Abstract
The history and species status of free-ranging goats inhabiting the Eastern Mediterranean islands is discussed with reference to morphometric, archaeological and genetic findings. A case study on the free-ranging goats on Crete (Capra aegagrus cretica) is presented. The phenotype of the Cretan goat resembles that of the wild bezoar goat (C. aegagrus). However, the mitochondrial DNA of cytochrome b and d-loop sequences shows affinity with domestic goats. It has been suggested that the Cretan goat represents a feral animal that was introduced onto the island during the 6th millennium b.c. as a primitive domestic, and has retained the wild morphotype but has undergone significant genetic change. An alternative explanation, and the one that is proposed here, is that the goat was introduced onto the island in wild form and released as a food source. Subsequent introgressions with domestic animals, especially ewes, have influenced its genotype. These conclusions are applicable to other free-living goats and sheep which inhabit islands in the Eastern Mediterranean.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Humans play an important role in the dispersion of mammalian species through the introduction of animals into new regions and habitats. Animals chosen for translocation may be wild, ‘managed’ or domestic. They may be introduced for food, sport, pest control or even for religious or other symbolic reasons (e.g., 22, 60, 64, 73). Accidental or passive translocation, which Vigne (73) called ‘facilitated immigration,’ includes animals escaping from confinement, shipwrecks and landfalls or the transport of ‘stowaways’ such as commensals and pests.
Examples of translocation of taxa in continental regions include the introduction of fallow deer (Dama dama) from Asia Minor into North-West Africa (7), and the introduction of the genet (Genetta genetta) from North Africa into Europe (14). However, the impact of introductions of non-indigenous faunal is most evident in insular settings since these are ‘closed’ systems colonised via temporary landbridges, radiation of taxa which swim/drift/fly, or else via human maritime activity [e.g., (18, 22, 44, 64, 70, 72, 73)]. This is especially visible on the Mediterranean islands, due to the antiquity and intensity of human activity. Species that were extraneous to the Quaternary insular fauna were introduced and resulted in the gradual replacement of the island fauna, especially endemic elements (18, 44, 46, 70, 73). Indeed, the vulnerability of insular fauna to allochthonous domestic herbivores and carnivores is evident on islands even today [e.g., (3, 6, 52)]. Although commonly thought to be a Neolithic phenomenon, recent research has shown that the extinction of most insular taxa, may pre-date the Neolithic period. This accords well with a growing corpus of information attesting to pre-Neolithic occupation of many islands (47, 50).
Of the numerous anthropochorous taxa on Mediterranean islands, the history of the tribe Caprini (subfamily Caprinae) is especially interesting given their historical and present-day importance in the economy of the region as providers of meat, milk, fibre and skin (21). Aside from Myotragus balearicus, an endemic caprine of the Balearic Islands that became extinct ca. 4,500 years ago (38), it is evident today that no authochtonous caprines are found on the Mediterranean islands (43, 44, 71, 74, 76). However, today, in addition to domestic sheep (Ovis aries) and goat (Capra hircus), free-ranging caprines are found on several Mediterranean islands: sheep on Cyprus, Sardinia and Corsica; and goats on Crete, Montecristo and islets in the Sporades and Cylades (Fig. 1).
Map of Eastern Mediterranean showing location of islands with present-day free-living sheep (1–3) and goats (a–g). (Based on (44): Fig. 3). 1 – Corsica; 2 – Sardinia; 3 – Cyprus; a – Montecristo; b – Samothrace; c – Youra; d – Antimilos; e – Crete (including the islets of Theodorou, Dia, Agii Pantes).
Until recently, it was thought that the free-living caprines of the Mediterranean islands were in fact authochtonous forms. Today, it is widely accepted that they were introduced by humans during the Neolithic period when the islands were first settled (9, 10, 24, 43, 57, 72, 74). Despite the shift in paradigm, their taxonomic and genetic status (wild, domestic, feral) at the time of their introduction as well as today, remains unclear. The situation is complicated by the fact that they closely resemble their Near Eastern wild progenitors – the Asiatic mouflon (O. orientalis, also termed O. gmelini) and the bezoar goat (C. aegagrus) – in morphology, colouration and behaviour (Fig. 2). As noted by Ciani et al. (10), the classification of insular caprines as specific or sub-specific geographic forms has, until now, been based on “arbitrary criteria and on examination of scattered materials.” More substantive investigations are needed to rectify this situation. This paper offers a brief resume of the origin and current specific attribution of free-ranging goats inhabiting islands in the Eastern Mediterranean today, and presents a case study of the free-ranging Cretan goat (C. a. cretica).
Free-Ranging Goats
Present Populations
Today, free-ranging goats are found only on four islands in the Eastern Mediterranean – Montecristo, Antimilos, Youra and Crete (Fig. 1). Until historic times, free-living goats also inhabited three other islands: Samothrace (Greece); Tavolara (Italy); La Galite (Tunisia) (13, 44, 46, 63). It is likely that goats were present on other islands but became extinct in antiquity as suggested by evidence from the Late Palaeolithic/Mesolithic site of Grava on Corfu where evidence was found for “a wild goat economy showing a tendency of domestication” [cited in Mavridis (50)]. Free-ranging goats are not found today on this island.
The island of Montecristo in the Tuscany Archipelago, Tyrrhenian Sea off Italy is a small island some 10 km2. The goat population on Montecristo consisted of approximately 300–350 individuals (9). The origin of these goats is unknown but it is suggested that they were first introduced in antiquity with further stock brought in as game in the second half of the 19th century (59). The goats exhibit a relatively uniform phenotype (9).
Antimilos (Erimomilos) is a very small island located just northeast of the large Cycladic island of Milos. Today it serves as a national park for free-living goats.
Couturier (13) notes that frequent hybridization occurred between the free-living Capra and domestic goats which inhabited the island. It is not surprising then that of the 400–500 animals found on the island in the 1960s, Schultze-Westrum (63) considered only 100 to truly resemble the typical wild bezoar goat in terms of morphology and pelage. He attributed Antimilos goats to C. a. pictus.
