Abstract
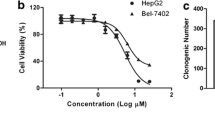
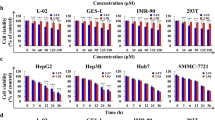
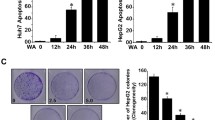
Chlorogenic acid (CGA), a phenylpropanoid derived from Eucommia ulmoides Oliver, has been shown to exhibit potent cytotoxic and anti-proliferative activities against several human cancers. However, the effects of CGA on hepatocellular carcinoma (HCC) and the underlying mechanisms have not been intensively studied. In this study, the CGA treatment effects on the viability of human hepatoma cells were investigated by MTT assay. Our data showed that CGA could dose-dependently inhibit the activity of human hepatoma cells Hep-G2 and Huh-7, but did not affect the activity and growth of normal human hepatocyte QSG-7701. The genes and pathways influenced by CGA treatment were explored by RNA sequencing and bioinformatics analysis, which identified 323 differentially expressed genes (DEGs) involved in multiple pharmacological signaling pathways such as MAPK, NF-κB, apoptosis and TGF-β signaling pathways. Further analyses by real-time quantitative PCR, Western blot and flow cytometry revealed that CGA effectually suppressed the noncanonical NF-κB signaling pathway, meanwhile it activated the mitochondrial apoptosis of HCC by upregulation of the BH3-only protein Bcl-2 binding component 3 (BBC3). Our findings demonstrated the potential of CGA in suppressing human hepatoma cells and provided a new insight into the anti-cancer mechanism of CGA.





Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424. https://doi.org/10.3322/caac.21492
Forner A, Reig M, Bruix J (2018) Hepatocellular carcinoma. Lancet 391:1301–1314. https://doi.org/10.1016/s0140-6736(18)30010-2
Vibert E, Schwartz M, Olthoff KM (2020) Advances in resection and transplantation for hepatocellular carcinoma. J Hepatol 72:262–276. https://doi.org/10.1016/j.jhep.2019.11.017
Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, Schwartz M, Porta C, Zeuzem S, Bolondi L, Greten TF, Galle PR, Seitz JF, Borbath I, Häussinger D, Giannaris T, Shan M, Moscovici M, Voliotis D, Bruix J (2008) Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 359:378–390. https://doi.org/10.1056/NEJMoa0708857
Spallanzani A, Orsi G, Andrikou K, Gelsomino F, Rimini M, Riggi L, Cascinu S (2018) Lenvatinib as a therapy for unresectable hepatocellular carcinoma. Expert Rev Anticancer Ther 18:1069–1076. https://doi.org/10.1080/14737140.2018.1524297
Bruix J, Qin S, Merle P, Granito A, Huang YH, Bodoky G, Pracht M, Yokosuka O, Rosmorduc O, Breder V, Gerolami R, Masi G, Ross PJ, Song T, Bronowicki JP, Ollivier-Hourmand I, Kudo M, Cheng AL, Llovet JM, Finn RS, LeBerre MA, Baumhauer A, Meinhardt G, Han G (2017) Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 389:56–66. https://doi.org/10.1016/s0140-6736(16)32453-9
Yang PM, Lin LS, Liu TP (2020) Sorafenib inhibits ribonucleotide reductase regulatory subunit M2 (RRM2) in hepatocellular carcinoma cells. Biomolecules. https://doi.org/10.3390/biom10010117
Wu J, Chen H, Li H, Tang Y, Yang L, Cao S, Qin D (2016) Antidepressant potential of chlorogenic acid-enriched extract from Eucommia ulmoides Oliver Bark with neuron protection and promotion of serotonin release through enhancing Synapsin I expression. Molecules 21:260. https://doi.org/10.3390/molecules21030260
Yao X, Gongyu G, Chen G (2006) Determination of active constituents in Lonicera confusa DC. by capillary electrophoresis with amperometric detection. Biomed Chromatogr 20:1192–1199. https://doi.org/10.1002/bmc.684
