Abstract
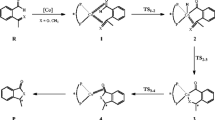
In this paper, we used density functional theory (DFT) computations to study the mechanisms of the hydroacylation reaction of an aldehyde with an alkene catalyzed by Wilkinson’s catalyst and an organic catalyst 2-amino-3-picoline in cationic and neutral systems. An aldehyde’s hydroacylation includes three stages: the C-H activation to form rhodium hydride (stage I), the alkene insertion into the Rh-H bond to give the Rh-alkyl complex (stage II), and the C-C bond formation (stage III). Possible pathways for the hydroacylation originated from the trans and cis isomers of the catalytic cycle. In this paper, we discussed the neutral and cationic pathways. The rate-determining step is the C-H activation step in neutral system but the reductive elimination step in the cationic system. Meanwhile, the alkyl group migration-phosphine ligand coordination pathway is more favorable than the phosphine ligand coordination-alkyl group migration pathway in the C-C formation stage. Furthermore, the calculated results imply that an electron-withdrawing group may decrease the energy barrier of the C-H activation in the benzaldehyde hydroacylation.
Similar content being viewed by others
References
Kakiuchi F, Murai S. Catalytic C-H/olefin coupling. Acc Chem Res, 2002, 35: 826–834
Lei M, Feng WL, Du HG, Xu ZF. Theoretical investigation on regioselectivity of aromatic ketones in the addition with olefin catalyzed by RuH2(CO)(PPh3)3. Sci China Ser B, 2000, 43: 412–420
Crabtree RH. The organometallic chemistry of alkanes. Chem Rev, 1985, 85: 245–269
Ritleng V, Sirlin C, Pfeffer M. Ru-, Rh-, and Pd-catalyzed C-C bond formation involving C-H activation and addition on unsaturated substrates: reactions and mechanistic aspects. Chem Rev, 2002, 102: 1731–1770
Dermenci A, Dong GB. Decarbonylative C-C bond forming reactions mediated by transition metals. Sci China Chem, 2013, 56: 685–701
Suggs JW. Isolation of a stable acylrhodium (III) hydride intermediate formed during aldehyde decarbonylation. Hydroacylation. J Am Chem Soc, 1978, 100: 640–641
Fairlie DP, Bosnich B. Homogeneous catalysis. Mechanism of catalytic hydroacylation: the conversion of 4-pentenals to cyclopentanones. Organometallics, 1988, 7: 946–954
Chung LW, Wiest O, Wu YD. A theoretical study on the trans-addition intramolecular hydroacylation of 4-alkynals catalyzed by cationic rhodium complexes. J Org Chem, 2008, 73: 2649–2655
Morgan JP, Kundu K, Doyle MP. A readily prepared neutral heterobimetallic titanium (IV)-rhodium (I) catalyst for intramolecular hydroacylation. Chem Commun, 2005: 3307–3309
Hyatt IFD, Anderson HK, Morehead Jr AT, Sargent AL. Mechanism of rhodium-catalyzed intramolecular hydroacylation: a computational study. Organometallics, 2008, 27:135–147
Larock R, Oertle K, Potter G. A convenient synthesis of cyclopentanones via rhodium (I)-catalyzed intramolecular hydroac-ylation of unsaturated aldehydes. J Am Chem Soc, 1980, 102: 190–197
Omura S, Fukuyama T, Horiguchi J, Murakami Y, Ryu I. Ruthenium hydride-catalyzed addition of aldehydes to dienes leading to β, Γ-unsaturated ketones. J Am Chem Soc, 2008, 130: 14094–14095
Marder TB, Roe DC, Milstein D. Transition-metal-catalyzed carbon-carbon bond formation via carbon-hydrogen activation. Intermolecular hydroacylation: the addition of aldehydes to alkenes. Organometallics, 1988, 7: 1451–1453