Free-living goats are found on the islet of Youra (also known as Jura/Yioura/Yura/Giura) and neighbouring islets of Peristera (Phthiotis), Lamia, Atalantonisi (Evoikos) and Psili (Argolikos) in the Sporades (63). According to Ciani and Masseti (9), they number 300. They have been attributed to C. a. dorcas (13), and more recently they were assigned by Schultze-Westrum (63) to C. a. f. hircus since he considered them to be feral animals and as a local subspecies by (9) Ciani and Masseti, who designated them as C. a. jourensis.
The largest population of free-living goats on Mediterranean islands is found on Crete (Greece) and its offshore islets. Populations of C. a. cretica, colloquially known as the Cretan goat or Agrimi, are concentrated in the White Mountains of Western Crete (elevation 2,400 m), especially in the vicinity of cliffs at the head of Samaria Gorge, the Samaria National Forest and on the three islets of Dia, Theodorou and Agii Pantes that serve as reserves for the goats (Fig. 3a, b) (51 and references therein; 55, 63, 77). By 1960, the Cretan goat was a threatened species with an estimated population size of only 300 animals (63). Today, the population size on Crete and the islets is ca. 1,000 animals (58).
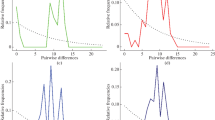
Locations from which samples of agrimi were taken showing the relationship between the three agrimi populations sampled and the polymorphic sites on two regions of the cytochrome b gene. Samaria Gorge on Crete is the parent population from which Theodorou Island population is derived; the Hai-Bar population is derived from Theodorou Island. (a) Crete and Hai-Bar, Israel, (b) Crete: Samaria Gorge and Theodorou Island, (c) Phylogenetic analyses of the three agrimi populations, minimum evolution using MEGA. [Two regions of the cytochrome b (353 bp).
Insular goats are smaller than mainland wild bezoar goat, C. aegagrus, averaging 85 cm in withers height compared to 95 cm in the wild bezoar (15). However, they resemble the wild form in coat colour and patterning, with a distinct black line along the spine and base of neck forming a collar (Fig. 2). Flanks are brown with a white underbelly, but pelage colour varies seasonally – from brown-grey in winter to yellow-chestnut brown in summer, as well as with age. Males are bearded.
All insular goat populations have homonymous horns (right-handed spiral), whereas only a few Asiatic bezoar goats sprout horns with a distinct homonymous twist, usually in the upper third of the horn (15). The wild bezoar has well-developed scimitar shaped horns that curve backwards, with a long, sharp keel on the front surface. The tips turn slightly inward, facing each other. In this latter feature, insular goats differ among themselves – e.g., those on Antimilos are distinguished by a slight outward turn of the horn tip, while those on Crete more closely resemble bezoar goats (15). Groves (17) interprets this as a sign that insular goats “originate from different breeds.” However, differences in selective pressures as well as in the extent to which they have inter-bred with domestic animals are more likely to be the causes. Horn size in all insular ewes is markedly reduced, as in the wild form.
Groves (17) examined brain capacity in free-living goats from Antimilos, Youra and Crete, and reported that (a) there were no marked differences between the three island samples and that (b) all three samples were intermediate between true wild and domestic goats “overlapping marginally with both”. It is interesting to note that the Mediterranean insular goats all have far larger brain capacities than feral goats from Juan Fernandez Island off Chile, established some 400 years ago. Consequently, Groves (17) concluded that the wild goats of the Aegean “are derived from very primitive domestic goats.” Epstein (15) notes that the crania of wild bezoar goats is short and high (‘egg-shaped’) compared to those from Youra which are long and low – another domestic characteristic which emphasizes their feral origin.
Past Populations
According to Cherry (8; Table 2), the island of Montecristo was probably never inhabited. The origin and the time of the introduction of goats onto Montecristo are unknown but the goats clearly represent an ancient population. Over time, they have interbred with domestic animals introduced onto the island, especially in the second part of the 19th century. Montecristo goats are today considered feral (59).
The origin of the Capra on Antimilos is not known. However, the neighbouring, far-larger island of Milos was first occupied in the 4th or 3rd millennium b.c. (8; Table 1) such that if Antimilos was occupied by humans, it probably did not ante-date that of the larger island. A Cretan bronze arrow sharpener dating to the 16–17th centuries b.c. attests to the antiquity of hunting on Antimilos (63).
On the island of Youra, the Upper Mesolithic levels of Cyclops Cave have yielded remains of Capra (50). The Mesolithic period in Cyclops Cave spans the period 8500–6500 uncal. b.c. such that the goats probably fall in the 6500 b.c. range (62). As discussed by Masseti and Trantalidou (48), this represents one of the earliest introductions of C. aegagrus into the Aegean. At present, no further published information is available on these remains.
The earliest, securely dated goat bones from Crete derive from Stratum X (the ‘Camp’) at Knossos dating to the end of the 7th millennium b.c. – 6,100 ± 180 uncal. b.c.; 5,980 ± 130 b.c. They were found together with remains of other domesticates – sheep, cattle, pig and dog (32, 33). Goats in this sample and in the overlying Early Neolithic Ia were fewer in number than sheep, but considerably larger in size. Given the paucity of these goat samples (5 and 4 bones, respectively), no comparative analyses could be carried out with mainland goats. However, the size of sheep in the ‘Camp’ assemblage was comparable to that of contemporaneous domestic sheep populations from the Greek mainland and other domestic sheep samples from Europe. The combined caprine sample showed a biased cull with 60% slaughtered by 24 months, 90% by 36 months. Bones of large sized goats (identified as agrimi) are found together with those of domestic sheep and goat in various excavations on Crete dating from the 6th millennium b.c. onwards (32, 33, 75). In the Minoan period (ca. 3,650–1,170 uncal. b.c.) the agrimi is clearly depicted in art works as a game animal (43, 58). These data indicate that the agrimi was well established as a hunted species following its introduction in the Neolithic.
