Abstract
Introduction
Tumor-induced osteomalacia (TIO) is an uncommon paraneoplastic syndrome due to the overproduction of fibroblast growth factor 23 (FGF23). It is predominantly caused by mesenchymal tumors and cured upon their complete removal. Non-surgical treatment is an alternative option but limited to specific clinical conditions.
Methods
We report a challenging case of TIO caused by a tumor involving the occipital bone. We also performed a literature review of TIO caused by tumors localized at this site, focusing on clinical findings, treatment, and outcomes.
Results
The patient, a 62-year-old male, presented with a long-lasting history of progressive weakness. Biochemical evaluation revealed severe hypophosphatemia due to low renal tubular reabsorption of phosphate with raised intact FGF23 values. A 68 Ga-DOTATATE PET/TC imaging showed a suspicious lesion located in the left occipital bone that MRI and selective venous catheterization confirmed to be the cause of TIO. Stereotactic gamma knife radiosurgery was carried out, but unfortunately, the patient died of acute respiratory failure.
To date, only seven additional cases of TIO have been associated to tumors located in the occipital bone. Furthermore, the tumor involved the left side of the occipital bone in all these patients.
Conclusion
The occipital region is a difficult area to access so a multidisciplinary approach for their treatment is required. If anatomical differences could be the basis for the predilection of the left side of the occipital bone, it remains to be clarified.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tumor-induced osteomalacia (TIO) is an uncommon paraneoplastic syndrome predominantly due to the overproduction of fibroblast growth factor 23 (FGF23) by mesenchymal tumors usually involving bone and soft tissues [1]. An increased number of patients with TIO have been recently reported, possibly reflecting a raised awareness of the disease. A step-by-step clinical approach is fundamental to diagnose TIO and localize the culprit lesion [1, 2]. Non-surgical treatment is an alternative option but is limited to specific clinical conditions, such as inability to locate the tumor, tumor located in sites difficult to be treated surgically, contraindications to surgery, and incomplete previous tumor resection [1, 3,4,5].
We report a challenging case of TIO located in the occipital bone. Moreover, we also performed a literature review of TIO located in this specific bone focusing on clinical findings, treatment, and outcomes.
Case presentation
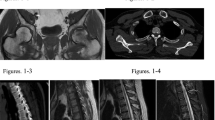
A 62-year-old male presented with an 8 years long-lasting history of weakness. He referred to both hip fractures. Biochemical evaluation at admission showed hypophosphatemia due to low renal tubular reabsorption of phosphate (TmP/GFR: 1.70 mg/dL) and increased serum intact FGF23 value (iFGF23) (190 pg/mL, n.v.: < 95 pg/mL, DiaSorin kit, Stillwater, MN, USA) suggestive of TIO. A 99mTc-labeled octreotide HYNIC-TOC scintigraphy showed increased uptake in the proximal left tibia suggesting it was the culprit site lesion. Even though the targeted MRI was negative, surgery was performed, but the tumor was not histologically detected. In addition, intra-operative assay of serum FGF23 did not show any significant decrease. Then, we performed a 68Ga-DOTATE PET/CT, through which a suspicious lesion was detected in the left occipital bone (Fig. 1a). It was also confirmed by brain MRI (Fig. 1b). Venous blood sampling (VBS) for measuring serum iFGF23 on both sides of the brain showed a slight gradient in respect to the contralateral site, suggesting that this was the site of FGF23 overproduction (Fig. 1c). Surgery was refused by the patient (mainly because of the increased risk of mortality, possible irreversible neurological consequences), and stereotactic gamma knife radiosurgery (SGKRS) was carried out. The patient died a few days later owing to acute respiratory failure. Autopsy was not performed.
Literature review
We searched for all original and review articles in MEDLINE till December 2022. Individual search was carried out using the following terms: “oncogenic osteomalacia,” “tumor induced osteomalacia,” “FGF23,” “skull base,” and “occipital bone.” Syndromic conditions associated with high FGF23 (e.g., fibrous dysplasia/McCune-Albright syndrome) were excluded. Only articles in English were considered. We were able to identify only other seven patients with TIO caused by a tumor located in the occipital bone [6,7,8,9,10,11,12].
