Abstract
Purpose
In urban areas, humus quantity and quality depend less on natural environmental factors than on anthropogenic ones. The aim of the study was to assess the impact of different land use types of urban soils on the properties of soil organic matter (SOM).
Materials and methods
Thirty-five sites involving four ways of soil use were examined: lawns, allotment gardens, fallows, and arable lands. The study was conducted in Pruszków Town in the Warsaw Agglomeration, Central Poland. Lawns and allotment gardens were located in the central part of the town, whereas fallows and arable lands were in the peripheral zones. Humus horizons, to a depth of 0–20 cm, were analyzed. Using Na-pyrophosphate extraction, we determined the soluble SOM compounds (PY), and the organic matter in the extraction residue, considered as humins (HM). In a separate extraction (with 0.05 M H2SO4), low molecular weight (LMW) humus compounds were determined. The quantity of humic acids (HA) precipitated during Na-pyrophosphate extraction was determined as well. A spectroscopic method (UV-Vis) was used to characterize HA properties. The absorption coefficients E4/E6 were calculated based on the results of absorbance measurements involving 465- and 665-nm wavelengths of UV-Vis light. Statistical analyses were performed to find similarities and differences between soils differently used in Pruszków.
Results and discussion
The dominant part of the humus in the studied soils were humins HMs. There were two times more HM in the central part of the town than that in the peripheral zones. The same observation was made for soluble humus compounds (PY). The amount of LMW fractions was similar in soils of all uses. The degree of humification was small and averaged about 30% for all soils. Fulvic acid (FA) concentrations predominated over HA concentrations in all soils. The least condensed HA occurred in the allotment gardens with an E4/E6 ratio of 5.7, whereas the most condensed HAs were present in soil on arable lands (E4/E6 ratio of 4.7).
Conclusions
The studies have shown that the type of land use affects humus properties. The main differences were found to be in the quantity of humus compounds. Soils from the central part of the town contained more stable (HM) and soluble (PY) compounds than soils in the outskirts of town. PY compounds were characterized by a simple structure. Fulvic acids (FA) dominated in all of the studied soils (low HA/FA ratio). A high E4/E6 ratio indicates low maturity of humic fractions with low molecular weight compounds.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Urban soils show a great variety (Greinert 2015; Greinert 2017), mainly due to the way they are used and the various functions they perform. The most common uses are residential, commercial, and transport-related, but also include food production and recreational uses, e.g., parks, forests, and green lands can be distinguished (Burghardt et al. 2015; Greinert 2017). Urban soils may have several simultaneous uses. For example, allotment gardens, representing a highly common land use in many cities all over the world, were established for food production. They are also increasingly considered as places of rest, allowing contact with nature (Ferris et al. 2001) and performing pro-environmental functions through biodiversity conservation (Matteson et al. 2008; Goddard et al. 2010; Speak et al. 2015) or ecological corridors for fragmented habitats in cities. The most common type of greenery in many Polish cities and towns is the lawn (Charzyński et al. 2013b). Within city borders, a small percentage of soils is used as farmland.
Soils in urban areas are usually transformed, destroyed, or often completely man-made (Charzyński et al. 2013a; Burghardt et al. 2015; Hulisz et al. 2018). A common phenomenon is covering the soil surface with humus materials, which are transported from other places (Greinert 2013). Such treatments are often applied by owners of garden plots in order to improve the condition of their soils and during the establishment of green areas. The migration of people from city centers to the suburbs has made the latter areas increasingly integrated into cities and transformed by human intervention (Oktaba et al. 2014). This has caused a decline in their original agricultural, forest-related, or other natural character. These processes have occurred for hundreds of years and; in many present urban soils, signs of past activity can be found (He and Zhang 2009; Krupski et al. 2017).
