Abstract
Purpose
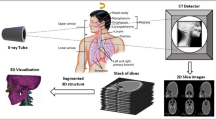
Accurate segmentation of the mandibular canal in cone beam CT data is a prerequisite for implant surgical planning. In this article, a new segmentation method based on the combination of anatomical and statistical information is presented to segment mandibular canal in CBCT scans.
Methods
Generally, embedding shape information in segmentation models is challenging. The proposed approach consists of three main steps as follows: At first, a method based on low-rank decomposition is proposed for preprocessing. Then, a conditional statistical shape model is trained, and mandibular bone is segmented with high accuracy. In the final stage, fast marching with a new speed function is utilized to find the optimal path between mandibular and mental foramen. Fast marching tries to find the darkest tunnel close to the initial segmentation of the canal, which was obtained with conditional SSM model. In this regard, localization of mandibular canal is performed more accurately.
Results
The method is applied to the identification of mandibular canal in 120 sets of CBCT images. Conditional statistical model is evaluated by calculating the compactness capacity, specificity and generalization ability measures. The capability of the proposed model is evaluated in the segmentation of mandibular bone and canal. The framework is effective in noisy scans and is able to detect canal in cases with mild bone resorption.
Conclusion
Quantitative analysis of the results shows that the method performed better than two other recent methods in the literature. Experimental results demonstrate that the proposed framework is effective and can be used in computer-guided dental implant surgery.











Similar content being viewed by others
Notes
Peak Signal-to-Noise Ratio.
References
Roberts JA, Drage NA, Davies J, Thomas DW (2014) Effective dose from cone beam CT examinations in dentistry. Br J Radiol 82(973):35–40
Robinson PP (1988) Observations on the recovery of sensation following inferior alveolar nerve injuries. Br J Oral Maxillofac Surg 26:177–189
Stein W, Hassfeld S, Muhling J (1998) Tracing of thin tubular structures in computer tomographic data. Comput Aided Surg 3:83–88
Hanssen N, Burgielski Z, Jansen T, Liévin M, Ritter L, von Rymon-Lipinski B, Keeve E (2004) Nerves-level sets for interactive 3D segmentation of nerve channels. In: IEEE international symposium on biomedical imaging nano macro 2004. IEEE, pp 201–204
Kondo T, Ong SH, Foong KW (2004) Computer-based extraction of the inferior alveolar nerve canal in 3-D space. Comput Methods Programs Biomed 76:181–191
Rueda S, Gil JA, Pichery R, Alcañiz M (2006) Automatic segmentation of jaw tissues in CT using active appearance models and semi-automatic landmarking. In: Larsen R, Nielsen M, Sporring J (eds) Medical image computing and computer-assisted intervention 2006. Springer, Heidelberg, pp 167–174
Yau HT, Lin YK, Tsou LS, Lee CY (2008) An adaptive region growing method to segment inferior alveolar nerve canal from 3D medical images for dental implant surgery. Comput Aided Des Appl 5:743–752
Orth RC, Wallace MJ, Kuo MD (2008) C-arm cone-beam CT: general principles and technical considerations for use in interventional radiology. J Vasc Interv Radiol 19:814–820
Kainmueller D, Lamecker H, Seim H, Zinser M, Zachow S (2009) Automatic extraction of mandibular nerve and bone from cone-beam CT data. In: Yang G-Z, Hawkes D, Rueckert D, Noble A, Taylor C (eds) Medical image computing and computer-assisted intervention 2009. Springer, Heidelberg, pp 76–83
Lloréns R, Naranjo V, López F, Alcañiz M (2012) Jaw tissues segmentation in dental 3D CT images using fuzzy-connectedness and morphological processing. Comput Methods Programs Biomed 108:832–843
Kim G, Lee J, Lee H, Seo J, Koo Y-M, Shin Y-G, Kim B (2011) Automatic extraction of inferior alveolar nerve canal using feature-enhancing panoramic volume rendering. IEEE Trans Biomed Eng 58:253–264
Gerlach NL, Meijer GJ, Kroon D-J, Bronkhorst EM, Bergé SJ, Maal TJJ (2014) Evaluation of the potential of automatic segmentation of the mandibular canal using cone-beam computed tomography. Br J Oral Maxillofac Surg 52:838–844
