Abstract
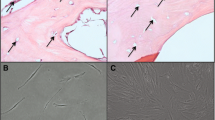
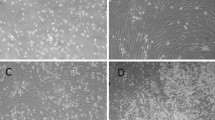
Bone repair is a major concern in reconstructive surgery. Transplants containing osteogenically committed mesenchymal stem cells (MSCs) provide an alternative source to the currently used autologous bone transplants which have limited supply and require additional surgery to the patient. A major drawback, however is the lack of a critical mass of cells needed for successful transplantation. The purpose of the present study was to test the effects of FGF2 and FGF9 on expansion and differentiation of MSCs in order to establish an optimal culture protocol resulting in sufficient committed osteogenic cells required for successful in vivo transplantation. Bone marrow-derived MSCs cultured in αMEM medium supplemented with osteogenic supplements for up to three passages (control medium), were additionally treated with FGF2 and FGF9 in various combinations. Cultures were evaluated for viability, calcium deposition and in vivo osteogenic capacity by testing subcutaneous transplants in nude mice. FGF2 had a positive effect on the proliferative capacity of cultured MSCs compared to FGF9 and control medium treated cultures. Cultures treated with FGF2 followed by FGF9 showed an increased amount of extracted Alizarin red indicating greater osteogenic differentiation. Moreover, the osteogenic capacity of cultured cells transplanted in immunodeficient mice revealed that cells that were subjected to treatment with FGF2 in the first two passages and subsequently to FGF9 in the last passage only, were more successful in forming new bone. It is concluded that the protocol using FGF2 prior to FGF9 is beneficial to cell expansion and commitment, resulting in higher in vivo bone formation for successful bone tissue engineering.




Similar content being viewed by others
References
Agata H.; Asahina I. et al. Effective bone engineering with periosteum-derived cells. J Dent Res 86: 79–83; 2007.
Bloemers F. W.; Blokhuis T. J. et al. Autologous bone versus calcium-phosphate ceramics in treatment of experimental bone defects. J Biomed Mater Res B Appl Biomater 66: 526–531; 2003.
Caterson E. J.; Nesti L. J. et al. Human marrow-derived mesenchymal progenitor cells: isolation, culture expansion, and analysis of differentiation. Mol Biotechnol 20: 245–256; 2002.
Colter D. C.; Class R. et al. Rapid expansion of recycling stem cells in cultures of plastic-adherent cells from human bone marrow. Proc Natl Acad Sci USA 97: 3213–3218; 2000.
De Moerlooze L.; Dickson C. Skeletal disorders associated with fibroblast growth factor receptor mutations. Curr Opin Genet Dev 7: 378–385; 1997.
Ever L.; Zhao R. J. et al. Fibroblast growth factor receptor 2 plays an essential role in telencephalic progenitors. Dev Neurosci 30: 306–318; 2008.
Fakhry A.; Ratisoontorn C. et al. Effects of FGF-2/-9 in calvarial bone cell cultures: differentiation stage-dependent mitogenic effect, inverse regulation of BMP-2 and noggin, and enhancement of osteogenic potential. Bone 36: 254–266; 2005.
Fujita M.; Kinoshita Y. et al. Proliferation and differentiation of rat bone marrow stromal cells on poly(glycolic acid)-collagen sponge. Tissue Eng 11: 1346–1355; 2005.
Govindarajan V.; Overbeek P. A. FGF9 can induce endochondral ossification in cranial mesenchyme. BMC Dev Biol 6: 7; 2006.
Gregory C. A.; Gunn W. G. et al. An Alizarin red-based assay of mineralization by adherent cells in culture: comparison with cetylpyridinium chloride extraction. Anal Biochem 329: 77–84; 2004.
Haynesworth S. E.; Goldberg V. M. et al. Diminution of the number of mesenchymal stem cells as a cause for skeletal aging. Musculoskeletal Soft-tissue Aging: Impact on Mobility. In: Buckwalter J. A., Goldberg V. M.; Y. W. SL-Y. (eds) Rosemont, American Academy of Orthopaedic Surgeons. pp. 79–87, 1994.
Haynesworth S. E.; Goshima J. et al. Characterization of cells with osteogenic potential from human marrow. Bone 13: 81–88; 1992.
Hecht D.; Zimmerman N. et al. Identification of fibroblast growth factor 9 (FGF9) as a high affinity, heparin dependent ligand for FGF receptors 3 and 2 but not for FGF receptors 1 and 4. Growth Factors 12: 223–233; 1995.
Hori Y.; Inoue S. et al. Effect of culture substrates and fibroblast growth factor addition on the proliferation and differentiation of rat bone marrow stromal cells. Tissue Eng 10: 995–1005; 2004.
Hung S. C.; Chen N. J. et al. Isolation and characterization of size-sieved stem cells from human bone marrow. Stem Cells 20: 249–258; 2002.
Hurley M. M.; Abreu C. et al. Expression and regulation of basic fibroblast growth factor mRNA levels in mouse osteoblastic MC3T3-E1 cells. J Biol Chem 269: 9392–9396; 1994.
Hurley M. M.; Tetradis S. et al. Parathyroid hormone regulates the expression of fibroblast growth factor-2 mRNA and fibroblast growth factor receptor mRNA in osteoblastic cells. J Bone Miner Res 14: 776–783; 1999.
Ignelzi Jr. M. A.; Wang W. et al. Fibroblast growth factors lead to increased Msx2 expression and fusion in calvarial sutures. J Bone Miner Res 18: 751–759; 2003.
