003). Laser spectroscopy: basic concepts and instrumentation. Springer. p. 31.
Acknowledgments
Authors acknowledge Research Center for Modeling and Simulations for providing supercomputer facility for DFT studies.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nosheen, B., Perveen, F., Ashraf, Z. et al. Charge transfer and opto-electronic properties of some newly designed polycatenar discotic liquid crystal derivatives: a DFT study. J Mol Model 26, 291 (2020). https://doi.org/10.1007/s00894-020-04550-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-020-04550-x