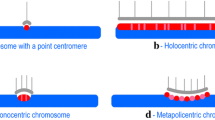
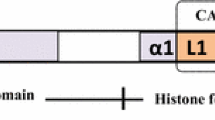
Abstract
The centromere, which is one of the essential parts of a chromosome, controls kinetochore formation and chromosome segregation during mitosis and meiosis. While centromere function is conserved in eukaryotes, the centromeric DNA sequences evolve rapidly and have few similarities among species. The histone H3 variant CENH3 (CENP-A in human), which mostly exists in centromeric nucleosomes, is a universal active centromere mark in eukaryotes and plays an essential role in centromere identity determination. The relationship between centromeric DNA sequences and centromere identity determination is one of the intriguing questions in studying centromere formation. Due to the discoveries in the past decades, including “neocentromeres” and “centromere inactivation”, it is now believed that the centromere identity is determined by epigenetic mechanisms. This review will present recent progress in plant centromere biology.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ekwall K. Epigenetic control of centromere behavior. Annu Rev Genet, 2007, 41: 63–81
Pluta A, Mackay A, Ainsztein A, Goldberg I, Earnshaw W. The centromere: hub of chromosomal activities. Science, 1995, 270: 1591–1594
Fitzgerald-Hayes M, Clarke L, Carbon J. Nucleotide sequence comparisons and functional analysis of yeast centromere DNAs. Cell, 1982, 29: 235–244
Schueler MG, Sullivan BA. Structural and functional dynamics of human centromeric chromatin. Annu Rev Genomics Hum Genet, 2006, 7: 301–313
Dawe RK, Henikoff S. Centromeres put epigenetics in the driver’s seat. Trends Biochem Sci, 2006, 31: 662–669
Birchler JA, Han FP. Maize centromeres: structure, function, epigenetics. Annu Rev Genet, 2009, 43: 287–303
Henikoff S, Furuyama T. Epigenetic inheritance of centromeres. Cold Spring Harb Symp Quant Biol, 2010, 75: 51–60
Allshire RC, Karpen GH. Epigenetic regulation of centromeric chromatin: old dogs, new tricks? Nat Rev Genet, 2008, 9: 923–937
Furuyama S, Biggins S. Centromere identity is specified by a single centromeric nucleosome in budding yeast. Proc Natl Acad Sci USA, 2007, 104: 14706–14711
Maddox PS, Oegema K, Desai A, Cheeseman IM. “Holo”er than thou: chromosome segregation and kinetochore function in C. elegans. Chromosome Res, 2004, 12: 641–653
Steiner FA, Henikoff S. Holocentromeres are dispersed point centromeres localized at transcription factor hotspots. eLIFE, 2014, 3: e02025
Copenhaver GP, Nickel K, Kuromori T, Benito MI, Kaul S, Lin X, Bevan M, Murphy G, Harris B, Parnell LD. Genetic definition and sequence analysis of Arabidopsis centromeres. Science, 1999, 286: 2468–2474
Nagaki K, Talbert PB, Zhong CX, Dawe RK, Henikoff S, Jiang J. Chromatin immunoprecipitation reveals that the 180-bp satellite repeat is the key functional DNA element of Arabidopsis thaliana centromeres. Genetics, 2003, 163: 1221–1225
Cheng Z, Dong F, Langdon T, Ouyang S, Buell CR, Gu M, Blattner FR, Jiang J. Functional rice centromeres are marked by a satellite repeat and a centromere-specific retrotransposon. Plant Cell, 2002, 14: 1691–1704
Ananiev E, Phillips R, Rines H. Chromosome-specific molecular organization of maize (Zea mays L.) centromeric regions. Proc Natl Acad Sci USA, 1998, 95: 13073–13078
Jin W, Melo JR, Nagaki K, Talbert PB, Henikoff S, Dawe RK, Jiang J. Maize centromeres: organization and functional adaptation in the genetic background of oat. Plant Cell, 2004, 16: 571–581
Wu Y, Kikuchi S, Yan H, Zhang W, Rosenbaum H, Iniguez AL, Jiang J. Euchromatic subdomains in rice centromeres are associated with genes and transcription. Plant Cell, 2011, 23: 4054–4064
Wang G, Li H, Cheng Z, Jin W. A novel translocation event leads to a recombinant stable chromosome with interrupted centromeric domains in rice. Chromosoma, 2013, 122: 295–303
