Abstract
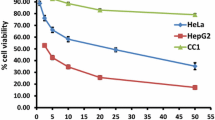
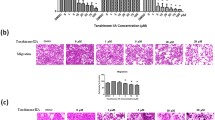
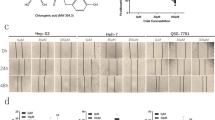
Tanshinone IIA (Tan IIA), a natural product from herb Salvia miltiorrhiza Bunge, has potential anti-tumor activity. The aim of this study was to pinpoint the molecular mechanisms underlying Tan IIA-induced cancer cell apoptosis. Human hepatoma BEL-7402 cells treated with Tan IIA underwent assessment with MTT assay for cell viability, 10-day culture for colony formation, flow cytometry and fluorescence microscopy for apoptosis and cell cycle analysis. Changes in intracellular [Ca2+] and mitochondrial membrane potential (∆ψ) reflected the calcium-dependent apoptosis pathway. RT-PCR was used to detect gene expression of Bad and metallothionein 1A (MT 1A). Cytotoxicity of Tan IIA was tested in human amniotic mesenchymal stem cells (HAMCs). Tan IIA exhibited dose-dependent and time-dependent anticancer effects on BEL-7402 cells through apoptosis and G0/G1 arrest. Cells treated with Tan IIA increased their intracellular calcium, decreased their mitochondrial membrane potential and induced Bad and MT 1A mRNA expression. No adverse effects of Tan IIA were found in HAMCs. In conclusion, these results indicate that Tan IIA-induced cancer cell apoptosis acts via activation of calcium-dependent apoptosis signaling pathways and upregulation of MT 1A expression.






Similar content being viewed by others
Abbreviations
- HAMCs:
-
Human amniotic mesenchymal stem cells
- HCC:
-
Hepatocellular carcinoma
- MT 1A:
-
Metallothionein 1A
- SMB:
-
Salvia miltiorrhiza Bunge
- Tan IIA:
-
Tanshinone IIA
References
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM (2010) Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 127:2893–2917
Farazi PA, DePinho RA (2006) Hepatocellular carcinoma pathogenesis: from genes to environment. Nat Rev Cancer 6:674–687
Zhou L, Zuo Z, Chow MS (2005) Danshen: an overview of its chemistry, pharmacology, pharmacokinetics, and clinical use. J Clin Pharmacol 45:1345–1359
Niu XL, Ichimori K, Yang X, Hirota Y, Hoshiai K, Li M, Nakazawa H (2000) Tanshinone II-A inhibits low density lipoprotein oxidation in vitro. Free Radic Res 33:305–312
Yan FF, Liu YF, Liu Y, Zhao YX (2009) Sulfotanshinone Sodium Injection could decrease fibrinogen level and improve clinical outcomes in patients with unstable angina pectoris. Int J Cardiol 135:254–255
Dong X, Dong J, Zhang R, Fan L, Liu L, Wu G (2009) Anti-inflammatory effects of tanshinone IIA on radiation-induced microglia BV-2 cells inflammatory response. Cancer Biother Radiopharm 24:681–687
Kapoor S (2009) Tanshinone IIA: a potent, natural anti-carcinogenic agent for the management of systemic malignancies. Chin J Integr Med 15:153
Yoon Y, Kim YO, Jeon WK, Park HJ, Sung HJ (1999) Tanshinone IIA isolated from Salvia miltiorrhiza BUNGE induced apoptosis in HL60 human premyelocytic leukemia cell line. J Ethnopharmacol 68:121–127
Lee WY, Cheung CC, Liu KW, Fung KP, Wong J, Lai PB, Yeung JH (2010) Cytotoxic effects of tanshinones from Salvia miltiorrhiza on doxorubicin-resistant human liver cancer cells. J Nat Prod 73:854–859
Tang Z, Tang Y, Fu L (2003) Growth inhibition and apoptosis induction in human hepatoma cells by tanshinone II A. J Huazhong Univ Sci Technol Med Sci 23:166–168, 172
Zhu B, Zhai Q, Yu B (2010) Tanshinone IIA protects rat primary hepatocytes against carbon tetrachloride toxicity via inhibiting mitochondria permeability transition. Pharm Biol 48:484–487
Shi SL, Li QF, Liu QR, Xu DH, Tang J, Liang Y, Zhao ZL, Yang LM (2009) Nuclear matrix protein, prohibitin, was down-regulated and translocated from nucleus to cytoplasm during the differentiation of osteosarcoma MG-63 cells induced by ginsenoside Rg1, cinnamic acid, and tanshinone IIA (RCT). J Cell Biochem 108:926–934
Yeruva L, Hall C, Elegbede JA, Carper SW (2010) Perillyl alcohol and methyl jasmonate sensitize cancer cells to cisplatin. Anticancer Drugs 21:1–9