Species Status
Like that of sheep, the systematics of the free-ranging goats of the Mediterranean islands has been controversial. Researchers such as Couturier (12), Nowak and Paradiso (53) and Mason (49) recognised five wild sub-species of wild bezoar goat (C. aegagrus), with two forms occurring only on the Mediterranean islands – C. aegagrus cretica on Crete and C. a. picta on Antimilos. Even in 1991, Harrison and Bates (19) listed C. a. cretica as a wild form of C. aegagrus, while Nowak (54) listed C. a. cretica as a separate species but not C. a. picta. Similarly, Van den Brink (69) suggested that the agrimi are “wild or mainly wild,” but that the goats on Youra and Montecristo are feral and those on Antimilos “are certainly not completely pure.” Heptner et al. (24) also specifically noted that the goats of Youra pertain to domestic animals turned wild. According to Schultze-Westrum (63), all the Sporades and Cycladic goats, including those on Youra and Antimilos, represent feral animals, whereas all Cretan free-living goats were classified as a subspecies of wild goat endemic to Crete. In an on-line report on taxonomy of caprinae, the IUCN/SSC Caprinae specialist group (31) concluded that “the Cretan agrimi (C. a. cretica) is a domestic form and should not be considered a subspecies of wild goat” – a finding supported by other researchers (9, 11, 17, 35, 39, 43, 46).
Couturier (13) suggested that the goats of Montecristo are the result of hybridization of wild and domestic forms. This has been given greater credence by biochemical genetic analyses which have demonstrated that the Montecristo goats have very high levels of genetic variability, indicating that the gene pool of this population has been affected by multiple introductions of domestic goats (59). Selective pressures, high inbreeding and decreased genetic variation have acted on the Montecristo goats to reconstruct a “wild” phenotype although the goats are actually feral.
Despite their phenotypic similarity, results of a preliminary study on genetic affinities between goats from Montecristo, Samothrace and Crete using microsatellites (10) demonstrated marked variability in the pattern of their alleles. The Montecristo specimens also exhibited low variability while those from Crete had a greater degree of polymorphism, suggesting marked inter-population variation at the genetic level.
The resume presented above illustrates that there is some consensus concerning the feral status of goats from Youra, Montecristo and Antimilos, but that of the Cretan goats remain disputed. In this respect, based on the archaeozoological record, Jarman (32) noted that “in view of the dearth of available data on the osteological relationships between wild, feral and domestic populations of the same species this would be a difficult proposition to test.’’ Consequently, the resolution of this issue lies with genetic studies.
In their analysis of mitochondrial DNA (mtDNA) of wild and domestic goats, Manceau et al. (41) corroborated previous research by Takada et al. (67), who found that domestic goats and wild C. aegagrus were monophyletic, thereby supporting C. aegagrus as the progenitor of the domestic form. Moreover, the single specimen of C. aegagrus cretica, which Manceau et al. (41) had analyzed, had an almost identical haplotype to that of the domestic goat, indicating that the agrimi may be a feral animal. To further evaluate this claim, we examined the mtDNA of C. aegagrus cretica populations, to test for the extent of divergence between populations within a relatively short time scale (5).
Case Study: The Genetics of the Agrimi
Samples
For this study, blood samples were taken from 10 tranquilized individuals of C. a. cretica that were managed in the Hai-Bar Reserve on Mount Carmel, Northern Israel. These individuals were part of a breeding herd that was established in August 1975, when six goats were brought to Israel from Theodorou Island, Crete. The samples will be related to as “Hai-Bar”.
In addition, in 1996 skeletons of modern C. a. cretica specimens from Samaria Gorge and from the islet of Theodorou were collected (Fig. 3a, b). The skeletons represent animals that were found in the wild and are assumed to have died of natural causes. The osteological remains were characterized based on their morphology, measured and then sampled for DNA analysis. The samples comprise teeth, bones and dry skin that was still adhering to the bones. A maximum of 21 samples, representing different animals, were brought to Israel. Using ancient DNA techniques (27, 36), we extracted DNA from a total of 12 samples: 7 from Theodorou Island and 5 from Samaria Gorge (34, 35).
Methods
Blood taken from the Hai-Bar animals was put in a 3-ml tube containing 7.5% EDTA to prevent clotting. DNA was extracted by using a method of isolation of DNA from peripheral blood (16). DNA from the modern skeletons was extracted from either bone, teeth or skin using guanidine thiocyanate (GuHCL) (4) and silica (28). The extracted DNA was analyzed by the PCR reaction (61). PCR amplification was performed with different sets of primers and under conditions specific for each reaction. DNA from blood was amplified in a volume of 50 μl through 25 cycles by using overlapping primers as described in (35). The DNA extracted from bones, teeth and skins was amplified in a volume of 25 μl through 40 cycles. Only two sets of primers amplified successfully in most individual [primer set 86–92 and G11–G12; see (35)]. The main PCR programme was as follows: 95°C for 10 min, 94°C for 30 s, 90 s annealing at 46–52°C (as set for each set of primers), 90 s elongation at 72°C. The double-stranded PCR products were run on 1.5% low melting agarose gels to determine whether there was a successful amplification in the PCR reaction. For all the PCR positive reactions of the Hai-Bar samples, DNA was purified by using the Boehringer DNA purification kit. The positive bands of the contemporary skeletal samples were cored directly from the gel using Pasteur pipettes. All the samples, purified and cored bands, were sequenced by the direct sequencing reaction using the Thermo Sequenase kit (Amersham).