Jackson R. A.; Nurcombe V. et al. Coordinated fibroblast growth factor and heparan sulfate regulation of osteogenesis. Gene 379: 79–91; 2006.
Jones E.; McGonagle D. Human bone marrow mesenchymal stem cells in vivo. Rheumatology (Oxford) 47: 126–131; 2008.
Kotobuki N.; Hirose M. et al. Cultured autologous human cells for hard tissue regeneration: preparation and characterization of mesenchymal stem cells from bone marrow. Artif Organs 28: 33–39; 2004.
Li Z. G.; Mathew P. et al. Androgen receptor-negative human prostate cancer cells induce osteogenesis in mice through FGF9-mediated mechanisms. J Clin Invest 118: 2697–2710; 2008.
Luria E. A.; Owen M. E. et al. Bone formation in organ cultures of bone marrow. Cell Tissue Res 248: 449–454; 1987.
Matsubara T.; Suardita K. et al. Alveolar bone marrow as a cell source for regenerative medicine: differences between alveolar and iliac bone marrow stromal cells. J Bone Miner Res 20: 399–409; 2005.
Maurice S.; Srouji S. et al. Isolation of progenitor cells from cord blood using adhesion matrices. Cytotechnology 54: 121–133; 2007.
Montero A.; Okada Y. et al. Disruption of the fibroblast growth factor-2 gene results in decreased bone mass and bone formation. J Clin Invest 105: 1085–1093; 2000.
Muraglia A.; Martin I. et al. A nude mouse model for human bone formation in unloaded conditions. Bone 22: 131S–134S; 1998.
Ornitz D. M.; Marie P. J. FGF signaling pathways in endochondral and intramembranous bone development and human genetic disease. Genes Dev 16: 1446–1465; 2002.
Quarto N.; Longaker M. T. Differential expression of specific FGF ligands and receptor isoforms during osteogenic differentiation of mouse adipose-derived stem cells (mASCs) recapitulates the in vivo osteogenic pattern. Gene 424: 130–140; 2008.
Quarto N.; Wan D. C. et al. Molecular mechanisms of FGF-2 inhibitory activity in the osteogenic context of mouse adipose-derived stem cells (mASCs). Bone 42: 1040–1052; 2008.
Reyes M.; Lund T. et al. Purification and ex vivo expansion of postnatal human marrow mesodermal progenitor cells. Blood 98: 2615–2625; 2001.
Sabbieti M. G.; Marchetti L. et al. Prostaglandins regulate the expression of fibroblast growth factor-2 in bone. Endocrinology 140: 434–444; 1999.
Sekiya I.; Larson B. L. et al. Expansion of human adult stem cells from bone marrow stroma: conditions that maximize the yields of early progenitors and evaluate their quality. Stem Cells 20: 530–541; 2002.
Slater B. J.; Kwan M. D. et al. Mesenchymal cells for skeletal tissue engineering. Expert Opin Biol Ther 8: 885–893; 2008.
Smith J. R.; Pochampally R. et al. Isolation of a highly clonogenic and multipotential subfraction of adult stem cells from bone marrow stroma. Stem Cells 22: 823–831; 2004.
Solchaga L. A.; Penick K. et al. FGF-2 enhances the mitotic and chondrogenic potentials of human adult bone marrow-derived mesenchymal stem cells. J Cell Physiol 203: 398–409; 2005.
Sotiropoulou P. A.; Perez S. A. et al. Characterization of the optimal culture conditions for clinical scale production of human mesenchymal stem cells. Stem Cells 24: 462–471; 2006.
Sullivan R.; Klagsbrun M. Purification of cartilage-derived growth factor by heparin affinity chromatography. J Biol Chem 260: 2399–2403; 1985.
Toai T. C.; Thao H. D. et al. In vitro culture and differentiation of osteoblasts from human umbilical cord blood. Cell Tissue Bank 11: 269–280; 2009.
Tsutsumi S.; Shimazu A. et al. Retention of multilineage differentiation potential of mesenchymal cells during proliferation in response to FGF. Biochem Biophys Res Commun 288: 413–419; 2001.
van den Bos C.; Mosca J. D. et al. Human mesenchymal stem cells respond to fibroblast growth factors. Hum Cell 10: 45–50; 1997.
Young C. S.; Abukawa H. et al. Tissue-engineered hybrid tooth and bone. Tissue Eng 11: 1599–1610; 2005.
Yu K.; Xu J. et al. Conditional inactivation of FGF receptor 2 reveals an essential role for FGF signaling in the regulation of osteoblast function and bone growth. Development 130: 3063–3074; 2003.
Yu L. Y.; Pei Y. et al. Effect of fibroblast growth factor 9 on Runx2 gene promoter activity in MC3T3-E1 and C2C12 cells. Chin Med J (Engl) 120: 491–495; 2007.
Acknowledgments
The authors wish to acknowledge MAGNET, Israel Ministry of commerce & Industry, No. 2007992 for supporting this research. The excellent technical assistance of Janette Zavin, Pessia Shenzer is also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: J. Denry Sato
Rights and permissions
About this article
Cite this article
Kizhner, T., Ben-David, D., Rom, E. et al. Effects of FGF2 and FGF9 on osteogenic differentiation of bone marrow-derived progenitors. In Vitro Cell.Dev.Biol.-Animal 47, 294–301 (2011). https://doi.org/10.1007/s11626-011-9390-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-011-9390-y