A clinical synopsis of these patients and of the patient reported here is shown in Table 1. Four patients were males and four were females. Average age was 52.75 ± 5.06 years. Putative duration of the disease was 3.0 ± 2.13 years. All patients complained of at least one typical symptom of chronic hypophosphatemia (i.e., bone pain, weakness, fractures). Mean value of serum phosphate at admission was 1.76 ± 0.26 mg/dL. TmP/GFR values were not available for all patients. Basal circulating FGF23 levels were reported in 6 out of 8 patients. C-terminal assay was used in all cases [8,9,10,11,12] except that reported here in which iFGF23 was measured. As in our case, the tumor was detected in other six patients by functional imaging [7,8,9,10,11,12]. The suspicious lesion was confirmed by MRI in our case and in other five patients [7,8,9,10] and by CT scan in another one [11]. Interestingly, the tumor involved invariably the left side of the occipital bone. In five patients [6, 8,9,10, 12], the clivus was also involved. In one patient, data on treatment and follow-up were not available [12]. All other previously reported cases were initially treated by surgery. In one of them [9], surgery was performed after angioembolization. Histologically, all excised tumors were consistent with the phosphaturic mesenchymal tumor (PMT) [1]. In three patients [9,10,11], a postoperative drop of serum FGF23 was documented after surgery. The complete surgical removal of the tumor was obtained in three patients [7, 9, 10], with at least 3 months of follow-up for the two of them. Surgery was unsuccessful in one case [8]. After 4 months of follow-up, a second surgery was performed but again ineffectively. An alternative medical strategy was implemented with phosphate supplements and, for the first time in TIO, with peptide receptor radionuclide therapy (PRRT) 177Lu-DOTATATE. Three months after the first cycle, the authors reported improvement of symptoms and lesion’s standard uptake values (SUVs) reduction at 68Ga DOTATATE PET/CT. Disease persistence after two surgeries was reported for another case [11]. Then, PRRT177Lu-DOTATATE was employed leading to a stable disease after two cycles. Recurrence after 2 years since surgery was reported in one case [6]. The patient was treated with SGKRS followed by subcutaneous octreotide without effect on serum phosphate and FGF23.
Discussion
The patient reported here is the eighth case of TIO caused by a tumor located in the occipital bone. All patients shared clinical and laboratory findings consistent with TIO caused by tumors located elsewhere and a long delay before final diagnosis, a common finding of patients with TIO [1].
In all surgically treated patients [6,7,8,9,10], the histological diagnosis was consistent with PMT, the most common tumor associated with TIO [1]. It is likely that even in the case reported by Luthra and colleagues [12] and in our case this would have been the diagnosis if the tumor had been excised. Indeed, through 68Ga-DOTATE PET/CT, a suspicious area was detected in the left occipital bone. Whole-body 68Ga-DOTATATE PET/CT has been reported to have an accuracy for localizing tumors causing TIO greater than that of octreo-SPECT-CT for the higher affinity of 68Ga-DOTATATE for somatostatin receptors (in particular type 2) compared to octreotide [1, 13]. However, the expression of somatostatin receptors is not specific for TIO-causing tumors [1, 13]. Indeed, in our case, a histologically proven false-positive increased radiotracer uptake for tumor-causing TIO was detected in the proximal left tibia, likely as a result of osteomalacia-related intramedullary fracture. Furthermore, we were able to confirm tumor localization by VBS (Fig. 1c). Even though a significant threshold to secure the diagnosis has not been established, the detected gradient supported the anatomical origin of FGF23 from the left side. In the patient reported by Hana et al. in which the tumor was located in the anterior skull base, the FGF23 gradient was only 10 pg/mL [14].