Chen L, Lan MS (2010) Studies on the chemical constituents of Teyou 2 Folium Mori. J Chin Med Mater 33:380–382
Meinhart AD, Damin FM, Caldeirão L, da Silveira TFF, Filho JT, Godoy HT (2017) Chlorogenic acid isomer contents in 100 plants commercialized in Brazil. Food Res Int 99:522–530. https://doi.org/10.1016/j.foodres.2017.06.017
Jiang Y, Kusama K, Satoh K, Takayama E, Watanabe S, Sakagami H (2000) Induction of cytotoxicity by chlorogenic acid in human oral tumor cell lines. Phytomedicine 7:483–491. https://doi.org/10.1016/s0944-7113(00)80034-3
Yagasaki K, Miura Y, Okauchi R, Furuse T (2000) Inhibitory effects of chlorogenic acid and its related compounds on the invasion of hepatoma cells in culture. Cytotechnology 33:229–235. https://doi.org/10.1023/a:1008141918852
Yun N, Kang JW, Lee SM (2012) Protective effects of chlorogenic acid against ischemia/reperfusion injury in rat liver: molecular evidence of its antioxidant and anti-inflammatory properties. J Nutr Biochem 23:1249–1255. https://doi.org/10.1016/j.jnutbio.2011.06.018
Santana-Galvez J, Cisneros-Zevallos L, Jacobo-Velazquez DA (2017) Chlorogenic acid: recent advances on its dual role as a food additive and a nutraceutical against metabolic syndrome. Molecules. https://doi.org/10.3390/molecules22030358
Gong XX, Su XS, Zhan K, Zhao GQ (2018) The protective effect of chlorogenic acid on bovine mammary epithelial cells and neutrophil function. J Dairy Sci 101:10089–10097. https://doi.org/10.3168/jds.2017-14328
Chen J, Xie H, Chen D, Yu B, Mao X, Zheng P, Yu J, Luo Y, Luo J, He J (2018) Chlorogenic acid improves intestinal development via suppressing mucosa inflammation and cell apoptosis in weaned pigs. ACS Omega 3:2211–2219. https://doi.org/10.1021/acsomega.7b01971
Kang TY, Yang HR, Zhang J, Li D, Lin J, Wang L, Xu X (2013) The studies of chlorogenic acid antitumor mechanism by gene chip detection: the immune pathway gene expression. J Anal Methods Chem 2013:617243. https://doi.org/10.1155/2013/617243
Naveed M, Hejazi V, Abbas M, Kamboh AA, Khan GJ, Shumzaid M, Ahmad F, Babazadeh D, FangFang X, Modarresi-Ghazani F, WenHua L, XiaoHui Z (2018) Chlorogenic acid (CGA): a pharmacological review and call for further research. Biomed Pharmacother 97:67–74. https://doi.org/10.1016/j.biopha.2017.10.064
Sadeghi Ekbatan S, Iskandar MM, Sleno L, Sabally K, Khairallah J, Prakash S, Kubow S (2018) Absorption and metabolism of phenolics from digests of polyphenol-rich potato extracts using the Caco-2/HepG2 co-culture system. Foods. https://doi.org/10.3390/foods7010008
Farah A, Monteiro M, Donangelo CM, Lafay S (2008) Chlorogenic acids from green coffee extract are highly bioavailable in humans. J Nutr 138:2309–2315. https://doi.org/10.3945/jn.108.095554
Refolo MG, Lippolis C, Carella N, Cavallini A, Messa C, D’Alessandro R (2018) Chlorogenic acid improves the regorafenib effects in human hepatocellular carcinoma cells. Int J Mol Sci. https://doi.org/10.3390/ijms19051518
Deka SJ, Gorai S, Manna D, Trivedi V (2017) Evidence of PKC binding and translocation to explain the anticancer mechanism of chlorogenic acid in breast cancer cells. Curr Mol Med 17:79–89. https://doi.org/10.2174/1566524017666170209160619
Belkaid A, Currie JC, Desgagnés J, Annabi B (2006) The chemopreventive properties of chlorogenic acid reveal a potential new role for the microsomal glucose-6-phosphate translocase in brain tumor progression. Cancer Cell Int 6:7. https://doi.org/10.1186/1475-2867-6-7