It is important to note that for all the skeleton samples, utmost care was taken to prevent contemporary contamination during DNA extraction and amplification. Each stage of the procedure (DNA extraction and the PCR reaction) was carried out in a dedicated laboratory for ancient DNA studies. The extraction and amplification were carried out by using different UV hoods to eliminate contamination of contemporary DNA. Moreover, all the reagents and tubes were crossed-linked to prevent contemporary contamination. For the same reason, disposable sterile tubes, filtered tips and sterile reagents and solutions, and a special set of pipettes were used throughout the procedure. Multiple negative extraction and amplification control were included in each PCR reaction to detect contamination. To verify the authenticity of the sequences obtained through the research, DNA was sampled and studied for every specimen at least twice.
Analysis
A consensus sequence was determined for each sample by using the sequence obtained from the independent extractions/amplifications. A population genetic tree for the three populations of agrimi was generated via the MEGA programme (Fig. 3c). It is based on sequences of two regions of cytochrome b (i.e., all 3 bp mutations were analysed together). A distance matrix was calculated by using Kimura2 parameters, taking into account transitions and transversions – this included the three variable sites (position 216, 759 and 761 cytochrome b) and also parsimony informative sites. The tree is generated by using a minimum evolution (ME) approach obtained by neighbour joining (NJ).
The consensus sequences of the goats from the Hai-Bar were compared to caprine sequences in GeneBank. The comparison was accomplished by using the BLAST programme (Basic Local Alignment Search Tool) (1). The sequences from the contemporary skeletal samples were first compared to each other and then to the consensus sequence of the goats from the Hai-Bar (Fig. 3c), and finally to caprine sequences taken from GeneBank. All agrimi sequences generated by this project, those of unimproved domestic sheep (Awassi) and goat (Baladi) breeds from Israel (34), as well as published sequences of wild and domestic goats and sheep (2, 20, 31, 67), were aligned and a phylogenetic analysis run using the PAUP programme to study the relationships between the different species (Fig. 4b). A distance matrix was calculated by using Kimura2 parameters taking into account transitions and transversions. The tree is generated in MEGA using a ME approach obtained by neighbour joining (NJ) (Fig. 4a).
Phylogenetic analysis showing the distance between wild and domestic goat and sheep using minimum evolution model with Kimura 2 parameter test. (a) Phylogenetic analysis to distinguish between goat and sheep species. (b) Phylogenetic analysis of individuals representing different species of sheep and goats. The bootstrap values (shown on the branches) support the clade of domestic goats (C. hircus) and Agrimi, indicating a strong similarity between them, which in turn suggests that the Agrimi is a feral goat. Differences between sheep and goats are also supported by the bootstrap values. The representative sequences and their taxonomic identifications were taken from GeneBank. A full phylogenetic analysis using distance, maximum parsimony and maximum-likelihood using heuristic search and bootstrap analysis run on PAUP* Version 4.0 (66) can be found in the work of Kahila Bar-Gal (34) (Figures 2.1 and 2.2.1).
Results
Two mitochondrial regions, the cytochrome b and the d-loop, were sequenced for all three C. a. cretica populations sampled: Samaria Gorge (Crete), Theodorou Island and ten agrimi from the Hai-Bar, Israel. For the Hai-Bar population, all ten cytochrome b (1,140 bp) and d-loop sequences (217 bp) were found to be identical (Figs. 3c and 4a, b). The d-loop data are not presented in this paper, but can be obtained from Bar-Gal et al. (34, 35) upon request.
For the Samaria and Theodorou Island samples, with the two sets of primers for cytochrome b that were amplified, a total of 406 bp were sequenced. There is a similarity of 99.3% among all the sampled individuals with a maximum of 3 substitution differences (0.74%), which were found in positions 216, 759 and 761 (Fig. 3a, b, c). These three sites are polymorphic and vary among the contemporary sequences obtained from the skeletons. All 10 Hai-Bar goats have identical sequences. Two of the five Samaria Gorge specimens have thymine in position 759 and 761. The sequences of the seven samples from Theodorou Island show variability in all three positions. It was found that at position 216, four sequences have cytosine and one sequence has thymine. At positions 759 and 761, four sequences have adenine and two sequences have thymine. Specimen #21 from Theodorou Island is the only one whose sequence among the contemporary skeletons has an identical sequence to that of the Hai-Bar goats. Since the agrimi population at the Hai-Bar Carmel was derived from the Theodorou population, it is likely that the dominant sequence type was common among the founder animals that established the Hai-Bar Carmel population.
Comparison of the d-loop sequences showed 16 out of 17 specimens to have identical sequences. For the d-loop primer Gd1–Gd2, an exception was Theodorou specimen #20, which differed in two substitutions (a transition and a transversion) and one deletion. This high similarity among the d-loop sequences, a noncoding region, is surprising especially when differences were found among the cytochrome b sequences. It is possible that there are inter-populations differences but that these differences are not reflected in this study and that further research on the entire d-loop or microsatellite markers needs to be done.
As illustrated in Figs. 3c and 4a,b, the sequences obtained from a total of 17 specimens sampled from these different populations indicate that the C. a. cretica cytochrome b and d-loop sequences are closer to domestic goat (C. hircus) sequences and identical to that of the unimproved ‘Baladi’ breed of domestic goat from the Southern Levant (15). They differ significantly from published sequences for the wild bezoar goat (C. aegagrus).
Conclusion
Based on the palaeontological and archaeological data outlined here, it is clear that all insular free-living goats represent anthropochorous introductions into the Eastern Mediterranean islands. However, the timing of their introduction as well as their status, i.e., wild or domestic, and their further evolution on the islands requires clarification.
In terms of their mtDNA profiles, insular, free-ranging goats and sheep clade with domestic animals rather than with their wild counterparts (26, 35, 36, 40, 41). This has been interpreted by most researchers as reflecting the feral status of insular caprines. To wit, that they were introduced during the Neolithic period – even as early as the 9th millennium BP on Cyprus (74) – at an early stage in their domestication (‘primitive domesticates’), and then escaped or were released, thereby establishing feral populations.