Styner MA, Rajamani KT, Nolte L-P, Zsemlye G, Székely G, Taylor CJ, Davies RH (2003) Evaluation of 3D correspondence methods for model building. In: Biennial international conference on information processing in medical imaging. Springer, pp 63–75
Roohi SF, Zoroofi RA (2013) 4D statistical shape modeling of the left ventricle in cardiac MR images. Int J Comput Assist Radiol Surg 8:335–351
Gerlach NL, Meijer GJ, Maal TJ, Mulder J, Rangel FA, Borstlap WA, Bergé SJ (2010) Reproducibility of 3 different tracing methods based on cone beam computed tomography in determining the anatomical position of the mandibular canal. J Oral Maxillofac Surg 68:811–817
Ong F, Lustig M (2016) Beyond low rank + sparse: multi-scale low rank matrix decomposition. IEEE J Sel Top Sign Proces 10(4):672–687
Wackerly D, Mendenhall W, Scheaffer RL (2008) Mathematical statistics with applications, 7th edn. Thomson Brooks/Cole, Scarborough
Wang Z, Bovik AC, Sheikh HR, Simoncelli EP (2004) Image quality assessment: from error visibility to structural similarity. IEEE Trans Image Process 13:600–612
Lorensen WE, Cline HE (1987) Marching cubes: a high resolution 3D surface construction algorithm. In: Stone MC (ed) ACM siggraph computer graphics. ACM, New York, pp 163–169
Rueckert D, Sonoda LI, Hayes C, Hill DL, Leach MO, Hawkes DJ (1999) Nonrigid registration using free-form deformations: application to breast MR images. IEEE Trans Med Imaging 18:712–721
Yokota F, Okada T, Takao M, Sugano N, Tada Y, Tomiyama N, Sato Y (2013) Automated CT segmentation of diseased hip using hierarchical and conditional statistical shape models. In: Mori K, Sakuma I, Sato Y, Barillot C, Navab V (eds) Medical image computing and computer-assisted intervention 2013. Springer, Heidelberg, pp 190–197
Yokota F, Okada T, Takao M, Sugano N, Tomiyama N, Sato Y, Tada Y (2012) Automated localization of pelvic anatomical coordinate system from 3D CT data of the hip using statistical atlas. Med Imaging Technol 30:43–52
Lamecker H, Lange T, Seebaß M (2004) Segmentation of the liver using a 3D statistical shape model. Konrad-Zuse-Zentrum für Informationstechnik, Apr 2
Sethian JA (1996) A fast marching level set method for monotonically advancing fronts. Proc Natl Acad Sci 93:1591–1595
Sethian JA (1999) Fast marching methods. SIAM Rev 41:199–235
Atkinson KE (2008) An introduction to numerical analysis. Wiley, London
MATLAB R2015a—Die Sprache für technische Berechnungen—MathWorks Deutschland (n.d.). http://de.mathworks.com/products/matlab/
Visual Studio 2013—Microsoft Developer Tools (n.d.). https://www.visualstudio.com/
Atkinson KE (2008) An introduction to numerical analysis. John Wiley & Sons
Training and Testing Data Sets (n.d.). https://msdn.microsoft.com/en-us/library/bb895173.aspx (2016)
Kroon D-J, Slump CH, Maal TJ (2010) Optimized anisotropic rotational invariant diffusion scheme on cone-beam CT. In: Jiang T, Navab N, Pluim JPW, Viergever MA (eds) Medical image computing and computer-assisted intervention 2010. Springer, Heidelberg, pp 221–228
Cootes TF, Edwards GJ, Taylor CJ (1998) Active appearance models. In: Computer vision—ECCV’98. Springer, pp 484–498
Abdolali F, Zoroofi RA, Otake Y, Sato Y (2016) Automatic segmentation of maxillofacial cysts in cone beam CT images. Comput Biol Med 72:108–119
Brechbühler C, Gerig G, Kübler O (1995) Parametrization of closed surfaces for 3-D shape description. Comput Vis Image Underst 61:154–170
Rissanen J (1978) Modeling by shortest data description. Automatica 14:465–471
Varadarajan VS (2013) Lie groups, Lie algebras, and their representations. Springer, Berlin
Davies R, Taylor C et al (2008) Statistical models of shape: optimisation and evaluation. Springer, Berlin
Kohavi R et al (1995) A study of cross-validation and bootstrap for accuracy estimation and model selection. In: Ijcai, pp 1137–1145
Dice LR (1945) Measures of the amount of ecologic association between species. Ecology 26:297–302
Heimann T, Van Ginneken B, Styner MA, Arzhaeva Y, Aurich V, Bauer C, Beck A, Becker C, Beichel R, Bekes G et al (2009) Comparison and evaluation of methods for liver segmentation from CT datasets. IEEE Trans Med Imaging 28:1251–1265
Acknowledgments
This work is partly supported by MEXT/JSPS Grant-in-Aid for Scientific Research Nos. 26108004 and 25242051.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights
For this type of study formal consent is not required because this study is a retrospective study.