Bandyopadhyay G, Biswas T, Roy KC, Mandal S, Mandal C, Pal BC, Bhattacharya S, Rakshit S, Bhattacharya DK, Chaudhuri U, Konar A, Bandyopadhyay S (2004) Chlorogenic acid inhibits Bcr-Abl tyrosine kinase and triggers p38 mitogen-activated protein kinase-dependent apoptosis in chronic myelogenous leukemic cells. Blood 104:2514–2522. https://doi.org/10.1182/blood-2003-11-4065
Huang MT, Smart RC, Wong CQ, Conney AH (1988) Inhibitory effect of curcumin, chlorogenic acid, caffeic acid, and ferulic acid on tumor promotion in mouse skin by 12-O-tetradecanoylphorbol-13-acetate. Cancer Res 48:5941–5946
Yamagata K, Izawa Y, Onodera D, Tagami M (2018) Chlorogenic acid regulates apoptosis and stem cell marker-related gene expression in A549 human lung cancer cells. Mol Cell Biochem 441:9–19. https://doi.org/10.1007/s11010-017-3171-1
Tian L, Su CP, Wang Q, Wu FJ, Bai R, Zhang HM, Liu JY, Lu WJ, Wang W, Lan F, Guo SZ (2019) Chlorogenic acid: a potent molecule that protects cardiomyocytes from TNF-α-induced injury via inhibiting NF-κB and JNK signals. J Cell Mol Med 23:4666–4678. https://doi.org/10.1111/jcmm.14351
Feng R, Lu Y, Bowman LL, Qian Y, Castranova V, Ding M (2005) Inhibition of activator protein-1, NF-kappaB, and MAPKs and induction of phase 2 detoxifying enzyme activity by chlorogenic acid. J Biol Chem 280:27888–27895. https://doi.org/10.1074/jbc.M503347200
Park JJ, Hwang SJ, Park JH, Lee HJ (2015) Chlorogenic acid inhibits hypoxia-induced angiogenesis via down-regulation of the HIF-1α/AKT pathway. Cell Oncol (Dordr) 38:111–118. https://doi.org/10.1007/s13402-014-0216-2
Yan Y, Li J, Han J, Hou N, Song Y, Dong L (2015) Chlorogenic acid enhances the effects of 5-fluorouracil in human hepatocellular carcinoma cells through the inhibition of extracellular signal-regulated kinases. Anticancer Drugs 26:540–546. https://doi.org/10.1097/cad.0000000000000218
Yan Y, Liu N, Hou N, Dong L, Li J (2017) Chlorogenic acid inhibits hepatocellular carcinoma in vitro and in vivo. J Nutr Biochem 46:68–73. https://doi.org/10.1016/j.jnutbio.2017.04.007
Burgos-Morón E, Calderón-Montaño JM, Orta ML, Pastor N, Pérez-Guerrero C, Austin C, Mateos S, López-Lázaro M (2012) The coffee constituent chlorogenic acid induces cellular DNA damage and formation of topoisomerase I- and II-DNA complexes in cells. J Agric Food Chem 60:7384–7391. https://doi.org/10.1021/jf300999e
Xue N, Zhou Q, Ji M, Jin J, Lai F, Chen J, Zhang M, Jia J, Yang H, Zhang J, Li W, Jiang J, Chen X (2017) Chlorogenic acid inhibits glioblastoma growth through repolarizating macrophage from M2 to M1 phenotype. Sci Rep 7:39011. https://doi.org/10.1038/srep39011
Huang S, Wang LL, Xue NN, Li C, Guo HH, Ren TK, Zhan Y, Li WB, Zhang J, Chen XG, Han YX, Zhang JL, Jiang JD (2019) Chlorogenic acid effectively treats cancers through induction of cancer cell differentiation. Theranostics 9:6745–6763. https://doi.org/10.7150/thno.34674
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub TR, Lander ES, Mesirov JP (2005) Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 102:15545–15550. https://doi.org/10.1073/pnas.0506580102
Zamzami N, Kroemer G (2003) Apoptosis: mitochondrial membrane permeabilization—the (w)hole story? Curr Biol 13:R71–R73. https://doi.org/10.1016/s0960-9822(02)01433-1
Wang L, Du H, Chen P (2020) Chlorogenic acid inhibits the proliferation of human lung cancer A549 cell lines by targeting annexin A2 in vitro and in vivo. Biomed Pharmacother 131:110673. https://doi.org/10.1016/j.biopha.2020.110673