Jun CH, Jo E, Park JW. Intermolecular hydroacylation by transition-metal complexes. Eur J Org Chem, 2007, 2007: 1869–1881
Kokubo K, Matsumasa K, Nishinaka Y, Miura M, Nomura M. Reaction of 2-hydroxybenzaldehydes with alkynes, alkenes, or allenes via cleavage of the aldehyde C-H bond using a Rhodium catalyst system. Bull Chem Soc Jpn, 1999, 72: 303–311
Stemmler RT, Bolm C. An unprecedented rhodium-catalyzed asymmetric intermolecular hydroacylation reaction with salicylaldehydes. Adv Synth Catal, 2007, 349: 1185–1198
Willis MC, Randell-Sly HE, Woodward RL, McNally SJ, Currie GS. Rhodium-catalyzed intermolecular chelation controlled alkene and alkyne hydroacylation: synthetic scope of β-S-substituted aldehyde substrates. J Org Chem, 2006, 71: 5291–5297
Willis MC, Randell-Sly HE, Woodward RL, Currie GS. Chelation-controlled intermolecular alkene and alkyne hydroacylation: the utility of β-thioacetal aldehydes. Org Lett, 2005, 7: 2249–2251
Wojcicki A. Insertion reactions of transition metal-carbon σ-bonded compounds I: carbon monoxide insertion. Adv Organomet Chem, 1973, 11: 87–145
Suggs JW. Activation of aldehyde carbon-hydrogen bonds to oxidative addition via formation of 3-methyl-2-aminopyridyl aldimines and related compounds: rhodium based catalytic hydroacylation. J Am Chem Soc, 1979, 101: 489–489
Jun CH, Lee H, Hong JB. Chelation-assisted intermolecular hydroacylation: direct synthesis of ketone from aldehyde and 1-alkene. J Org Chem, 1997, 62: 1200–1201
Jun CH, Lee DY, Lee H, Hong JB. A highly active catalyst system for intermolecular hydroacylation. Angew Chem Int Ed, 2000, 39: 3070–3072
Park YJ, Park JW, Jun CH. Metal-organic cooperative catalysis in C-H and C-C bond activation and its concurrent recovery. Acc Chem Res, 2008, 41: 222–234
Vautravers NR, Regent DD, Breit B. Inter-and intramolecular hydroacylation of alkenes employing a bifunctional catalyst system. Chem Commun, 2011, 47: 635–6637
Beletskiy EV, Sudheer C, Douglas CJ. Cooperative catalysis approach to intramolecular hydroacylation. J Org Chem, 2012, 77: 5884–5893
Jun CH, Lee H, Hong JB. Chelation-assisted intermolecular hydroacylation: direct synthesis of ketone from aldehyde and 1-alkene. J Org Chem, 1997, 62: 1200–1201
Jun CH, Huh CW, Na SJ. Direct synthesis of ketones from primary alcohols and 1-alkenes. Angew Chem Int Ed, 1998, 37: 145–147
Jun CH. Transition metal-catalyzed carbon-carbon bond activation. Chem Soc Rev, 2004, 33: 610–618
Li HX, Wang XT, Wen MW, Wang ZX. Computational insight into the mechanism of selective imine formation from alcohol and amine catalyzed by the ruthenium(II)-PNP pincer complex. Eur J Inor Chem, 2012, 2012: 5011–5020
Li HX, Wang XT, Huang F, Lu G, Jiang JL, Wang ZX. Computational study on the catalytic role of pincer ruthenium(II)-PNP complex in directly synthesizing amide from alcohol and amine: the origin of selectivity of amide over ester and imine. Organometallics, 2011, 30: 5233–5247
Marcé P, Godard C, Feliz M, Yáñez X, Bo C, Castillón S. Rhodium-catalyzed intermolecular hydroiminoacylation of alkenes: comparison of neutral and cationic catalytic systems. Organometallics, 2009, 28: 2976–2985
Yoo K, Jun CH, Choi CH, Sim E. Density functional study on the C-H bond cleavage of aldimine by a rhodium(I) catalyst. Bull Korean Chem Soc, 2008, 29: 1920–1926