However, the genetic data may also support a different scenario whereby caprines were introduced while still wild and released onto the islands. Subsequently, hey underwent extensive admixture with domesticates (also introduced, but at a later date). There are several points which would support this alternative scenario.
Firstly, the genetic data for free-living sheep and goats, including the agrimi, are restricted to mtDNA which reflects only the maternal contribution to the gene pool (26, 35, 40, 41, 56). Moreover, the genetic studies carried out to date are based on sequences from only two genes, cytochrome b and the d-loop, with the obtained sequence length often very short. This is certainly true for the agrimi; for cytochrome b, a maximum of 1,140 bp for Hai-Bar and only 406 bp for Theodorou, and for d-loop, a maximum of 217 bp. Longer, more complete sequences are needed to fully account for the inter-specific differences observed. Genetic studies that are based on mtDNA, and in particular cytochrome b, reflect a gene tree but conclusions are then drawn from this restricted data set for species status. In order to resolve the issue of inter-specific phylogenies, other nuclear genes, including the sex genes, need to be sequenced.
Based on morphological and biometric data, the insular caprines closely resemble their wild mainland progenitors from Asia Minor – the bezoar goat C. aegagrus and Asiatic mouflon O. gmelini. However, they do not precisely reproduce the wild form in phenotype, e.g., they are smaller in size, they differ in horn conformation. This may be due to founder effect and the small number of animals that have reproduced as well as to genetic drift resulting from their isolation rather than the fact that they represent ‘primitive domestics.’ It is interesting to note that more recent anthropochorous domesticates such as Soay sheep and goats on the San Juan Islands, have not reverted to the wild morphotype, but resemble the original domestic breed that was introduced (6, 11, 17).
It is well established that brain size in both captive-bred and feral animals is smaller than in wild animals, and that brain size in feral animals does not increase over time (37). However, the data provided by Groves (17) on brain size in insular caprines in support of their feral status is problematic. Most importantly, this study does not take into account the fact that brain size is plastic and influenced by environmental stimuli (23, 25). Sample sizes that were examined for both sheep and goats are extremely small and cannot account for intra-population variation. Moreover, a close examination of Groves’ (17; Fig. 5.3), where basi-cranial length in sheep is not logged but presented as raw data, clearly indicates that the different sheep populations follow a size cline, with domestic sheep having the shortest basi-cranial length, wild and insular mouflon overlapping and intermediate in size, while wild urials are the largest. Cranial capacity clearly follows the same pattern, suggesting that what is illustrated is the allometric relationship between brain case size and body size rather than a reduction in brain capacity. Moreover, mouflons are sexually dimorphic with skull length of 226–245 mm in males and 210–222 mm in females (males have far broader skulls) (19, 24). Since Groves did not state whether the studied specimens were male, female or of mixed sex, the role played by dimorphism in his findings is difficult to assess.
For goats a similar set of problems occur. Examination of Grove’s (17) Fig. 5.5 indicates that each goat population studied has an extremely wide range of variation in both cranial capacity and basi-cranial length, suggesting that there is no good correlation between these parameters within any of his groupings. Furthermore, his data does not take into account changes in skull shape, i.e., a skull with a shorter basi-cranium may be relatively broader. As for sheep, the contribution of sexual dimorphism is not accounted for.
A further issue relates to the archaeological record and the growing body of evidence attesting to pre-Neolithic human occupation/visits to islands in the Eastern Mediterranean, and the presence of introduced fauna in these levels, e.g., pig/boar in Akrotiri, Cyprus, and in Cyclops Cave, Youra. These new findings suggest that we need to rethink the timing and process of island colonisation. Consequently, free-living mouflon and goat populations may have been established in these early, less formal periods through the release of wild animals onto the islands. As such, they do not represent feral animals descended from ‘primitive domesticates’ transported to the island as has been suggested by other researchers [e.g., (11, 17, 23, 57, 68, 71, 73, 74)]. The subsequent Neolithic colonisation would then have heralded the arrival of managed caprine herds (together with other domesticates such as pig and cattle and cultivated plants). Free-ranging wild caprines would have continued to be obtainable only through hunting. Such a scenario would also offer enough time for wild caprine herds to establish themselves sufficiently and be exploited without becoming extinct. Thus, the Ovis and Capra found on the islands may not be feral animals (domesticates that turned wild), but relicts of wild taxa which were released/escaped on the islands and subsequently inter-bred with domesticates.
This scenario is supported by the fact that on most islands in the Eastern Mediterranean, a range of wild taxa that were never domesticated were also introduced into the islands, some together with caprines – deer, fox, and a range of felids and mustelids. For example, in Cyprus, fallow deer, which is larger than caprines and as difficult to transport, served as a staple food although it appears never to have been managed.
It is of special note that in all instances, the caprines found on the Mediterranean island closely resemble the wild type in colouration, horn conformation, sexual maturation age, frequencies of twinning. This suggests that they were introduced onto the islands as wild animals, i.e., before significant changes in morphology and biometry associated with domestication occurred. Subsequently, they underwent introgression with domestic animals that were introduced onto the islands – especially mating between wild males and domestic females – a feature that accounts for their mtDNA profile resembling that of domesticates than the wild type.
In a poster entitled “Islands as Natural Enclosures,” Masseti (45) raised the possibility that tamed or semi-domesticated herbivores were kept and bred in a free-ranging state on Mediterranean islands, serving as living repositories of animal protein. Indeed, there is extensive modern ethnographic evidence for transport of a large spectrum of wild taxa to islands to serve as ‘living larders.’ These often include animals that were never domesticated or even tamed. Horwitz et al. (29: 45) have argued that the initial founder stock brought to Cyprus were wild and that they “inter-bred with domestic animals which were brought from the mainland at a later date to replenish stock. The descendants of the first colonizing [wild] taxa are then the feral caprines found on many of the Mediterranean islands today...that have probably undergone extensive inbreeding with local domestic stock over time.” This is corroborated by the fact that the Cypriot anthropochorous faunal package was created over a long period of time with introduction of different taxa in several, separate introduction events.