Of note, only three patients had a complete recovery after surgery [7, 9, 10]. This percentage is below that reported in the literature [15] emphasizing that occipital bone is a difficult location to approach and for which combined treatments might be necessary. Medical treatments are second-line therapy for TIO patients with undetectable lesions, tumors not completely excised or located in sites difficult to be treated surgically, or with contraindications to surgery [1]. Traditionally, phosphate salts and active vitamin D metabolites are employed. Calcimimetics have been rarely used. Burosumab (an anti-FGF23 monoclonal antibody) has been recently approved for the treatment of patients with TIO based on the results obtained in two clinical trials [3, 4]. However, in these studies, the results were not entirely satisfactory. For example, mean serum phosphate was barely in the normal range, fractures persisted after 2 years of treatment, new fractures developed, and the effect on pain was absent in one study [3] and moderate in the other [4]. If these findings reflect the need to better define the appropriate dosing regimen [5] and/or the expression (and secretion) by tumor cells of other phosphatonins (e.g., MEPE, FGF7, sFRP-4) [1, 5, 16,17,18], it remains to be established. Interestingly, in two cases of TIO caused by tumors located in the occipital bone, PRRT177Lu-DOTATATE was used [8, 11]. Even though some short-term benefits were observed in both cases, further studies are needed to evaluate the safety and efficacy of PRRT177Lu-DOTATATE on a long-term period.
Finally, the finding is of interest in that in all patients, the tumor was localized in the left side of the occipital bone, and in five of them, the clivus was also involved. The existence of anatomical differences between the right and left sides of the occipital bone is well established. For example, mean values of condylar width and sagittal angle measurements were found significantly higher on the left side compared to the right one [19]. However, how these differences can reconcile with the development of the tumor remains questionable. The contribution of developmental anomalies involving the occipital bone, as in other more common, albeit rare, pathologic conditions occurring at this site (i.e., notochordal remnants/inclusions and chordoma) [20], seems unlikely. Indeed, PMTs fail to show any skeletal and soft tissue site–specific distribution having been reported virtually everywhere [1, 2].
In conclusion, we presented a new case of TIO caused by a tumor involving the occipital bone and a review of the pertinent literature regarding TIO associated with tumors occurring at this site. A meticulous patient analysis was performed to detail relevant clinical aspects. Major limitations are the retrospective nature of the study and the lack of important laboratory data such as pre- and/or post-treatment serum FGF23 levels and histological diagnosis in some cases. Overall, our analysis indicates that the occipital bone represents a challenging location for TIO-causing tumors for which a multidisciplinary approach is required to obtain the final cure of the patient.
Data availability
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Minisola S, Fukumoto S, Xia W, Corsi A, Colangelo L, Scillitani A, Pepe J, Cipriani C, Rv T (2022) Tumor-induced osteomalacia: a comprehensive review. Endocr Rev. https://doi.org/10.1210/Endrev/Bnac026
De Beur J, Sm MS, Wb X et al (2023) Global guidance for the recognition, diagnosis, and management of tumor-induced osteomalacia. J Intern Med 293:309–328. https://doi.org/10.1111/Joim.13593
Imanishi Y, Ito N, Rhee Y et al (2021) Interim analysis of a phase 2 open-label trial assessing burosumab efficacy and safety in patients with tumor-induced osteomalacia. J Bone Miner Res 36:262–270. https://doi.org/10.1002/Jbmr.4184
De Beur J, Sm MP, Tj W et al (2021) Burosumab for the treatment of tumor-induced osteomalacia. J Bone Miner Res 36:627–635. https://doi.org/10.1002/Jbmr.4233
Colangelo L, Sonato C, Riminucci M, Corsi A, Minisola S (2023) Appropriate dosing of burosumab in tumor-induced osteomalacia. Osteoporos Int 34:421. https://doi.org/10.1007/S00198-022-06617-2
