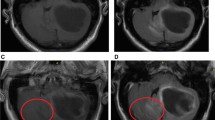
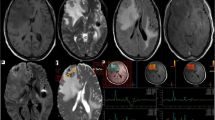
Abstract
Purpose
This study was done to investigate the usefulness of diffusion-weighted (DWI), perfusion-weighted (PWI) and proton magnetic resonance (MR) spectroscopy imaging in characterising solitary brain metastases.
Materials and methods
Fifty-nine solitary brain metastases were evaluated with conventional and nonmorphological MR imaging: DWI, PWI and MR spectroscopy. We evaluated size, signal intensity and contrast enhancement and calculated apparent diffusion coefficient (ADC), relative cerebral blood volume (rCBV), percentage of signal intensity recovery (PSR) and maximum values of N-acetylaspartate (NAA), choline (Cho), creatine (Cr), lipids (Lip), NAA/Cr and Cho/Cr. The nonmorphological parameters were compared with those from the literature for brain lesions that frequently enter the differential diagnosis with metastases.
Results
Signal intensity and contrast enhancement patterns were variable. There was a wide range of ADC values: min:max 0.59×10−3:1.88×10−3. Compared with normal white matter, rCBV was higher in lesions (3.30±1.59) and lower in perilesional oedema (0.42±0.15). Mean and minimum PSR were 57% and 48%, respectively; lip and Cho were elevated and NAA reduced.
Conclusions
Conventional MR findings of solitary metastases are heterogeneous, and some values of nonmorphological sequences are similar to those of other brain lesions. PWI seems to be the nonmorphological MR technique that may best contribute to the diagnosis of brain metastases.
Riassunto
Obiettivo
Scopo del presente lavoro è stato valutare l’utilità dell’imaging di diffusione (DWI), perfusione (PWI) e spettroscopia (MRS) nella caratterizzazione delle metastasi cerebrali solitarie.
Materiali e metodi
Abbiamo studiato 59 metastasi cerebrali uniche con sequenze di risonanza magnetica (RM) morfologiche e non morfologiche: DWI, PWI e MRS. Abbiamo valutato dimensioni, caratteristiche di segnale e contrast enhancement (CE), calcolato coefficiente di diffusione apparente (ADC), volume cerebrale ematico relativo (rCBV), percentuale di recupero della curva (RC), valori massimi di N-acetil-aspartato (NAA), colina (Cho), creatina (Cr), lipidi (lip), NAA/Cr, Cho/Cr. I parametri non morfologici sono stati confrontati con quelli della letteratura per le lesioni che frequentemente entrano in diagnosi differenziale con le metastasi.
Risultati
Caratteristiche di segnale e pattern di CE sono risultati variabili. Ampio il range di valori di ADC: minmax 0,59×10−3-1,88×10−3. Rispetto alla sostanza bianca normale il rCBV nelle lesioni era superiore (3,30±1,59) e nell’edema perilesionale inferiore (0,42±0,15). La percentuale media e minima di RC erano 57% e 48%. Sono risultati aumentati lip e Cho, ridotto l’NAA.
Conclusioni
Gli aspetti RM morfologici delle metastasi solitarie risultano variabili ed alcuni valori delle metodiche non morfologiche sovrapponibili a quelli di altre lesioni encefaliche. La perfusione sembra la metodica non morfologica RM in grado di contribuire più significativamente alla diagnosi di metastasi encefalica.
Similar content being viewed by others
References/Bibliografia
Nathoo N, Toms SA, Barnett GH (2004) Metastases to the brain: current management perspectives. Expert Rev Neurother 4:633–640
Barnholtz-Sloan JS, Sloan AE, Davis FG et al (2004) Incidence proportions of brain metastases in patients diagnosed (1973–2001) in the Metropolitan Detroit Cancer Surveillance System. J Clin Oncol 22:2865–2872
Gavrilovic IT, Posner JB (2005) Brain metastases: epidemiology and pathophysiology. J Neurooncol 75:5–14
Kamar FG, Posner JB (2010) Brain metastases. Semin Neurol 30:217–235
Posner JB, Chernik NL (1978) Intracranial metastases from systemic cancer. Adv Neurol 19:579–592
Le Chevalier T, Smith FP, Caille P (1985) Sites of primary malignancies in patients presenting with cerebral metastases: a review of 120 cases. Cancer 56:880–882
Kehrli P (1999) Epidemiology of brain metastases. Neurochirurgie 45:357–363
Harrison LE, Brennan MF, Newman E et al (1997) Hepatic resection for noncolorectal, non-neuroendocrine metastases: a fifteen-year experience with ninety-six patients. Surgery 121:625–632
Larson DA, Rubenstrein JL, McDermott MW (2005) Metasatic brain cancer. In: DeVita VT, Hellman S, Rosenberg SA (eds). Cancer, principles & practice of oncology. Lippincott Williams & Wilkins, Philadelphia, PA, pp 2328–2343
Jung S, Kim HW, Lee JH et al (2002) Brain tumor invasion model system using organotypic brain-slice culture as an alternative to in vivo model. J Cancer Res Clin Oncol 128:269–476
Falini A, Giovanna C, Origgi D et al (1996) Proton magnetic spectroscopy and intracranial tumors: clinical perspectives. J Neurology 243:706–714
Sijens PE, Knopp MV, Brunetti A et al (1995) 1H MR spectroscopy in patients with metastatic brain tumors: a multicenter study. Magn Reson Med 33:818–826
Poptani H, Gupta RK, Roy R et al (1995) Characterization of intracranial mass lesions with in vivo proton MR spectroscopy. AJNR Am J Neuroradiol 16:1593–1603