In conclusion, it is suggested here that although free-ranging sheep and goats currently inhabiting the islands of the Eastern Mediterranean represent anthropochorous species, they originate from wild and not domestic animals. Consequently, they retain the wild phenotype of Asiatic mouflon (O. gmelini) and the bezoar goat (C. aegagrus). However, mtDNA and biochemical analyses demonstrate a genotypic affinity to domestic forms – O. aries and Capra hircus, respectively. It is proposed that this may be due to admixture with domestic animals subsequently brought to the islands. However, more rigorous genetic research, particularly of nuclear DNA, is required in order to fully understand the phylogeny of insular caprines.
List of specimens used in phylogeny and source
GeneBank Number | Species Attribution | Reference |
AJ867261 | Ovis orientalis | |
DQ236091; AF217257; AY879560; Awassi Breed | Ovis aries | Meadows et al. (2005); Bunch n.d. (34) |
D84203 | Ovis musimon | Arai, K. n.d. |
AF034735; AJ010055 | Capra ibex | Manceau et al., (20) |
AF217256; AJ009879 | Capra ibex nubiana | |
AF034736; AB044309; AJ293418; D84202; AJ231413 | Capra falconeri | |
X56289; AB004074; AB004070; AB004072;AB004071; D84201; AB004070; AF217254; Baladi breed | Capra hircus | |
AB110593; AB110592; AF034739; D84204; AJ231409; AJ231408 | Capra aegagrus cretica | |
AF217255 | Capra aegagrus cretica | (34) |
References
Altschul SF, Gish W, Miller W, Myers W, Myers EW, Lipman DJ (1992) Basic Local Alignment Search Tool (BLAST). J Mol Bio 215:403–410
Arai K, Munechika I, Ito I, Kikkawa A, Nakamura K, Kanazawa T, Kosugiyama M (1997) Phylogenetic relationship of Caprini estimated by cytochrome b gene sequence analysis. Anim Soc Technol 68:148
Atkinson I (1989) Introduced animals and extinctions. In: Western D, Pearl MC (eds) Conservation for the Twenty-First Century. Oxford University Press, New York, pp 54–75
Boom R, Sol CJA, Salimans MMM, Jansen CL, Wertheim-van Dillen PME, van der Noordaa J (1990) Rapid and simple method for purification of nucleic acids. J Clin Microevol 28:495–503
Bruford MW, Bradley DG, Luikart G (2003) DNA markers reveal the complexity of livestock domestication. Nat Rev (Genet) 4:900–910
Campbell K, Donlan CJ (2005) Feral goat eradications on islands. Conserv Biol 19(5):1362–1374
Chapman NG, Chapman DI (1980) The distribution of fallow deer: a worldwide review. Mamm Rev 10:61–138
Cherry J (1981) Patttern and process in the earliest colonization of the Mediterranean islands. Proc Prehist Soc 47:41–68
Ciani F, Masseti M (1991) Considerazioni sull’origine della populazione ircina dell’isola di Montecristo, nel mar Tirreno settentrionale. Elementi per un confronto cronologico-culturale con l’antica diffusione artificiale dell’egagro (Capra aegagrus Erxleben, 1777) nelle isole del Mediterraneo orientale. Ric Biol Selvag 18(suppl.):123–133
Ciani F, Masseti M, Palazzo M, Cappuccio A, Matassino D (1999) Genetic variations in the Mediterranean wild goat (Capra aegagrus Erxleben, 1777). Contrib Zoogeogr Ecol East Mediterr Region 1:69–74
Clutton-Brock J (1999) Natural History of Domesticated Mammals, 2nd edn. Cambridge University Press, Cambridge
Corbet GB (1978) The Mammals of the Palaearctic Region. A Taxonomic Review. British Museum of Natural History, London
Couturier MAJ (1959) Statut actuel des représentants du genre Capra dans le basin Méditerranéen. Morges. Colloque du Service de Sauvegarde, Compte Rendu de la 7e Reunion technie de l’UICN 5:12–19
Dobson M (1998) Mammal distributions in the western Mediterranean: the role of human intervention. Mamm Rev 28:77–88
Epstein H (1971) The Origin of the Domestic Animals of Africa. Africana Publishing, New York
Filon D, Rund D, Rachmilewitz EA, Oppenheim A (1992) Advances in the prenatal diagnosis of beta-thalassemia in Israel. In: Bonne-Tamir B, Adam A (eds) Genetic Diversity among Jews. Oxford University Press, New York, pp 358–364
Groves CP (1989) Feral mammals of the Mediterranean islands: documents of early domestication. In: Clutton-Brock J (ed) The Walking Larder. Unwin Hyman, London, pp 46–58
Hadjisterkotis E, Masala B, Reese DS (2000) The origin and extinction of the large endemic Pleistocene mammals of Cyprus. Lav Soc Ital Biogeogr 21:593–606
Harrison DL, Bates PJ (1991) Mammals of Arabia, 2nd edn. Harrison Zoological Museum Publication, Seven Oaks Kent
Hassanin A, Pasquet E, Vigne J-D (1998) Molecular systematics of the subfamily Caprinae (Artiodactyla, Bovidae) as determined from Cytochrome b sequences. J Mamm Evol 5(3):217–236
Hatziminaoglu Y, Boyazoglu J (2004) The goat in ancient civilisation: from the fertile crescent to the Aegean Sea. Small Rumin Res 51:123–129
Heinsohn TE (2001) Human influence on vertebrate zoogeography: animal translocation and biological invasions across and to the east of Wallace’s Line. In: Metcalfe I, Smith JMB, Morwood M, Davidson I (eds) Faunal and Floral Migrations and Evolution in SE Asia–Australasia. A.A. Balkema Publishers, Rotterdam, pp 154–170
Hemmer H (1990) Domestication. Cambridge University Press, Cambridge
Heptner VG, Nasimovich AA, Bannikov AC (1989) Mammals of the Soviet Union. Artiodactyls and Perissodactyls, vol. I. E.J. Brill, Leiden
Herre W, Röhrs M (1990) Haustiere-Zoologische Gesehen. 2nd edn. Gustav Fischer, Stuttgart
Hiendleder S, Kaupe B, Wassmuth R, Janke A (2002) Molecular analysis of wild and domestic sheep questions current nomenclature and provides evidence for domestication from two different subspecies. Proc R Soc Lond, B Biol Sci 269:893–904