Yang JS, Liu CW, Ma YS, Weng SW, Tang NY, Wu SH, Ji BC, Ma CY, Ko YC, Funayama S, Kuo CL (2012) Chlorogenic acid induces apoptotic cell death in U937 leukemia cells through caspase- and mitochondria-dependent pathways. In Vivo 26:971–978
Rakshit S, Mandal L, Pal BC, Bagchi J, Biswas N, Chaudhuri J, Chowdhury AA, Manna A, Chaudhuri U, Konar A, Mukherjee T, Jaisankar P, Bandyopadhyay S (2010) Involvement of ROS in chlorogenic acid-induced apoptosis of Bcr-Abl+ CML cells. Biochem Pharmacol 80:1662–1675. https://doi.org/10.1016/j.bcp.2010.08.013
Sun SC (2011) Non-canonical NF-κB signaling pathway. Cell Res 21:71–85. https://doi.org/10.1038/cr.2010.177
Jiang X, Li C, Lin B, Hong H, Jiang L, Zhu S, Wang X, Tang N, Li X, She F, Chen Y (2017) cIAP2 promotes gallbladder cancer invasion and lymphangiogenesis by activating the NF-kappaB pathway. Cancer Sci 108:1144–1156. https://doi.org/10.1111/cas.13236
Lau R, Niu MY, Pratt MA (2012) cIAP2 represses IKKalpha/beta-mediated activation of MDM2 to prevent p53 degradation. Cell Cycle 11:4009–4019. https://doi.org/10.4161/cc.22223
Gressot LV, Doucette T, Yang Y, Fuller GN, Manyam G, Rao A, Latha K, Rao G (2017) Analysis of the inhibitors of apoptosis identifies BIRC3 as a facilitator of malignant progression in glioma. Oncotarget 8:12695–12704. https://doi.org/10.18632/oncotarget.8657
Moron-Calvente V, Romero-Pinedo S, Toribio-Castello S, Plaza-Diaz J, Abadia-Molina AC, Rojas-Barros DI, Beug ST, LaCasse EC, MacKenzie A, Korneluk R, Abadia-Molina F (2018) Inhibitor of apoptosis proteins, NAIP, cIAP1 and cIAP2 expression during macrophage differentiation and M1/M2 polarization. PLoS One 13:e0193643. https://doi.org/10.1371/journal.pone.0193643
Wang CY, Mayo MW, Korneluk RG, Goeddel DV, Baldwin AS Jr (1998) NF-kappaB antiapoptosis: induction of TRAF1 and TRAF2 and c-IAP1 and c-IAP2 to suppress caspase-8 activation. Science 281:1680–1683. https://doi.org/10.1126/science.281.5383.1680
Han J, Flemington C, Houghton AB, Gu Z, Zambetti GP, Lutz RJ, Zhu L, Chittenden T (2001) Expression of bbc3, a pro-apoptotic BH3-only gene, is regulated by diverse cell death and survival signals. Proc Natl Acad Sci USA 98:11318–11323. https://doi.org/10.1073/pnas.201208798
Wei JS, Whiteford CC, Cenacchi N, Son CG, Khan J (2005) BBC3 mediates fenretinide-induced cell death in neuroblastoma. Oncogene 24:7976–7983. https://doi.org/10.1038/sj.onc.1208947
Singh N, Sarkar J, Sashidhara KV, Ali S, Sinha S (2014) Anti-tumour activity of a novel coumarin–chalcone hybrid is mediated through intrinsic apoptotic pathway by inducing PUMA and altering Bax/Bcl-2 ratio. Apoptosis 19:1017–1028. https://doi.org/10.1007/s10495-014-0975-2
Acknowledgements
We thank the Teaching and Research Core Facility at College of Life Sciences, NWAFU, for their support in this work.
Funding
This work is supported by 13th Five Year National Key Research and Development Program of China (2017YFD0600702).
Author information
Authors and Affiliations
Contributions
HC and JD conceived and designed this research. YJ, HN and NS performed the experiments. YJ, HC and WH analyzed the data. HC, YJ and JD wrote the manuscript. All authors reviewed and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest/competing interests.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jiang, Y., Nan, H., Shi, N. et al. Chlorogenic acid inhibits proliferation in human hepatoma cells by suppressing noncanonical NF-κB signaling pathway and triggering mitochondrial apoptosis. Mol Biol Rep 48, 2351–2364 (2021). https://doi.org/10.1007/s11033-021-06267-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06267-3