Informed consent
Written informed consent was not required for this study because this study is a retrospective study.
Appendix A: Performance evaluation metrics
Appendix A: Performance evaluation metrics
To investigate the statistical behavior of conditional SSM, we utilize compactness, specificity and generalization measures. Compactness is an estimation of parameters required to generate a valid instance of the modeled object [37]. The compactness of the shape model is calculated as the cumulated variance for the first \(i=1,\ldots ,M\) modes:
where \(C(\tau )\) and \({\lambda }_i \) are the compactness capacity and i-th largest eigenvalue, respectively. Generalization of a model measures the ability to represent unseen instances of the object class modeled [37], and it is defined as follows:
where \(N_s \) is the number of training data, \(t_k \) is the training sample that is eliminated in leave-one-out procedure, and \(r_k \) is the reconstructed shape using \({\tau }\) parameters. The specificity of a shape model is described as how much it can represent valid instances of the modeled class of object [37] and it is formulated as following:
where \(s_k (\uptau )\) is an arbitrary sample constructed by \(\uptau \) parameters, \(N_r \) is the number of data, and \({t}^{\prime }_k \) is the closest sample in training datasets to \(s_k (\uptau )\). Leave-one-out crossvalidation [38] is employed to compare the segmented mandible bone with the gold standard. To create the gold standard dataset, each mandible and the mandibular canal was manually segmented by two radiologists in a slice-by-slice fashion. In order to compare the automatic segmentation results with the gold standard, two criteria are employed: (1) Dice’s coefficient [39] and (2) average symmetric surface distance [40]. Dice’s coefficient measures the overlap between the automatic segmentation result and reference manual annotations. This similarity measure is defined as following:
where A and M are segmentation results obtained by automatic segmentation and gold standard, respectively. This criterion is one of the most well-known methods in evaluating different segmentation methods.
Average symmetric surface distance (ASSD) [40] is defined as the space between two segmentations A and M in millimeters. If we assume that \(S_\mathrm{A}\) and \(S_{\mathrm{M}}\) are surface voxels of A and M, the Euclidean distance for each surface voxel of \(S_{\mathrm{A}}\) to the closest surface voxel of \(S_{\mathrm{M} }\) is calculated. To preserve symmetry, the same process is applied for the surface voxels of \(S_{\mathrm{M}}\) to \(S_{\mathrm{A}}\). Therefore, ASSD is expressed as the average of all stored distances as follows:
where d is the shortest distance of voxel v to surface S, \(\left\| . \right\| \) and \(\left| . \right| \) represent vector norm and number of vertices, respectively. ASSD provides a volumetric-based evaluation criterion for the assessment of segmentation result.
Rights and permissions
About this article
Cite this article
Abdolali, F., Zoroofi, R.A., Abdolali, M. et al. Automatic segmentation of mandibular canal in cone beam CT images using conditional statistical shape model and fast marching. Int J CARS 12, 581–593 (2017). https://doi.org/10.1007/s11548-016-1484-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-016-1484-2