Zhong CX, Marshall JB, Topp C, Mroczek R, Kato A, Nagaki K, Birchler JA, Jiang J, Dawe RK. Centromeric retroelements and satellites interact with maize kinetochore protein CENH3. Plant Cell, 2002, 14: 2825–2836
Ma J, Wing RA, Bennetzen JL, Jackson SA. Plant centromere organization: a dynamic structure with conserved functions. Trends Genet, 2007, 23: 134–139
Henikoff S, Ahmad K, Malik HS. The centromere paradox: stable inheritance with rapidly evolving DNA. Science, 2001, 293: 1098–1102
Malik HS, Henikoff S. Adaptive evolution of Cid, a centromere-specific histone in Drosophila. Genetics, 2001, 157: 1293–1298
Talbert PB, Bryson TD, Henikoff S. Adaptive evolution of centromere proteins in plants and animals. J Biol, 2004, 3: 18
Palmer DK, O’Day K, Wener MH, Andrews BS, Margolis RL. A 17-kD centromere protein (CENP-A) copurifies with nucleosome core particles and with histones. J Cell Biol, 1987, 104: 805–815
Palmer DK, O’Day K, Trong H, Charbonneau H, Margolis RL. Purification of the centromere-specific protein CENP-A and demonstration that it is a distinctive histone. Proc Natl Acad Sci USA, 1991, 88: 3734–3738
Talbert PB, Masuelli R, Tyagi AP, Comai L, Henikoff S. Centromeric localization and adaptive evolution of an Arabidopsis histone H3 variant. Plant Cell, 2002, 14: 1053–1066
Morey L, Barnes K, Chen Y, Fitzgerald-Hayes M, Baker RE. The histone fold domain of Cse4 is sufficient for CEN targeting and propagation of active centromeres in budding yeast. Eukaryot Cell, 2004, 3: 1533–1543
Chen Y, Baker RE, Keith KC, Harris K, Stoler S, Fitzgerald-Hayes M. The N terminus of the centromere H3-like protein Cse4p performs an essential function distinct from that of the histone fold domain. Mol Cell Biol, 2000, 20: 7037–7048
Vermaak D, Hayden HS, Henikoff S. Centromere targeting element within the histone fold domain of Cid. Mol Cell Biol, 2002, 22: 7553–7561
Sullivan KF, Hechenberger M, Masri K. Human CENP-A contains a histone H3 related histone fold domain that is required for targeting to the centromere. J Cell Biol, 1994, 127: 581–592
Black BE, Foltz DR, Chakravarthy S, Luger K, Woods VL Jr., Cleveland DW. Structural determinants for generating centromeric chromatin. Nature, 2004, 430: 578–582
Ravi M, Kwong PN, Menorca RM, Valencia JT, Ramahi JS, Stewart JL, Tran RK, Sundaresan V, Comai L, Chan SW. The rapidly evolving centromere-specific histone has stringent functional requirements in Arabidopsis thaliana. Genetics, 2010, 186: 461–471
Ravi M, Chan SW. Haploid plants produced by centromere-mediated genome elimination. Nature, 2010, 464: 615–618
Sanei M, Pickering R, Kumke K, Nasuda S, Houben A. Loss of centromeric histone H3 (CENH3) from centromeres precedes uniparental chromosome elimination in interspecific barley hybrids. Proc Natl Acad Sci USA, 2011, 108: 498–505
Ravi M, Shibata F, Ramahi JS, Nagaki K, Chen C, Murata M, Chan SW. Meiosis-specific loading of the centromere-specific histone CENH3 in Arabidopsis thaliana. PLoS Genet, 2011, 7: e1002121
Harrington JJ, Van Bokkelen G, Mays RW, Gustashaw K, Willard HF. Formation of de novo centromeres and construction of first-generation human artificial microchromosomes. Nat Genet, 1997, 15: 345–355
Phan BH, Jin W, Topp CN, Zhong CX, Jiang J, Dawe RK, Parrott WA. Transformation of rice with long DNA-segments consisting of random genomic DNA or centromere-specific DNA. Transgenic Res, 2007, 16: 341–351
Platero JS, Ahmad K, Henikoff S. A distal heterochromatic block displays centromeric activity when detached from a natural centromere. Mol Cell, 1999, 4: 995–1004
Marshall OJ, Chueh AC, Wong LH, Choo K. Neocentromeres: new insights into centromere structure, disease development, and karyotype evolution. Am J Hum Genet, 2008, 82: 261–282
Nasuda S, Hudakova S, Schubert I, Houben A, Endo TR. Stable barley chromosomes without centromeric repeats. Proc Natl Acad Sci USA, 2005, 102: 9842–9847
Topp CN, Okagaki RJ, Melo JR, Kynast RG, Phillips RL, Dawe RK. Identification of a maize neocentromere in an oat-maize addition line. Cytogenet Genome Res, 2009, 124: 228–238
