Abstract
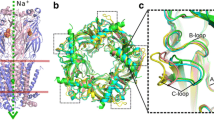
Compared with traditional structure-based approaches for the identification of species-specific ligands, the ab initio approach, based on large-scale protein sequences from different species, has been used to locate specific sites that may be important to the molecular selectivity of species. Statistically significant differences in the distribution of residues in different species and differences in the physicochemical properties of residue-specific sites may largely account for species selectivity. The nicotinic acetylcholine receptor (nAChR), an important neuro-receptor with significantly different ligand selectivity in different species, was used to test our method. Because of the lack of nAChR structural information, the mechanism of ligand discrimination is unclear which hinders attempts at novel molecular design. In this study, the specific site residues 186 and 189 in the principal subunits and residues 34, 55, 56, 57, 106 and 112 in complementary subunits of nAChR were identified by applying our method with stringent statistical cutoffs. These sites were predicted to contribute to ligand selectivity and this result coincides well with the known experimental data.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Blouin C, Boucher Y, Roger A J. Inferring functional constraints and divergence in protein families using 3D mapping of phylogenetic information. Nucleic Acids Res, 2003, 31: 790–797
Pang P S, Jankowsky E, Wadley L M, et al. Prediction of functional tertiary interactions and intermolecular interfaces from primary sequence data. J Exp Zool B Mol Dev Evol, 2005, 304: 50–63
Atchley W R, Zhao J. Molecular architecture of the DNA-binding region and its relationship to classification of basic helix-loop-helix proteins. Mol Biol Evol, 2007, 24: 192–202
Millar N S. Assembly and subunit diversity of nicotinic acetylcholine receptors. Biochem Soc Trans, 2003, 31: 869–874
Clementi F, Fornasari D, Gotti C. Neuronal Nicotinic Recetpors. In: Born G V R, Ganten D, Herken H, et al., eds. Handbook of Experimental Pharmacology. Heidelberg: Springer-Verlag, 2000
Zouridakis M, Zisimopoulou P, Poulas K, et al. Recent advances in understanding the structure of nicotinic acetylcholine receptors. IUBMB Life, 2009, 61: 407–423
Thany S H. Insect nicotinic acetylcholine receptors. In: Thany S H, ed. Advances in Experimental Medicine and Biology. New York: Springer Science+Business Media, LLC, 2010
Liu Z W, Yao X M, Zhang Y X. Insect nicotinic acetylcholine receptors (nAChRs): Important amino acid residues contributing to neonicotinoid insecticides selectivity and resistance. Afr J Biotechnol, 2008, 7: 4935–4939
Sattelle D B. Invertebrate nicotinic acetylcholine receptors-targets for chemicals and drugs important in agriculture, veterinary medicine and human health. J Pestic Sci, 2009, 34: 233–240
Matsuda K, Sattelle D B. Mechanism of selective actions of neonicotinoids on insect nicotinic acetylcholine receptors. In: Clark J M, Ohkawa H, eds. New Discoveries in Agrochemicals. Washington DC: American Chemical Society Publishers, 2005. 172–182
Tomizawa M, Lee D L, Casida J E. Neonicotinoid insecticides: Mo lecular features conferring selectivity for insect versus mammalian nicotinic receptors. J Agric Food Chem, 2000, 48: 6016–6024
Matsuda K, Shimomura M, Ihara M, et al. Neonicotinoids show selective and diverse actions on their nicotinic receptor targets: Electrophysiology, molecular biology, and receptor modeling studies. Biosci Biotechnol Biochem, 2005, 69: 1442–1452
Matsuda K, Buckingham S D, Kleier D, et al. Neonicotinoids: Insecticides acting on insect nicotinic acetylcholine receptors. Trends Pharmacol Sci, 2001, 22: 573–580
Narahashi T. Neuronal ion channels as the target sites of insecticides. Pharmacol Toxicol, 1996, 79: 1–14
Tomizawa M, Casida J E. Neonicotinoid insecticide toxicology: Mechanisms of selective action. Annu Rev Pharmacol Toxicol, 2005, 45: 247–268
Unwin N. Refined structure of the nicotinic acetylcholine receptor at 4A resolution. J Mol Biol, 2005, 346: 967–989
Breer H, Sattelle D B. Molecular properties and functions of insect acetylcholine receptors. J Insect Physiol, 1987, 33: 771–790
Celie P H, van Rossum-Fikkert S E, van Dijk W J, et al. Nicotine and carbamylcholine binding to nicotinic acetylcholine receptors as studied in AChBP crystal structures. Neuron, 2004, 41: 907–914