Soils of urban areas play a significant role in the daily lives of the respective inhabitants (Morel et al. 2017). Therefore, knowing their properties is particularly important. Recently, attention has been paid to the possibility of accumulating large amounts of organic carbon in urban soils (Pouyat et al. 2002, 2006; Churkina et al. 2010; Brown et al. 2012; Edmondson et al. 2012, 2014; Lorenz and Lal 2015; Burghardt and Schneider 2018). However, there are limited data on the amounts of different organic matter fractions and the humus quality in urban areas. Soil organic matter (SOM) is the most important soil component, responsible for many soil properties, such as sorption capacity, water retention, thermal properties, and soil resistance to pollution (Schnitzer 1978). Generally, three main fractions of humus compounds are distinguished according to their solubility in different solutions into: (I) humic acids (HA), which are soluble in diluted alkali and precipitated on the acidification of the alkaline extract; (II) fulvic acids (FA), which remain in the solution when the alkaline extract is acidified; and (III) humins HM, which cannot be extracted from the soil or sediment by diluted alkali or acid (Schnitzer and Khan 1972).
SOM content and quality differ depending on climate and soil conditions. The fractional composition is dependent on soil type; but, in an urban environment, it can be modified by anthropogenic substrates, such as coal, charcoal, char, soot (known as black carbon (BC) or elemental carbon (EC)), plastics, or different pollutants, e.g., polycyclic aromatic hydrocarbons (PAHs) (Lorenz et al. 2006; Agarwal and Bucheli 2011; Dębska et al. 2012; Klimkowicz-Pawlas et al. 2016). However, SOM content and quality in urban soils may be also dependent on the way they are used. The aim of the present study is to assess the impact of different urban land uses on the properties of SOM taking a selected town in Poland as an example.
2 Materials and methods
2.1 Site description and soil sampling
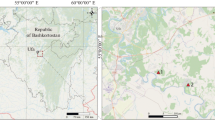
The study was carried out in Pruszków town which is located in the south-west part of the Warsaw Agglomeration, Central Poland. The town population in 2017 was 60,993 (Central Statistical Office 2017), whereas the total area in 2016 was 19 km2 (Central Statistical Office 2016). Soils occurring in areas representing four land uses were included in this study: lawns, allotment gardens, fallows, and arable lands within the administrative boundaries of the town. Lawns (in parks and along the streets) and allotment gardens were located in the central zone of the town. Arable lands and fallow lands were mostly located on the outskirts of Pruszków. The largest area of Pruszków is occupied by Pleistocene deposits, represented mainly by sand and gravels in the northern and central parts. Kame sands, sands and silts on fluvioglacial sands, and isolated areas were located in the southern part of the town. There are also sands and silts on the glacial tills in the southern part of the investigated area. Compact deposits are found only in the southern part of the town. These are mainly small areas occupied by Pleistocene glacial tills. The predominant types of soils in the study area are Haplic Luvisol, Albic Luvisol (Arenic), and Haplic Cambisol (Dystric) (Fic et al. 2004).
Humus horizons to a depth of 0–20 cm from 35 sites were analyzed (Fig. 1). Samples were collected from soils by means of a stainless-steel tube. Each of the bulk samples was mixed from numerous subsamples to obtain a total weight of 1 kg. Soil samples were air-dried, ground, and sieved by a 1-mm sieve in order to obtain fine earth.
2.2 Na-pyrophosphate extraction
SOM fractions were obtained by chemical extraction using the Kononova and Belchikova method (Dziadowiec and Gonet 1999). This involves performing Na-pyrophosphate extraction to separate the soluble SOM bound to the mineral surface via iron, aluminum, and calcium fraction (PY) from the non-hydrolysing residue, usually called humins (HM) (supposed to be organic matter occluded in mineral aggregates, and mainly comprises clay and silt of high mechanical stability).
A brief procedure is as follows. Samples of dried fine earth were ground to obtain particles of less than 0.25 mm. Each sample was mixed with a 0.1 M sodium pyrophosphate solution and a 0.1 M NaOH solution, then left overnight. After 24 h, the samples were filtered. Depending on the amount of carbon in the sample, extracts of 5 ml (dark extracts with higher carbon content) or 10 ml (bright extracts with less carbon content) were neutralized to pH 7.0 by the addition of 1 M of H2SO4. Next, the samples were evaporated to dryness on a water bath. The carbon content of this fraction was determined using the Tyurin titrimetric method (Ponomareva and Plotnikova 1980). The extracted humic substances included HA and FA. The amount of carbon in the remaining residuum, HM, was calculated by subtraction as follows: HM = SOC − PY, where SOC represents the total organic carbon and PY represents the carbon of the soluble fraction obtained during Na-pyrophosphate extraction.
