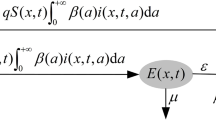
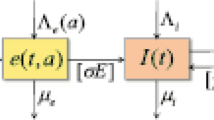Abstract
Demographic structure and latent phenomenon are two essential factors determining the rate of tuberculosis transmission. However, only a few mathematical models considered age structure coupling with disease stages of infectious individuals. This paper develops a system of delay partial differential equations to model tuberculosis transmission in a heterogeneous population. The system considers demographic structure coupling with the continuous development of disease stage, which is crucial for studying how aging affects tuberculosis dynamics and disease progression. Here, we determine the basic reproduction number, and several numerical simulations are used to investigate the influence of various progression rates on tuberculosis dynamics. Our results support that the aging effect on the disease progression rate contributes to tuberculosis permanence.













Similar content being viewed by others
References
Aagaard C, Hoang T, Dietrich J, Cardona P, Izzo A, Dolganov G, Schoolnik G, Cassidy J, Billeskov R, Andersen P (2011) A multistage tuberculosis vaccine that confers efficient protection before and after exposure. Nat Med 17(2):189
Anderson R, Trewhella W (1985) Population dynamics of the badger (Meles meles) and the epidemiology of bovine tuberculosis (Mycobacterium bovis). Philos Trans RSoc B Biol Sci 310(1145):327–381
Arregui S, Iglesias M, Samper S, Marinova D, Martin C, Sanz J, Moreno Y (2018) Data-driven model for the assessment of mycobacterium tuberculosis transmission in evolving demographic structures. P Natl Acad Sci USA 115(14):E3238–E3245
Barry C 3rd, Boshoff H, Dartois V, Dick T, Ehrt S, Flynn J, Schnappinger D, Wilkinson R, Young D (2009) The spectrum of latent tuberculosis: rethinking the biology and intervention strategies. Nat Rev Microbiol 7(12):845
Blower S, Mclean A, Porco T, Small P, Hopewell P, Sanchez M, Moss A (1995) The intrinsic transmission dynamics of tuberculosis epidemics. Nat Med 1(8):815
Cai J, Wang X, Ma A, Wang Q, Han X, Li Y (2015) Factors associated with patient and provider delays for tuberculosis diagnosis and treatment in asia: a systematic review and meta-analysis. PloS one 10(3):e0120088
Cardona P (2016) Reactivation or reinfection in adult tuberculosis: Is that the question? Int J Mycobacteriol 5(4):400–407
Cardona P, Ruiz-Manzano J (2004) On the nature of mycobacterium tuberculosis-latent bacilli. Eur Respir J 24(6):1044–1051
Castillo-Chavez C, Feng Z (1998) Global stability of an age-structure model for TB and its applications to optimal vaccination strategies. Math Biosci 151(2):135–154
Castillo-Chavez C, Song B (2004) Dynamical models of tuberculosis and their applications. Math Biosci Eng 1(2):361–404
Census and Statistics Department (2001) 2001 population by-census main report: volume I. Hong Kong Census and Statistics Department
Census and Statistics Department of the Government of the Hong Kong SAR (2018) Fertility trend in hong kong, 1981 to 2017. https://www.censtatd.gov.hk/hkstat/sub/sp160.jsp?productCode=FA100090
Census and Statistics Department of the Government of the Hong Kong SAR: Statistics on population (2018) https://www.censtatd.gov.hk/hkstat/sub/so20.jsp
Chan-Yeung M, Noertjojo K, Tan J, Chan S, Tam C (2002) Tuberculosis in the elderly in hong kong. Int J Tuberc Lung D 6(9):771–779
Cohen T, Colijn C, Finklea B, Murray M (2006) Exogenous re-infection and the dynamics of tuberculosis epidemics: local effects in a network model of transmission. J R Soc Interface 4(14):523–531
Department of Health of the Government of the Hong Kong SAR: Statistics on tuberculosis (1995-2018). https://www.info.gov.hk/tb_chest/en/index_2.htm (2018)
Feng Z, Castillo-Chavez C, Capurro A (2000) A model for tuberculosis with exogenous reinfection. Theor Popul Biol 57(3):235–247
Feng Z, Huang W, Castillo-Chavez C (2001) On the role of variable latent periods in mathematical models for tuberculosis. J Dyn Differ Equ 13(2):425–452
Feng Z, Iannelli M, Milner F (2002) A two-strain tuberculosis model with age of infection. SIAM J Appl Math 62(5):1634–1656
Flynn J, Chan J (2001) Tuberculosis: latency and reactivation. Infec Immun 69(7):4195–4201
Houben R, Dodd P (2016) The global burden of latent tuberculosis infection: a re-estimation using mathematical modelling. PLoS Med 13(10):e1002152
Jabbari A, Castillo-Chavez C, Nazari F, Song B, Kheiri H (2016) A two-strain tb model with multiple latent stages. Math Biosci Eng 13:741–785
Kapitanov G (2015) A double age-structured model of the co-infection of tuberculosis and HIV. Math Biosci Eng 12(1):23–40
Kasaie P, Andrews J, Kelton W, Dowdy D (2014) Timing of tuberculosis transmission and the impact of household contact tracing. An agent-based simulation model. Am J Resp Crit Care 189(7):845–852
Kirschner D, Pienaar E, Marino S, Linderman J (2017) A review of computational and mathematical modeling contributions to our understanding of mycobacterium tuberculosis within-host infection and treatment. Curr Opin Syst Biol 3:170–185
Kuniya T (2011) Global stability analysis with a discretization approach for an age-structured multigroup SIR epidemic model. Nonlinear Anal-Real 12(5):2640–2655
Liu Q, Jiang D, Hayat T, Alsaedi A (2018) Dynamics of a stochastic tuberculosis model with antibiotic resistance. Chaos Soliton Fract 109:223–230