Brejc K, van Dijk W J, Klaassen R V, et al. Crystal structure of an ACh-binding protein reveals the ligand-binding domain of nicotinic receptors. Nature, 2001, 411: 269–276
Talley T T, Harel M, Hibbs R E, et al. Atomic interactions of neonicotinoid agonists with AChBP: Molecular recognition of the distinctive electronegative pharmacophore. Proc Natl Acad Sci USA, 2008, 105: 7606–7611
Ihara M, Okajima T, Yamashita A, et al. Crystal structures of Lymnaea stagnalis AChBP in complex with neonicotinoid insecticides imidacloprid and clothianidin. Invert Neurosci, 2008, 8: 71–81
Celie P H, Klaassen R V, van Rossum-Fikkert S E, et al. Crystal structure of acetylcholine-binding protein from Bulinus truncatus reveals the conserved structural scaffold and sites of variation in nicotinic acetylcholine receptors. J Biol Chem, 2005, 280: 26457–26466
Nishiwaki H, Nakagawa Y, Kuwamura M, et al. Correlations of the electrophysiological activity of neonicotinoids with their binding and insecticidal activities. Pest Manag Sci, 2003, 59: 1023–1030
Matsuda K, Buckingham S D, Freeman J C, et al. Effects of the alpha subunit on imidacloprid sensitivity of recombinant nicotinic acetylcholine receptors. Br J Pharmacol, 1998, 123: 518–524
Kazuhiko M, Steven D B, John C F, et al. Role of the alpha subunit of nicotonic acetylcholine receptor in the selective action of imidacloprid. Pestic Sci, 1999, 55: 211–213
Tomizawa M, Casida J E. Molecular recognition of neonicotinoid insecticides: The determinants of life or death. Acc Chem Res, 2009, 42: 260–269
Casida J E. Neonicotinoid metabolism: Compounds, substituents, pathways, enzymes, organisms, and relevance. J Agric Food Chem, 2011, 59: 2923–2931
Liu Z W, Williamson M S, Lansdell S J, et al. A nicotinic acetylcholine receptor mutation conferring target-site resistance to imidacloprid in Nilaparvata lugens (brown planthopper). Proc Natl Acad Sci USA, 2005, 102: 8420–8425
Tan J, Salgado V L, Hollingworth R M. Neural actions of imidacloprid and their involvement in resistance in the Colorado potato beetle, Leptinotarsa decemlineata (Say). Pest Manag Sci, 2008, 64: 37–47
Tomizawa M, Zhang N, Durkin K A, et al. The neonicotinoid electronegative pharmacophore plays the crucial role in the high affinity and selectivity for the Drosophila nicotinic receptor: An anomaly for the nicotinoid cation-pi interaction model. Biochemistry, 2003, 42: 7819–7827
Shimomura M, Yokota M, Okumura M, et al. Combinatorial mutations in loops D and F strongly influence responses of the alpha7 nicotinic acetylcholine receptor to imidacloprid. Brain Res, 2003, 991: 71–77
Shimomura M, Okuda H, Matsuda K, et al. Effects of mutations of a glutamine residue in loop D of the alpha7 nicotinic acetylcholine receptor on agonist profiles for neonicotinoid insecticides and related ligands. Br J Pharmacol, 2002, 137: 162–169
Matsuda K, Shimomura M, Kondo Y, et al. Role of loop D of the alpha7 nicotinic acetylcholine receptor in its interaction with the insecticide imidacloprid and related neonicotinoids. Br J Pharmacol, 2000, 130: 981–986
Shimomura M, Yokota M, Matsuda K, et al. Roles of loop C and the loop B-C interval of the nicotinic receptor alpha subunit in its selective interactions with imidacloprid in insects. Neurosci Lett, 2004, 363: 195–198
Thompson J D, Gibson T J, Plewniak F, et al. The CLUSTAL_X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res, 1997, 25: 4876–4882
Casida J E, Yamamoto I. Nicotinoid Insecticides and the Nicotinic Acetylcholine Receptor. Tokyo: Springer-Verlag, 1999
Mecozzi S, West A P Jr, Dougherty D A. Cation-pi interactions in aromatics of biological and medicinal interest: Electrostatic potential surfaces as a useful qualitative guide. Proc Natl Acad Sci USA, 1996, 93: 10566–10571
Wang F, Gerzanich V, Wells G B, et al. Assembly of human neuronal nicotinic receptor alpha5 subunits with alpha3, beta2, and beta4 subunits. J Biol Chem, 1996, 271: 17656–17665
Ma J C, Dougherty D A. The cation-pi interaction. Chem Rev, 1997, 97: 1303–1324
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Wang, Y., Li, Z. & Qian, X. Comparative and statistical analysis of nAChR sequences: An ab initio approach to the origin of molecular discrimination. Chin. Sci. Bull. 57, 479–486 (2012). https://doi.org/10.1007/s11434-011-4778-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4778-3