2.3 Humic acid extraction
HAs were separated from the 0.1 M Na4P2O7 and 0.1 M NaOH extracts. In order to precipitate HA, a drop of concentrated H2SO4 was added to each sample to bring about a turbidity of the sample. For the complete precipitation of HA, the extracts were allowed to stand overnight. The samples were filtered and the precipitates were washed with 0.01 M of H2SO4 until a colorless filtrate was obtained. The HA precipitates collected on the filters were then dissolved with warm 0.02 M NaOH, heated to 80 °C. The flasks with HA precipitates were then filled up to a volume of 100 ml with 0.02 M NaOH and mixed (HA extracts). Next, subsamples of the extracts were taken and neutralized with 1 M of H2SO4 to pH 7.0. These were set on a water bath and evaporated to dryness. Carbon from the individual HA samples was determined using the Tyurin titrimetric method (Ponomareva and Plotnikova 1980).
Calculation of the quantity of carbon in FA structures was made on the basis of the equation FA= PY − HA, where PY represents the carbon of the soluble fraction obtained during Na-pyrophosphate extraction and HA represents the quantity of carbon in the HA structures. The HA/FA ratio was calculated as the amount of FA divided by the amount of HA. The percentage of carbon from each fraction in orgaSOC was calculated.
2.4 Spectroscopic method
HA extracts obtained in step 2.3 were transferred to cuvettes. A solution of 0.02 M of NaOH was used as the blank. The absorbance of solutions at wavelengths of 465 and 665 nm was measured. Measurements were made in duplicate. The color indexes (E4/E6) were calculated as the ratio of E465/E665 nm. Spectral properties of solutions were measured using an UV-Vis spectrometer (Genesys 10vis, Thermo Electron Corporation).
2.5 Sulfuric acid extraction
Separate soil samples were treated with 0.05 M of H2SO4. The samples were mixed and left until the next day. After 24 h, the samples were filtered, while the residues were washed with acidified water (one drop of concentrated H2SO4 mixed with 1 l of distilled water). This action was performed to remove calcium from the filtrate. Distilled water was added up to a volume of 250 ml to each sample. Then, the extracts were neutralized with NaOH and evaporated to dryness. The carbon was determined by the Tyurin titrimetric method (Ponomareva and Plotnikova 1980). The carbon compounds obtained in this extraction were considered as highly mobile LMW fractions of simple structures.
2.6 Statistical analysis
Multiple-sample comparison tests were conducted with the application of Statgraphics Plus 4.1 software. After verification of the occurrence of any significant differences between the mean values by means of the F test in the analysis of variance (ANOVA) table, multiple-range tests were applied in order to determine which mean values were significantly different from the remaining ones. In the cases where standard deviations were different, the Kruskal-Wallis test was applied to compare medians. Additionally, a simple regression analysis was used to describe the relationship between some soil properties such as soil organic carbon (SOC), total nitrogen (NT), C/N ratio, cation exchange capacity (CEC), content of silt and clay (particle < 0.05 mm), and content of different humus fractions or E4/E6 ratio.
The physical, physicochemical, and chemical properties of examined soils were described in detail in a previous works (Oktaba and Kusinska 2011; Oktaba et al. 2014), but some data were briefly described and used in this work to explain some phenomena or to find correlations with different humus compounds. Due to the lack of carbonates in the examined soils, all of the carbon in the samples was assumed to be organic carbon.
3 Results
3.1 Soil organic carbon and total nitrogen content for different land uses
The largest concentrations of SOC were found in the soils of allotment gardens and lawns. Statistically significant differences were found in the SOC quantities in soils of allotment gardens and lawns when compared to the average SOC content in the soils of arable lands and fallows (Table 1). Nitrogen content in the studied soils exhibited a slightly different tendency. The largest concentrations of this element were noted in the soils of allotment gardens, deviating significantly from the contents found in other soils (Table 1). The C/N ratio was high and similar in soils of four land uses. The cation exchange capacity (CEC) varied significantly between soils of allotment gardens and other soils. The clay content was extremely low in all studied soils (Table 1).