Hofreiter M, Serre D, Poinar HN, Kuch M, Paabo S (2001) Ancient DNA. Nat Rev (Genet) 2:253–260
Hoos M, Paabo S (1993) DNA extraction from Pleistocene bones by silica-based purification method. Nucleic Acids Res 21:3913–3914
Horwitz LK, Tchernov E, Hongo H (2004) The domestic status of the early Neolithic fauna of Cyprus: a view from the mainland. In: Peltenburg E, Wasse A (eds) Neolithic Revolution. New Perspectives on Southwest Asia in Light of Recent Discoveries on Cyprus. Levant Supplementary Series 1. Oxbow Books, Oxford, pp 35–48
Irwin DM, Kocher TD, Wilson AC (1991) Evolution of the cytochrome b gene of mammals. J Mol Evol 32:128–144
IUCN/SSC Caprinae Specialist Group (2000) A summary of discussion on the taxonomy of mountain ungulates and its conservation implications (prepared by M. Festa-Bianchet). Workshop on Caprinae Taxonomy, Ankara, Turkey, May 8–10. Available on website: http://callisto.si.usherb.ca:8080/caprinae/taxo.htm
Jarman M (1996) Human influence in the development of the Cretan mammalian fauna. In: Reese DS (ed) Pleistocene and Holocene Fauna of Crete and Its First Settlers. Prehistory Press, Madison, WI, pp 211–229
Jarman MR, Jarman HN (1968) The fauna and economy of Early Neolithic Knossos. Ann Br Sch Athens 63:241–276
Kahila Bar-Gal, G (2000) Genetic changes in Capra species of Southern Levant over the past 10,000 years as studied by DNA analysis of ancient and modern populations. Unpublished PhD dissertation, The Hebrew University of Jerusalem
Bar-Gal KG, Smith P, Tchernov E, Greenblatt C, Docus P, Gardesein A, Horwitz LK (2002a) Genetic evidence for the origin of the Agrimi goat (Capra aegagrus cretica). J Zool Lond 256:369–377
Kahila Bar-Gal G, Khalaily H, Marder O, Ducos P, Horwitz LK (2002b) Ancient DNA evidence for the transition from wild to domestic status in Neolithic goats: a case study from the site of Abu Gosh, Israel. Anc Biomol 4:9–17
Kruska D, Röhrs M (1974) Comparative–quantitative investigations on brains of feral pigs from the Galapagos islands and of European domestic pigs. Z Anat Entwicklungsgesch 144:61–73
Lalueza-Fox C, Castresana J, Sampietro L, Marques-Bonet T, Alcover JA, Bertranpetit J (2005) Molecular dating of caprines using ancient DNA sequences of Myotragus balearicus, an extinct endemic Balearic mammal. BioMed Cent Evol Biol 5(70):1–11
Logan GT, Brown JH, Husband TP, Nicholson MC (1994) Conservation biology of the Cretan agrimi. Biol Gallo-Hell 22:241–246
Luikart G, Gielly L, Excoffier L, Vigne J-D, Bouvet J, Taberlet P (2001) Multiple maternal origins and weak phylogeographic structure in domestic goats. Proc Natl Acad USA 98:5927–5932
Manceau V, Despres L, Bouvet J, Taberlet P (1999) Systematics of the genus Capra inferred from mitochondrial DNA sequence data. Mol Phylogenet Evol 13:504–510
Mannen H, Nagata Y, Tsuji S (2001) Mitochondrial DNA reveal that domestic goat (Capra hircus) are genetically affected by two subspecies of bezoar (Capra aegagrus). Biochem Genet 39:145–154
Masseti M (1997) The prehistorical diffusion of the Asiatic mouflon, Ovis gmelini Blyth, 1841, and of the Bezoar goat, Capra aegagrus Erxleben, 1777, in the Mediterranean region beyond their natural distributions. In: Hadjisterkotis E (ed) Proceedings of the 2nd International Symposium on Mediterranean Mouflon, The Mediterranean Mouflon: Management, Genetics and Conservation. Ministry of Interior, Nicosia, pp 12–27
Masseti M (1998) Holocene endemic and anthropochorous wild mammals of the Mediterranean islands. Anthropozoology 28:3–20
Masseti M (2002) Mediterranean islands as natural enclosures. Poster presented at 9th ICAZ meeting, Durham, UK, 23rd–28th August
Masseti M (2003) Holocene endemic and non-endemic mammals of the Aegean islands. In: Katjabopoulou E, Hamilakis Y, Halstead P, Gamble C, Elefanti P (eds) Zooarchaeology in Greece. Recent Advances. Br Sch Athens Stud 9:53–63
Masseti M, Darlas A (1999) Pre-Neolithic man and other mammals on the Eastern Mediterranean islands. In: Cruz AR, Milliken S, Oosterbeek L, Peretto C (eds) Human Population Origins in the Circum-Mediterranean Area. Arkeos (5):pp 189–204
Masseti M, Trantalidou K (2002) Boars and goats in a Mesolithic fishing community: Youra (Northern Sporades, Greece), a case study. Poster presented at 9th ICAZ meeting, Durham, U.K., 23rd–28th August 2002
Mason IL (1984) Goats. In: Mason IL (ed) Evolution of Domestic Animals. Longman, London
Mavridis F (2003) Early island archaeology and the extinction of endemic fauna in the eastern Mediterranean: problems of interpretation and methodology. In: Kotjabopoulou E, Hamilakis Y, Halstead P, Gamble C, Elefanti P (eds) Zooarchaeology in Greece. Recent Advances. Br Sch Athens Stud 9:65–74
Nicholson MC, Husband TP (1992) Diurnal behaviour of the agrimi, Capra aegagrus. J Mamm 73(1):135–142
Nogales M, Martin A, Tershy BR, Donlan CJ, Veitch D, Puerta N, Woods B, Alonso J (2004) A review of feral cat eradication on islands. Conserv Biol 18:310–319
Nowak RM, Paradiso (1983) Walkers Mammals of the World, Vol II, 4th edn. The Johns Hopkins University Press, Baltimore
Nowak RM (1991) Walker’s Mammals of the World, Part 1. The Johns Hopkins University Press, Baltimore
Papageorgiou M (1974) Population energy relationships of the agrimi (Capra aegagrus cretica) on Theodorou Island, Greece. PhD dissertation, Ann Arbor Microfilms, Michigan
Pedrosa S, Uzun M, Arranz J-J, Gutiérrez-Gil B, San Primitivo F, Bayón Y (2005) Evidence of three maternal lineages in Near Eastern sheep supporting multiple domestication events. Proceedings of the Royal Society, Series B, 275: 2211–2217.