De Edelenyi FS, Rubin C, Esteve F et al (2000) A new approach for analyzing proton magnetic resonance spectroscopic images of brain tumors: nosologic images. Nat Med 6:1287–1289
Law M, Cha S, Knopp EA et al (2002) High-grade gliomas and solitary metastases: differentiation by using perfusion and proton spectroscopic MR imaging. Radiology 222:715–721
Brem SS, Bierman PJ, Brem H et al (2011) Central nervous system cancers. J Natl Compr Canc Netw 9:352–400
Niwińska A, Tacikowska M, Murawska M (2010) The effect of early detection of occult brain metastases in HER2-positive breast cancer patients on survival and cause of death. Int J Radiat Oncol Biol Phys 77:1134–1139
Oshiro S, Tsugu H, Komatsu F et al (2008) Metastatic adenocarcinoma in the brain: magnetic resonance imaging with pathological correlations to mucin content. Anticancer Res 28:407–413
Isiklar I, Leeds NE, Fuller GN, Kumar AJ (1995) Intracranial metastatic melanoma: correlation between MR imaging characteristics and melanin content. AJR Am J Roentgenol 165:1503–1512
Law M (2009) Advanced imaging techniques in brain tumors. Cancer Imaging 9(Spec No A):S4–S9
Hayashida Y, Hirai T, Morishita S et al (2006) Diffusion-weighted imaging of metastatic brain tumors: comparison with histologic type and tumor cellularity. AJNR Am J Neuroradiol 27:1419–1425
Duygulu G, Ovali GY, Calli C et al (2010) Intracerebral metastasis showing restricted diffusion: correlation with histopathologic findings. Eur J Radiol 4:117–120
Server A, Kulle B, Maehlen J (2009) Quantitative apparent diffusion coefficients in the characterization of brain tumors and associated peritumoral edema. Acta Radiol 50:682–689
Rizzo L, Crasto SG, Moruno PG (2009) Role of diffusion- and perfusionweighted MR imaging for brain tumour characterisation. Radiol Med 114:645–659
Bink A, Gaa J, Franz K et al (2005) Importance of diffusion-weighted imaging in the diagnosis of cystic brain tumors and intracerebral abscesses. Zentralbl Neurochir 66:119–125
Wetzel SG, Cha S, Johnson G et al (2002) Relative cerebral blood volume measurements in intracranial mass lesions: interobserver and intraobserver reproducibility study. Radiology 224:797–803
Sorensen AG, Reimer P (2001) Imaging RM di perfusione cerebrale. Principi ed applicazioni correnti. CIC Edizioni Internazionali, Roma
Boxerman JL, Schmainda KM, Weisskoff RM (2006) Relative cerebral blood volume maps corrected for contrast agent extravasation significantly correlate with glioma tumor grade, whereas uncorrected maps do not. AJNR Am J Neuroradiol 27:859–867
Uematsu H, Maeda M (2006) Double echo perfusion-weighted MR imaging: basic concepts and application in brain tumors for the assessment of tumor blood volume and vascular permeability. Eur Radiol 16:180–186
Chan JH, Tsui EY, Chau LF (2002) Discrimination of an infected brain tumor from a cerebral abscess by combined MR perfusion and diffusion imaging. Comput Med Imaging Graph 26:19–23
Cha S, Knopp EA, Johnson G et al (2002) Intracranial mass lesions: dynamic contrast-enhanced susceptibility-weighted echo-planar perfusion MR imaging. Radiology 223:11–29
Hakyemez B, Erdogan C, Bolca N et al (2006) Evaluation of different cerebral mass lesions by perfusion-weighted MR imaging. J Magn Reson Imaging 24:817–824
Zonari P, Baraldi P, Crisi G (2007) Multimodal MRI in the characterization of glial neoplasms: the combined role of single-voxel MR spectroscopy, diffusion imaging and echo-planar perfusion imaging. Neuroradiology 49:795–803
Mangla R, Kolar B, Zhu T et al (2011) Percentage signal recovery derived from MR dynamic susceptibility contrast imaging is useful to differentiate common enhancing malignant lesions of the brain. AJNR Am J Neuroradiol 32:1004–1010
Hossman KA, Blöink M (1981) Blood flow and regulation of blood flow in experimental peritumoral edema. Stroke 12:211–217
Uematsu H, Maeda M, Itoh H (2003) Peritumoral brain edema in intracranial meningiomas evaluated by dynamic perfusion-weighted MR imaging: a preliminary study. Eur Radiol 13:758–762
Wang S, Kim S, Chawla S et al (2011) Differentiation between glioblastomas, solitary brain metastases, and primary cerebral lymphomas using diffusion tensor and dynamic susceptibility contrast-enhanced MR imaging. AJNR Am J Neuroradiol 32:507–514
Server A, Orheim TE, Graff BA et al (2011) Diagnostic examination performance by using microvascular leakage, cerebral blood volume, and blood flow derived from 3-T dynamic susceptibility-weighted contrastenhanced perfusion MR imaging in the differentiation of glioblastoma multiforme and brain metastasis. Neuroradiology 53:319–330
Chawla S, Zhang Y, Wang S et al (2010) Proton magnetic resonance spectroscopy in differentiating glioblastomas from primary cerebral lymphomas and brain metastases. J Comput Assist Tomogr 34:836–841
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gaudino, S., Di Lella, G.M., Russo, R. et al. Magnetic resonance imaging of solitary brain metastases: main findings of nonmorphological sequences. Radiol med 117, 1225–1241 (2012). https://doi.org/10.1007/s11547-012-0846-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-012-0846-2