3.2 PY, HM, and LMW concentrations in soil representing different land uses
Humins (HM), which are humus compounds tightly bound to the mineral part of soil, predominated across form of humus in the studied soils (Fig. 2). The lowest concentrations of humins (5.7 g kg−1 of soil) were found in soil from arable land and fallows (6.3 g kg−1 of soil). The content of HM was two times higher in the soils of lawns and allotment gardens (Table 3).
Similarly, the concentrations of compounds extracted with sodium pyrophosphate (PY) in soils increased in the following order: arable land < fallows < lawns < allotment gardens (Table 2). Homogeneous groups were distinguished using statistical analyses (Kruskal-Wallis test). Both HM and PY fractions were the most abundant in the soils of allotment gardens and lawns that are in the central zone of the town, with their quantities differencing statistically from the amounts found in the soils of arable lands. The HM and PY contents in fallow soils were similar to those in the central zone as well as in arable land (Table 2). The amounts of each fraction in all of the studied soils correlated most strongly with the amount of SOC (Table 3).
In the case of low molecular weight (LMW) fractions, their values were very similar in soils of all uses and ranged from 0.4 g kg−1 of soil in allotment gardens and arable lands to 0.6 g kg−1 of soil in fallows (Table 2).
3.3 Percentage of carbon of PY, HM, and LMW compounds to soil organic carbon
Despite significant differences in the content of PY and HM in soils characterized by different uses, the percentage of carbon from individual humus compounds in the SOC content of the soil was very similar in all studied samples (Fig. 2). In the case of HM, it amounted to 65–71% of SOC. Regardless of such minor differences, a slightly higher percentage of HM carbon in SOC was observed in the central zone (average share of 70%) than in the peripheral zone (65%). The degree of humification, defined as the combined contribution of fulvic and humic acids (PY) in SOC, was low and averaged about 30% for all soils (Fig. 2). The share of LMW carbon to SOC ranged from 1.8 and 2% in the soils of allotment gardens and lawns to 4.5 and 5.0% in the soils of fallows and arable land.
3.4 Humic and fulvic acids in soils characterized by different land uses
The content of humic acids (HA) was two times higher in soils of allotment gardens or lawns when compared to that of arable lands (Table 2). Fulvic acids, however, prevailed in all soils, with an HA/FA ratio (where HA is the amount of humic acid carbon and FA is the amount of fulvic acid carbon) of less than one. The amount of fulvic acids was about seven times higher than the amount of humic acids, both in the central and peripheral zones.
3.5 UV-VIS spectra and E4/E6 ratio
On the basis of the E4/E6 ratio, we can observe that the least condensed humic acid molecules with lower molecular weight occurred in the soils of allotment gardens with an E4/E6 ratio of 5.6, followed by lawn and fallow soils (5.3 and 5.1 respectively). The most condensed humic acid molecules (E4/E6 ratio of 4.7) were present in the soil of arable land (Table 4).
4 Discussion
Humus substances are complex organic materials, which are important in many soil processes and they are the main components responsible for soil fertility. Soil organic matter (SOM) distribution in urban ecosystems is more variable than that in natural soils, due to the high heterogeneity of soils in urban areas (Lorenz et al. 2006). The formation of humic compounds with different structures and various properties is determined primarily by the chemical and physicochemical properties of the soil (Łabaz 2007). In agricultural environments, the additional factors modifying the original soil properties are fertilization, the cultivation method, and the types of plants cultivated (Medyńska-Juraszek 2016; West and Post 2002; Lorenz et al. 2006).