Ma J, Teng X, Wang X, Fan X, Wu Y, Tian M, Zhou Z, Li L (2017) A multistage subunit vaccine effectively protects mice against primary progressive tuberculosis, latency and reactivation. EBioMedicine 22:143–154
Macdonald N (1978) Time lags in biological models. In: Lecture notes in biomathmatics, Springer-Verlag, Heidelberg
Makinodan T, Kay M (1980) Age influence on the immune system. Adv Immunol 29:287–330
Marais B, Gie R, Schaaf H, Hesseling A, Obihara C, Starke J, Enarson D, Donald P, Beyers N (2004) The natural history of childhood intra-thoracic tuberculosis: a critical review of literature from the pre-chemotherapy era [state of the art]. Int J Tuberc Lung D 8(4):392–402
Mossong J, Hens N, Jit M, Beutels P, Auranen K, Mikolajczyk R, Massari M, Salmaso S, Tomba G, Wallinga J (2008) Social contacts and mixing patterns relevant to the spread of infectious diseases. PLoS Med 5(3):e74
Nahid P, Hopewell P (2017) Tuberculosis treatment. In: International encyclopedia of public health, 2nd edn. Academic Press, pp 267–276
Okuonghae D, Omosigho S (2011) Analysis of a mathematical model for tuberculosis: what could be done to increase case detection. J Theor Biol 269(1):31–45
Parrish N, Dick J, Bishai W (1998) Mechanisms of latency in mycobacterium tuberculosis. Trends Microbiol 6(3):107–112
Renardy M, Kirschner D (2020) A framework for network-based epidemiological modeling of tuberculosis dynamics using synthetic datasets. Bull Math Biol 82(6):1–20
Selwyn P, Hartel D, Lewis V, Schoenbaum E, Vermund S, Klein R, Walker A, Friedland G (1989) A prospective study of the risk of tuberculosis among intravenous drug users with human immunodeficiency virus infection. New Engl J Med 320(9):545–550
Storla D, Yimer S, Bjune G (2008) A systematic review of delay in the diagnosis and treatment of tuberculosis. BMC Public Health 8(1):15
Styblo K (1991) Epidemiology of tuberculosis, 2nd edn. Royal Netherlands Tuberculosis Association, The Hague
Væth M, Skriver M, Støvring H (2018) The impact of proportional changes in age-specific mortality on life expectancy when mortality is a log-linear function of age. Demogr Res 39:671–684
Vynnycky E, Fine P (1997) The natural history of tuberculosis: the implications of age-dependent risks of disease and the role of reinfection. Epidemiol Infect 119(2):183–201
Vynnycky E, Fine P (2000) Lifetime risks, incubation period, and serial interval of tuberculosis. Am J Epidemiol 152(3):247–263
Waaler H, Geser A, Andersen S (1962) The use of mathematical models in the study of the epidemiology of tuberculosis. Am J Public Health 52(6):1002–1013
World Health Organization (2006) Regional Office for the Eastern Mediterranean: diagnostic and treatment delay in tuberculosis. World Health Organization
World Health Organization (2018) Global tuberculosis report 2018. World Health Organization
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
A. Numerical Scheme
We first define a function Z(t, a) as
We rewrite system (1) into the following system
The initial and boundary conditions are
We first discretize the space of time, age and stage into mesh as followed
where \(\varDelta t, \varDelta a, \varDelta s\) are the step sizes of the time, age and stage, respectively. We suppose that all the age densities distribute in the interval \([0,a_{\max }]\) and all the stage densities distribute in the interval \([0,s_{\max }]\) and \(t\in [0,T]\), where T is the time span of the prediction period for the disease transmission. We have the following equalities for \(N, L_1, L_2\)
that is, \(N, L_1, L_2\) are the number of steps from 0 to T, \(a_{\max }\) and \(s_{\max }\) separately.
Here, we simulate the dynamics of system in the population with stationary structure and hence we suppose that the immigration rate and the birth rate are only related with age in the following simulations, that is, \(\beta (t,a) = \beta (a)\). Using i, j, k represent \(t_i, a_j,s_k\) for simplification, we have the following identities for the boundary conditions
where \(\beta _0\) is a positive constant representing the birth rate of the population. On the basis of the finite difference method for derivative and the trapezoidal rule for approximating integral, we discretize system (A.10) as follows.
Here,
For \(i = 0\), we utilize the initial epidemic data
For \(i = 1\), we do the following
-
1.
Calculating \(I^1_{j,0}\) for all j with
$$\begin{aligned} I^1_{j,0} = S^0_jG^0_j, \end{aligned}$$and
$$\begin{aligned} Z^1_j = I^1_{j,0}. \end{aligned}$$ -
2.
Calculating \(S^1_j\) for all j with formula (A.11) and \(I^1_{j,k}\) for all k with formula (A.12).
For \(2\le i\le N\), we do the following
-
1.
Calculating \(S^i_j\), \(I^i_{j,k}\) with the formula in (A.11) and (A.12) separately.
-
2.
Calculating \(Z^i_j\) with formula (A.13) and then replacing \(I^i_{j,0}\) with \(Z^i_j\), that is, \(I^i_{j,0} = Z^i_j\) for the boundary condition of infectious individuals.
In our numerical simulations, we set \(\varDelta t = 0.1\) and \(\varDelta a =\varDelta s = 0.01\).
Rights and permissions
About this article
Cite this article
Mu, Y., Chan, TL., Yuan, HY. et al. Transmission Dynamics of Tuberculosis with Age-specific Disease Progression. Bull Math Biol 84, 73 (2022). https://doi.org/10.1007/s11538-022-01032-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-022-01032-4