Poplin F (1979) Origine du Mouflon de Corse dans un nouvelle perspective: Par marronage. Ann Genet Sel Anim 11:133–143
Porter V (1996) The Cretan wild goat (Capra aegagrus cretica) and the ‘Theran antelopes’. In: Reese DS (ed) Pleistocene and Holocene Fauna of Crete and Its First settlers Madison. Prehistory Press, Wisconsin, pp 295–314
Randi E, Tosi G, Lorenzini R, Fusco G (1990) Genetic variability and conservation problems in Alpine inbex, domestic and feral goat populations (genus Capra). Z Säugetierkd 55:413–420
Sadler JP (1990) Beetles, boats and biogeography: insect invaders of the North Atlantic islands. Acta Archaeol 61:199–212
Saiki RD, Geelfand DH, Stoffel S, Scharf SJ, Higuchi R, Horn GT, Mullis KB, Erlich HA (1988) Primer-directed enzymatic amplification of DNA with Thermostable polymerase. Science 239:487–491
Sampson A (1998) The Neolithic and Mesolithic occupation of the Cave of Cyclope, Youra, Alonnesos, Greece. Annual of the British School at Athens, 94:1–22
Schultze-Westrum T (1963) Die wildziegen der ägäischen inseln. Saugetierkundl Mitt 11:145–182
Sondaar PY (1976) The island sweepstakes. Why did pygmy elephants, dwarf deer and large mice populate the Mediterranean? Nat Hist 95(9):50–57
Sultana S, Mannen H, Tsuji S (2003) Mitochondrial DNA diversity of Pakistani goats. Anim Genet 34:417–421
Swofford DL (1993) PAUP: Pylogenetic Analysis Using Parsimony Version 3.1.1. Illinois Natural History Survey, Champaign, IL
Takada T, Kikkawa Y, Yonekawa H, Kawakami S, Amano T (1997) Bezoar (Capra aegagrus) is a matriachal candidate for ancestor of domestic goat (Capra hircus): evidence from the mitochondrial DNA diversity. Biochem Genet 35:315–326
Valdez R (1982) The Wild Sheep of the World. Mesilla, New Mexico, pp 82–84
Van den Brink FH (1967) A Field Guide to the Mammals of Britain and Europe. Collins, London
Vigne J-D (1987) L’extinction Holocène du fond de peuplement mammalian indigène des îles de mediterranée occidentale. Mém Soc Géol Fr (New Series) 150:167–177
Vigne J-D (1988) Les mammiferes post-Glaciaires de corse. Étude archéozoologique. XXVI Supplement a Gallia Prehistoire. Editions du CNRS, Paris
Vigne J-D (1992) Zooarchaeology and biogeographical history of the mammals of Corsica and Sardinia since the last ice age. Mamm Rev 22(2):87–96
Vigne J-D (1999) The large “true” Mediterranean islands as a model for the Holocene human impact on the European vertebrate fauna? Recent data and new reflection. In: Benecke N (ed) The Holocene History of the European Vertebrate Fauna. Verlag Marie Leidorf, Rahden, pp 295–322
Vigne J-D (2002) Instabilite des premiers élevages Néolithiques: L’apport de la documentation insulaire Méditerranéenne. In: Maniers de faire...manieres de voir de l’object a l’Interpretation. hommage à noël pinzuti. XIe recontres culturelles interdisciplinaires. Publication du Musee de l’Alta Rocca, Editions Alain Piazzola, pp 77–84
Wilkens B (1996) Faunal remains from Italian excavations on crete. In: Reese DS (ed) Pleistocene and Holocene Fauna of Crete and Its First Settlers. Prehistory Press, Madison, WI, pp 241–254
Wilkins B, Delussu F (2002) Les mammiferes sauvages de la Sardaigne: Extinctions et nouvelles arrives au cours de l’Holocene. In: Gardeisen A (ed) Mouvements ou Déplacements de Population Animals en Méditerranée au cours de l’Holocène. BAR International Series 1017, Oxford, pp 23–31
Zimmerman K, Wettstein K, Stewert H, Pohle H (1942–1949). Die Wildsäuger von Kreta. Z Saugetierkd 17(1):1–72
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Horwitz, L.K., Bar-Gal, G.K. The Origin and Genetic Status of Insular Caprines in the Eastern Mediterranean: A Case Study of Free-Ranging Goats (Capra aegagrus cretica) on Crete. Human Evolution 21, 123–138 (2006). https://doi.org/10.1007/s11598-006-9015-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11598-006-9015-8