Properties of soils in Pruszków town varied both within the same way of use and between them, as presented elsewhere (Oktaba and Kusinska 2011; Oktaba et al. 2014). The differentiation of the texture within the studied soil was not substantial (Oktaba et al. 2014). ANOVA variance for mean silt and clay content (particles < 0.05 mm) using the F test did show differences only between the soils of allotment gardens and lawns (Table 1). Texture is one of the most important soil characteristics determining other soil properties, including the ability of the accumulation of SOM (Konen et al. 2003; Wang et al. 2010). West and Post (2002) also stated that texture, climate, and land use influence not only SOM content, but also its composition as well.
Despite the different kinds of soil use and different soil properties, such as texture and nitrogen content (Oktaba et al. 2014), soils of allotment gardens and lawns have been found to have a similar accumulation of organic carbon compounds. ANOVA (Fisher’s LSD procedure) allowed us to separate them into one homogeneous group. The second group, consisted of arable lands and fallows, indicated significant lower amount of soil organic carbon (SOC). The first group included soils from the central zone, whereas the second comprised soils located mainly on the outskirts of the town. Fallows and arable lands had common soil properties. In the case of allotment gardens and lawns, only the accumulation of SOC was similar, with the quality of humus expressed in the content of SOM fractions having a similar solubility. Thus, the manner of land use associated with the location of the soil and perhaps the degree of exposure to anthropogenic factors were more important factors determining the size of SOM accumulation and its composition than soil properties alone. Accumulation of SOC in allotment gardens could be related to organic fertilization of these soils in the form of compost, manure, or other organic fertilizers, which is a common practice among gardeners. Significant accumulation of carbon in soils of allotment gardens was noted by other authors (Burghardt and Schneider 2018). On the other hand, lawn soils are often covered with a peat layer before being installed.
Polish soils are generally characterized by low SOC content. Monitoring of the chemistry of Polish arable soils (website 2015) showed that the amount of SOM in 2015 was 1.12% on average (median 0.98%). The Masovian Voivodeship, in which Pruszków is located, is characterized by an average content of humus compared to Poland overall (< 2% SOM, which is about 1.2% SOC).
These types of soils made of silt and loam, as studied by Pisarek (2000), contained < 2% SOC. In the studies of other authors regarding Cambisols or Luvisols in the region of Pruszków, the amount of SOC was less than 1% (Chojnicki 1993a). It is only in anthropogenic soils that the carbon concentration exceeded 1% (Chojnicki 1993b). In the studied soils of Pruszków, quantities of SOC reached 2% on average, whereas in individual cases, the values exceeded 3% (Oktaba et al. 2014). It should be emphasized that these soils had sandy texture and low clay content, and they did not contain carbonates. Therefore, these are soils which properties are not favorable for the accumulation of SOM. In addition, organic matter in the studied soils of Pruszków was characterized by a high C/N ratio, which indicated the low availability of nitrogen for microorganisms and plants. In cultivated soils, C/N ratio is usually about 10 (10/1). In the studied agricultural soils, C/N ratio was above 21 (21/1), whereas in the soils of allotment gardens, which are usually enriched with mineral and organic fertilizers, the ratio was almost 20. In fallows and lawns, C/N ratios were even higher. A high C/N ratio results in disturbances in biological activity and disturbed processes of decomposition of SOM (Wang et al. 2010). The phenomenon of the increased accumulation of organic carbon in urban soils can be explained, among others, by insertion compounds derived from the combustion of fuels to soil. The enrichment of urban soils with organic carbon has been noted by other researchers (e.g., Pouyat et al. 2002; Edmondson et al. 2012).
The quality of soil humus is assessed on the basis of a number of different fractions and their composition. The quantitative and qualitative analysis of fractions of organic matter, differing in the degree of solubility, can provide valuable information on humus properties. For example, the solubility of SOM is related, to a certain degree, with the stability of SOM components (Ellerbrock and Kaiser 2005).