Fu S, Lv Z, Gao Z, Wu H, Pang J, Zhang B, Dong Q, Guo X, Wang XJ, Birchler JA. De novo centromere formation on a chromosome fragment in maize. Proc Natl Acad Sci USA, 2013, 110: 6033–6036
Zhang B, Lv Z, Pang J, Liu Y, Guo X, Fu S, Li J, Dong Q, Wu HJ, Gao Z. Formation of a functional maize centromere after loss of centromeric sequences and gain of ectopic sequences. Plant Cell, 2013, 25: 1979–1989
Zheng Y, Roseman RR, Carlson WR. Time course study of the chromosome-type breakage-fusion-bridge cycle in maize. Genetics, 1999, 153: 1435–1444
Han F, Lamb JC, Birchler JA. High frequency of centromere inactivation resulting in stable dicentric chromosomes of maize. Proc Natl Acad Sci USA, 2006, 103: 3238–3243
Han F, Gao Z, Birchler JA. Reactivation of an inactive centromere reveals epigenetic and structural components for centromere specification in maize. Plant Cell, 2009, 21: 1929–1939
Gao Z, Fu S, Dong Q, Han F, Birchler JA. Inactivation of a centromere during the formation of a translocation in maize. Chromosome Res, 2011, 19: 755–761
Fu S, Gao Z, Birchler J, Han F. Dicentric chromosome formation and epigenetics of centromere formation in plants. J Genet Genomics, 2012, 39: 125–130
Zhang W, Friebe B, Gill BS, Jiang J. Centromere inactivation and epigenetic modifications of a plant chromosome with three functional centromeres. Chromosoma, 2010, 119: 553–563
Birchler JA, Gao Z, Sharma A, Presting GG, Han FP. Epigenetic aspects of centromere function in plants. Curr Opin Plant Biol, 2011, 14: 217–222
Sullivan BA, Karpen GH. Centromeric chromatin exhibits a histone modification pattern that is distinct from both euchromatin and heterochromatin. Nat Struct Mol Biol, 2004, 11: 1076–1083
Dong Q, Han F. Phosphorylation of histone H2A is associated with centromere function and maintenance in meiosis. Plant J, 2012, 71: 800–809
Koo DH, Han F, Birchler JA, Jiang J. Distinct DNA methylation patterns associated with active and inactive centromeres of the maize B chromosome. Genome Res, 2011, 21: 908–914
Wong LH, Brettingham-Moore KH, Chan L, Quach JM, Anderson MA, Northrop EL, Hannan R, Saffery R, Shaw ML, Williams E, Choo KH. Centromere RNA is a key component for the assembly of nucleoproteins at the nucleolus and centromere. Genome Res, 2007, 17: 1146–1160
Topp CN, Zhong CX, Dawe RK. Centromere-encoded RNAs are integral components of the maize kinetochore. Proc Natl Acad Sci USA, 2004, 101: 15986–15991
Du Y, Topp CN, Dawe RK. DNA binding of centromere protein C (CENPC) is stabilized by single-stranded RNA. PLoS Genet, 2010, 6: e1000835
Gent JI, Dawe RK. RNA as a structural and regulatory component of the centromere. Annu Rev Genet, 2012, 46: 443–453
Choi ES, Stralfors A, Castillo AG, Durand-Dubief M, Ekwall K, Allshire RC. Identification of noncoding transcripts from within CENP-A chromatin at fission yeast centromeres. J Biol Chem, 2011, 286: 23600–23607
Wang K, Wu Y, Zhang W, Dawe RK, Jiang J. Maize centromeres expand and adopt a uniform size in the genetic background of oat. Genome Res, 2014, 24: 107–116
Birchler JA, Han F. Centromere epigenetics in plants. J Genet Genomics, 2013, 40: 201–204
Zhang J, Pawlowski WP, Han F. Centromere pairing in early meiotic prophase requires active centromeres and precedes installation of the synaptonemal complex in maize. Plant Cell, 2013, 25: 3900–3909
Zhang J, Zhang B, Su H, Birchler JA, Han F. Molecular mechanisms of homologous chromosome pairing and segregation in plants. J Genet Genomics, 2014, 41: 117–123
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at link.springer.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Feng, C., Liu, Y., Su, H. et al. Recent advances in plant centromere biology. Sci. China Life Sci. 58, 240–245 (2015). https://doi.org/10.1007/s11427-015-4818-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-015-4818-3