Ward MW, Rego AC, Frenguelli BG, Nicholls DG (2000) Mitochondrial membrane potential and glutamate excitotoxicity in cultured cerebellar granule cells. J Neurosci 20:7208–7219
Ni X, Sullivan GJ, Martin-Caraballo M (2007) Developmental characteristics of AMPA receptors in chick lumbar motoneurons. Dev Neurobiol 67:1419–1432
Miao Z, Jin J, Chen L, Zhu J, Huang W, Zhao J, Qian H, Zhang X (2006) Isolation of mesenchymal stem cells from human placenta: comparison with human bone marrow mesenchymal stem cells. Cell Biol Int 30:681–687
Mizokami T, Hisha H, Okazaki S, Takaki T, Wang XL, Song CY, Li Q, Kato J, Hosaka N, Inaba M, Kanzaki H, Ikehara S (2009) Preferential expansion of human umbilical cord blood-derived CD34-positive cells on major histocompatibility complex-matched amnion-derived mesenchymal stem cells. Haematologica 94:618–628
Yuan SL, Wei YQ, Wang XJ, Xiao F, Li SF, Zhang J (2004) Growth inhibition and apoptosis induction of tanshinone II-A on human hepatocellular carcinoma cells. World J Gastroenterol 10:2024–2028
Wang ZY (2003) Ham-Wasserman lecture: treatment of acute leukemia by inducing differentiation and apoptosis. Hematology Am Soc Hematol Educ Program 1–13
Wang X, Yuan S, Wang C (1996) A preliminary study of the anti-cancer effect of tanshinone on hepatic carcinoma and its mechanism of action in mice. Zhonghua Zhong Liu Za Zhi 18:412–414
Soleimanpour SA, Crutchlow MF, Ferrari AM, Raum JC, Groff DN, Rankin MM, Liu C, De Leon DD, Naji A, Kushner JA, Stoffers DA (2010) Calcineurin signaling regulates human islet {beta}-cell survival. J Biol Chem 285:40050–40059
Wang X, Wei Y, Yuan S, Liu G, Lu Y, Zhang J, Wang W (2005) Potential anticancer activity of tanshinone IIA against human breast cancer. Int J Cancer 116:799–807
Liu Y, Chen H, Jiang Y (2001) Protective effect of tanshinone IIA on acute hepatic injury in mice. Zhong Yao Cai 24:588–589
Liu Y, Wang X (2003) Protective effects of tanshinone IIA on injured primary cultured rat hepatocytes induced by CCl4. Zhong Yao Cai 26:415–417
Lu Q, Zhang P, Zhang X, Chen J (2009) Experimental study of the anti-cancer mechanism of tanshinone IIA against human breast cancer. Int J Mol Med 24:773–780
Olofsson MH, Havelka AM, Brnjic S, Shoshan MC, Linder S (2008) Charting calcium-regulated apoptosis pathways using chemical biology: role of calmodulin kinase II. BMC Chem Biol 8:2
Williams JP, Micoli K, McDonald JM (2010) Calmodulin - an often-ignored signal in osteoclasts. Ann N Y Acad Sci 1192:358–364
Devireddy LR, Green MR (2003) Transcriptional program of apoptosis induction following interleukin 2 deprivation: identification of RC3, a calcium/calmodulin binding protein, as a novel proapoptotic factor. Mol Cell Biol 23:4532–4541
Crabtree GR (2001) Calcium, calcineurin, and the control of transcription. J Biol Chem 276:2313–2316
Shibasaki F, McKeon F (1995) Calcineurin functions in Ca(2+)-activated cell death in mammalian cells. J Cell Biol 131:735–743
Wang HG, Pathan N, Ethell IM, Krajewski S, Yamaguchi Y, Shibasaki F, McKeon F, Bobo T, Franke TF, Reed JC (1999) Ca2+-induced apoptosis through calcineurin dephosphorylation of BAD. Science 284:339–343
Willis SN, Chen L, Dewson G, Wei A, Naik E, Fletcher JI, Adams JM, Huang DC (2005) Proapoptotic Bak is sequestered by Mcl-1 and Bcl-xL, but not Bcl-2, until displaced by BH3-only proteins. Genes Dev 19:1294–1305
Wei MC, Zong WX, Cheng EH, Lindsten T, Panoutsakopoulou V, Ross AJ, Roth KA, MacGregor GR, Thompson CB, Korsmeyer SJ (2001) Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 292:727–730
Shimizu S, Narita M, Tsujimoto Y (1999) Bcl-2 family proteins regulate the release of apoptogenic cytochrome c by the mitochondrial channel VDAC. Nature 399:483–487
Sullivan PG, Dragicevic NB, Deng JH, Bai Y, Dimayuga E, Ding Q, Chen Q, Bruce-Keller AJ, Keller JN (2004) Proteasome inhibition alters neural mitochondrial homeostasis and mitochondria turnover. J Biol Chem 279:20699–20707
Rivera A, Maxwell SA (2005) The p53-induced gene-6 (proline oxidase) mediates apoptosis through a calcineurin-dependent pathway. J Biol Chem 280:29346–29354