Extraction with pyrophosphate, which has an ability to extract humus-metal complexes, allowed us to separate humus forms connected with iron, aluminum, and calcium. Bindings of organic matter with trace elements are highly possible in the studied soils; however, this issue was not the subject of the present research. The number of compounds extracted in the course of pyrophosphate extraction (PY) was the largest in the soils of allotment gardens and lawns (central zone), but statistically differed from the amount in the soils located on the town’s periphery (arable lands and fallows). The dominant types of humus fractions in Polish soils are soluble compounds, PY fractions (Turski 1986). On the other hand, insoluble compounds such as humins (HM) predominate in soils of Pruszków, which are supposed to be mainly occluded in silt- and clay-sized microaggregates of high mechanical stability (Kayler et al. 2011). They constitute over 70% of the SOC in the central part of the town and 65% in the peripheral part, although, in the latter, the quantities of HM were half as much. Similar dependencies in urban soils were found in Warsaw and Łódź cities, central Poland (Kusińska et al. 1983; Kusińska 1991). According to these studies, HM accounted for 50–60% of SOC.
Due to dynamic development and the high level of traffic (Oktaba et al. 2014), soils in Pruszków town are strongly influenced by anthropogenic factors, in particular in the central part of the town.
According to Beyer et al. (2001), anthropogenic organic particles are possible sources of organic matter in urban soils, apart from natural humic substances. For example, in the majority of urban soil horizons, elemental carbon (EC) was found. EC is referred to by other researchers as black carbon (BC), which is produced by the incomplete combustion of fossil fuel and biomass contributing to the insertion of highly stable aromatic components to soils (Schmidt and Noack 2000). BC is ubiquitous in soil and sediment; however, it is present in larger quantities in urban environments. In Delhi, soil BC was found to be up to 23% of the SOC (Agarwal and Bucheli 2011). Apart from BC, other anthropogenic substrates may contribute to organic matter in urban soils (Lorenz et al. 2006). BC is very stable in the environment and has strong sorption properties.
The comparison of sorption properties in Pruszków soils with other soil properties (SOC, Nt, content of fraction < 0.05 mm, and various organic matter fractions) allowed us to determine those that were most related to the cation exchange capacity (CEC) of soils: there was no relationship between the granulometric composition and the sorption properties of these soils, but the presence of SOC (R = 0.64, p < 0.01) had significant influence on CEC. The fact that sorption abilities are associated with the SOM component was also noted by Ellerbrock and Kaiser (2005). From various forms of organic matter, the amount of carbon of HM (R = 0.54, p < 0.01) had a stronger effect on CEC than content of carbon occurring in easily soluble connections extracted by pyrophosphate solution, PY (R = 0.45, p < 0.01). This suggests that HM in the investigated soils may contain some BC. The concentration of BC in the urban area decreases from the inner parts of a city towards exterior areas (Liu et al. 2011), in the same way as HM content.
The mobile compounds of organic matter with the simplest structure (LMW) showed a small percentage in SOC of the studied soils. They accounted for 2% of SOC in the central zone of Pruszków town, and 5% of SOC in arable soils and fallows. This is consistent with the analysis of arable lands in Poland by Turski (1986).
In very young urban soil ecosystems, litter compounds and poorly humified FA fractions dominate in the SOM (Beyer et al. 1995). Pruszków used to be a small town about 100 years ago and has recently become part of the Warsaw agglomeration. In the studied soils of Pruszków, FAs comprised the predominant part of humified compounds. Their contents were seven times higher than the contents of HAs, as indicated by the low HA/FA ratio. In the cultivated Cambisols and Luvisols investigated by Pisarek (2000), the HA/FA ratio indicated the dominance of HA. Polláková et al. (2016) also found significant quantities of HA in Luvisols used as arable land. The low contents of HA in the studied soils (Table 2) may be caused by low contents of fine particles (e.g., clay fraction) and the lack of carbonates, which stabilize humus compounds. On the other hand, PAHs affect the deterioration in humus quality by changing the fractional composition of humus (Dębska et al. 2012; Banach-Szott et al. 2014). Therefore, pollutants may be another factors resulting in low contents of HA.
In the studied soils, HAs were characterized by a high E4/E6 ratio obtained by determining the optical density of HA solutions. This coefficient, as well as the study of the UV-Vis spectroscopic properties of HA solutions, is widely used. The method is simple and inexpensive, and provides important information on the structure of HA. Therefore, it is still commonly used throughout the world in studies on the properties of HA in soils and sediments (Giovanela et al. 2010; Polak et al. 2011; Traversa et al. 2014), as well as in the evaluation of stumps and root decomposition (Zhou et al. 2016). Optical density is one of the most informative indices of the properties and structure of humic substances and, consequently, reflects the direction of transformation of humus under different land use conditions (Zav’yalova 2012). This method was also used for HA assessment in urban soils (Markiewicz et al. 2017).