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Keith T, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ. Gaussian 09, Revision B.01, Wallingford CT: Gaussian, Inc., 2010
Zhao Y, Truhlar DG. Density functionals with broad applicability in chemistry. Acc Chem Res, 2008, 41: 157–167
Becke AD. Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys, 1993, 98: 5648–5652
Hay PJ, Wadt WR. Ab initio effective core potentials for molecular calculations. Potentials for K to Au including the outermost core orbitals. J Chem Phys, 1985, 82: 299–240
Sieffert N, Buhl M. Noncovalent interactions in a transition-metal triphenylphosphine complex: a density functional case study. Inorg Chem, 2009, 48: 4622–4624
Kulkarni AD, Truhlar DG. Performance of density functional theory and Møller-plesset second-order perturbation theory for structural parameters in complexes of Ru. J Chem Theory Comput, 2011, 7: 2325–2332
Guo XJ, Tang YH, Zhang X, Lei M. Concerted or stepwise hydrogen transfer in the transfer hydrogenation of acetophenone catalyzed by ruthenium-acetamido complex: a theoretical mechanistic investigation. J Phy Chem A, 2011, 115: 12321–12330
Feng R, Xiao A, Zhang X, Tang YH, Lei M. Origins of enantioselectivity in asymmetric ketone hydrogenation catalyzed by a RuH2 (binap)(cydn) complex: insights from a computational study. Dalton Trans, 2013, 42: 2130–2145
Chen Y, Tang YH, Lei M. A comparative study on the hydrogenation of ketones catalyzed by diphosphine-diamine transition metal complexes using DFT method. Dalton Trans, 2009: 2359–2364
Hassan M, Zhang X, Zhang WC, Guo XJ, Chen BH, Lei M. How does methanol assist the hydrogen transfer in Pd-catalyzed cyclocarbonylation of allylic alcohols? Insights from a DFT study. Chem Lett, 2012, 41: 693–695
Cioslowski J. A new population analysis based on atomic polar tensors. J Am Chem Soc, 1989, 111: 8333–8336
Barone V, Cossi M. Quantum calculation of molecular energies and energy gradients in solution by a conductor solvent model. J Phys Chem A, 1998, 102: 1995–2001
Strajbl M, Sham YY, Villa J, Chu ZT, Warshel A. Calculations of activation entropies of chemical reactions in solution. J Phys Chem B, 2000, 104: 4578–4584
Hermans J, Wang L. Inclusion of loss of translational and rotational freedom in theoretical estimates of free energies of binding. Application to a complex of benzene and mutant T4 lysozyme. J Am Chem Soc, 1997, 119: 2707–2714
Yu ZX, Houk K. Intramolecular 1,3-dipolar ene reactions of nitrile oxides occur by stepwise 1,1-cycloaddition/retro-ene mechanisms. J Am Chem Soc, 2003, 125: 13825–13830
Liang Y, Liu S, Xia Y. Z, Li YH, Yu ZX. Mechanism, regioselectivity, and the kinetics of phosphine-catalyzed [3+2] cycloaddition reactions of allenoates and electron-deficient alkenes. Chem Eur J, 2008, 14: 4361–4373
Tang SY, Gong TJ, Fu Y. Mechanistic study of copper-catalyzed intramolecular ortho-C-H activation/carbon-nitrogen and carbon-oxygen cyclizations. Sci China Chem, 2013, 56: 619–632
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wang, M., Zhang, X., Chen, Z. et al. A theoretical study on the mechanisms of intermolecular hydroacylation of aldehyde catalyzed by neutral and cationic rhodium complexes. Sci. China Chem. 57, 1264–1275 (2014). https://doi.org/10.1007/s11426-014-5102-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-014-5102-2