Chiu TL, Su CC (2010) Tanshinone IIA induces apoptosis in human lung cancer A549 cells through the induction of reactive oxygen species and decreasing the mitochondrial membrane potential. Int J Mol Med 25:231–236
Sastry KS, Smith AJ, Karpova Y, Datta SR, Kulik G (2006) Diverse antiapoptotic signaling pathways activated by vasoactive intestinal polypeptide, epidermal growth factor, and phosphatidylinositol 3-kinase in prostate cancer cells converge on BAD. J Biol Chem 281:20891–20901
She QB, Solit DB, Ye Q, O’Reilly KE, Lobo J, Rosen N (2005) The BAD protein integrates survival signaling by EGFR/MAPK and PI3 K/Akt kinase pathways in PTEN-deficient tumor cells. Cancer Cell 8:287–297
Theocharis SE, Margeli AP, Klijanienko JT, Kouraklis GP (2004) Metallothionein expression in human neoplasia. Histopathology 45:103–118
Alonso-Gonzalez C, Mediavilla D, Martinez-Campa C, Gonzalez A, Cos S, Sanchez-Barcelo EJ (2008) Melatonin modulates the cadmium-induced expression of MT-2 and MT-1 metallothioneins in three lines of human tumor cells (MCF-7, MDA-MB-231 and HeLa). Toxicol Lett 181:190–195
Saito T, Tezuka T, Konno R, Fujii N (2010) Protective effects of metallothionein I and II against metal- and ultraviolet radiation-induced damage in cultured lens epithelial cells. Jpn J Ophthalmol 54:486–493
Majumder S, Roy S, Kaffenberger T, Wang B, Costinean S, Frankel W, Bratasz A, Kuppusamy P, Hai T, Ghoshal K, Jacob ST (2010) Loss of metallothionein predisposes mice to diethylnitrosamine-induced hepatocarcinogenesis by activating NF-kappaB target genes. Cancer Res 70:10265–10276
de Gruijl FR (2000) Photocarcinogenesis: UVA vs UVB. Methods Enzymol 319:359–366
Ioachim E, Assimakopoulos D, Peschos D, Zissi A, Skevas A, Agnantis NJ (1999) Immunohistochemical expression of metallothionein in benign premalignant and malignant epithelium of the larynx: correlation with p53 and proliferative cell nuclear antigen. Pathol Res Pract 195:809–814
Klaassen CD, Liu J, Choudhuri S (1999) Metallothionein: an intracellular protein to protect against cadmium toxicity. Annu Rev Pharmacol Toxicol 39:267–294
Hu R, Hebbar V, Kim BR, Chen C, Winnik B, Buckley B, Soteropoulos P, Tolias P, Hart RP, Kong AN (2004) In vivo pharmacokinetics and regulation of gene expression profiles by isothiocyanate sulforaphane in the rat. J Pharmacol Exp Ther 310:263–271
Yeh CT, Yen GC (2005) Effect of sulforaphane on metallothionein expression and induction of apoptosis in human hepatoma HepG2 cells. Carcinogenesis 26:2138–2148
Cherian MG, Jayasurya A, Bay BH (2003) Metallothioneins in human tumors and potential roles in carcinogenesis. Mutat Res 533:201–209
Krizkova S, Masarik M, Majzlik P, Kukacka J, Kruseova J, Adam V, Prusa R, Eckschlager T, Stiborova M, Kizek R (2010) Serum metallothionein in newly diagnosed patients with childhood solid tumours. Acta Biochim Pol 57:561–566
Yap X, Tan HY, Huang J, Lai Y, Yip GW, Tan PH, Bay BH (2009) Over-expression of metallothionein predicts chemoresistance in breast cancer. J Pathol 217:563–570
Jin R, Huang J, Tan PH, Bay BH (2004) Clinicopathological significance of metallothioneins in breast cancer. Pathol Oncol Res 10:74–79
Xia WJ, Yang M, Fok TF, Li K, Chan WY, Ng PC, Ng HK, Chik KW, Wang CC, Gu GJ, Woo KS, Fung KP (2005) Partial neuroprotective effect of pretreatment with tanshinone IIA on neonatal hypoxia-ischemia brain damage. Pediatr Res 58:784–790
Acknowledgments
We thank Dr Li-Mei Yu at the Key Laboratory of Cell Engineering in Guizhou Province (Zunyi, China) for support and assistance in this study.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Z.-K. Dai and J.-K. Qin contributed equally to this work.
Rights and permissions
About this article
Cite this article
Dai, ZK., Qin, JK., Huang, JE. et al. Tanshinone IIA activates calcium-dependent apoptosis signaling pathway in human hepatoma cells. J Nat Med 66, 192–201 (2012). https://doi.org/10.1007/s11418-011-0576-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-011-0576-0