Yoshioka K, Nagata R, Ueda M, Yamaguchi T, Konishi Y, Hosoi M, Inoue T, Yamanaka K, Iwai Y, Sato T (2006) Phosphaturic mesenchymal tumor with symptoms related to osteomalacia that appeared one year after tumorectomy. Intern Med 45:1157–1160. https://doi.org/10.2169/Internalmedicine.45.1797
Mk M, Mk P (2017) Unusual calvarial tumour-oncogenic osteomalacia. Br J Neurosurg 31:495–496. https://doi.org/10.3109/02688697.2016.1161165
Basu S, Fargose P (2016) 177lu-Dotatate Prrt in recurrent skull-base phosphaturic mesenchymal tumor causing osteomalacia: a potential application of Prrt beyond neuroendocrine tumors. J Nucl Med Technol 44:248–250. https://doi.org/10.2967/Jnmt.116.177873
Mulani M, Somani K, Bichu S, Billa V (2017) Tumor-induced hypophosphatemia. Indian. J Nephrol 27:66–68. https://doi.org/10.4103/0971-4065.179302
Mishra T, Ma D, Patel K, Ga M (2019) Phosphaturic mesenchymal tumors involving skull bones: report of two rare cases. Asian J Neurosurg 14:253–255. https://doi.org/10.4103/Ajns.Ajns_176_17
Shah R, Ar L, Rs J et al (2019) Tumor induced osteomalacia in head and neck region: single center experience and systematic review. Endocr Connect 8:1330–1353. https://doi.org/10.1530/Ec-19-0341
Luthra K, Dk G, Lele V (2022) A pictorial essay of somatostatin receptor imaging in tumor-induced osteomalacia: a single institutional experience. Indian J Nucl Med 37:83–90. https://doi.org/10.4103/Ijnm.Ijnm_101_21
Sj R, Yeh R, Wong T, Dumeer S, Br M, Remotti F, Chikeka I, Ak R (2019) Tumor-induced osteomalacia - current imaging modalities and a systematic approach for tumor localization. Clin Imaging 56:114–123. https://doi.org/10.1016/J.Clinimag.2019.04.007
Hana T, Tanaka S, Nakatomi H, Shojima M, Fukumoto S, Ikemura M, Saito N (2017) Definitive surgical treatment of osteomalacia induced by skull base tumor and determination of the half-life of serum fibroblast growth factor 23. Endocr J 64:1033–1039. https://doi.org/10.1507/Endocrj.Ej17-0177
Li X, Jiang Y, Huo L et al (2020) Nonremission and recurrent tumor-induced osteomalacia: a retrospective study. J Bone Miner Res 35:469–477. https://doi.org/10.1002/Jbmr.3903
Riminucci M, Colangelo L, Ungari C, Cassoni A, Minisola S, Corsi A (2022) Naso-ethmoidal phosphaturic mesenchymal tumor: a rare tumor site for an uncommon paraneoplastic syndrome. Ear Nose Throat J 101:289–291. https://doi.org/10.1177/0145561320940869
Kritmetapak K, Kumar R (2023) Phosphatonins: from discovery to therapeutics. Endocr Pract 29:69–79. https://doi.org/10.1016/J.Eprac.2022.09.007
Palmisano B, Pace A, Colangelo L, Polimeni R, Sonato C, Messineo D, Minisola S, Magliulo G, Riminucci M, Corsi A (2023) "Non-Phosphaturic" variant of phosphaturic mesenchymal tumor of the middle ear expressing multiple phosphatonins. Ear Nose Throat J 1455613221149635. https://doi.org/10.1177/01455613221149635
Gumussoy I, Sb D (2020) Morphometric analysis of occipital condyles using alternative imaging technique. Surg Radiol Anat 42:161–169. https://doi.org/10.1007/S00276-019-02344-2
Ramesh T, Sv N, Gg T, Saker E, Shoja M, Loukas M, Rj O, Rs T (2017) Update on the notochord including its embryology, molecular development, and pathology: a primer for the clinician. Cureus 9:E1137. https://doi.org/10.7759/Cureus.1137
Funding
Open access funding provided by Università degli Studi di Roma La Sapienza within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics statement
All the clinico-pathologic investigations detailed in the manuscript have been conducted in accordance with the Declaration of Helsinki and its later amendments or comparable ethical standards.
Patient consent to participate
Written informed consent for the publication of data and images was obtained from the patient.
Conflicts of interest
Salvatore Minisola served as a speaker for Abiogen, Bruno Farmaceutici, Diasorin, Kyowa Kirin, UCB. He also served in the advisory board of Eli Lilly, Kyowa Kirin, UCB. SF received consulting fee Kyowa Kirin, Co., Ltd. Luciano Colangelo, Chiara Sonato, Cristiana Cipriani, Jessica Pepe, Giorgia Farinacci, Biagio Palmisano, Marco Occhiuto, Mara Riminucci, and Alessandro Corsi declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Colangelo, L., Sonato, C., Cipriani, C. et al. Occipital bone and tumor-induced osteomalacia: a rare tumor site for an uncommon paraneoplastic syndrome. Arch Osteoporos 18, 94 (2023). https://doi.org/10.1007/s11657-023-01305-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11657-023-01305-y