In many studies, a significant relationship has been found between the optical density of HA solutions and the aromaticity of HA (Tinoco et al. 2015). HA solutions containing more aromatic components are more condensed and characterized by higher optical density compared to acid solutions, where aliphatic chains are more abundant in the structure. However, Chen et al. (1977) found no relation between the E4/E6 ratios and the degree of aromaticity but observed inverse relationship with molecular weight for humic substances. The E4/E6 ratio appears to be independent of humus substances concentration, but varies with molecular size and the genesis of SOM in diverse soil environments (Schnitzer 1978). The lower ratio indicates a higher degree of humification (Gryndler et al. 2010; Zhou et al. 2016). The higher the E4/E6 ratio, the lower the hydrophobicity (Trubetskaya 2013). The structure and composition of HA are largely dependent on the type of anthropogenic impact (Gorbov et al. 2018).
A higher E4/E6 ratio was found in allotment gardens and lawns than in arable lands and fallows of the study area. Referring our data to the research cited above, we can conclude that the soils in the city center were characterized by humus with inferior properties, expressed in lower molecular weight or lower degree of aromaticity. It was surprising that the CEC of soil was not correlated with the ratio E4/E6. One might expect that the lower the ratio (i.e., the higher the HA maturity), the higher the sorption capacity of the soil would be.
5 Conclusions
The content of organic matter in urban soils of Pruszków town varied depending on the land use. The largest accumulation occurred in soils used as allotment gardens and lawns occurring in the central part of the town. Soil organic carbon (SOC) content in soils from that zone differed significantly from values obtained in the soils located in the peripheral zones of the town used as arable land and fallows. Organic matter in the investigated soils mainly consisted of humus compounds not soluble in Na-pyrophosphate solution of high stability, referred to as humins (HM). Concentration of carbon in PY compounds was minor. Content of carbon in PY and HM fractions was the highest in allotment gardens and lawns. Lower concentrations of these compounds were found in fallow soils, with the least noted in soils of arable lands. The amount of individual organic matter fractions was not correlated with the contents of particle-size fraction < 0.05 mm. SOC was most strongly correlated with the content of HM carbon. Weaker correlation was found between SOC and concentration of PY carbon.
Referring to previous studies (Oktaba et al. 2014), SOC was found to influence cation exchange capacity (CEC). Among the humus fractions, humins exerted the strongest influence on CEC. These data indicate that HM may include highly aromatic black carbon compounds. PY fraction compounds were characterized by a simple structure. The ratio of contents of humic acids to contents of fulvic acids (HA/FA) was extremely low (0.10–0.20 on average), which means that fulvic acids predominated in all analyzed soils. Humic acids under study had a low aromatic structure as indicated by optical density measurements of HA solutions. A high E4/E6 ratio indicates low maturity of this humic fraction with low molecular weight compounds.
Change history
30 August 2018
The article “The impact of different land uses in urban area on humus quality”, written by Lidia Oktaba, Diana Odrobińska, and Łukasz Uzarowicz was originally published electronically on 7 April 2018 with incorrect copyright line in the publisher’s internet portal (currently SpringerLink).
30 August 2018
The article ���The impact of different land uses in urban area on humus quality���, written by Lidia Oktaba, Diana Odrobi��ska, and ��ukasz Uzarowicz was originally published electronically on 7 April 2018 with incorrect copyright line in the publisher���s internet portal (currently SpringerLink).
References
Agarwal T, Bucheli TD (2011) Is black carbon a better predictor of polycyclic aromatic hydrocarbon distribution in soils than total organic carbon? Environ Pollut 159:64–70
Banach-Szott M, Dębska B, Rosa E (2014) Effect of soil pollution with polycyclic aromatic hydrocarbons on the properties of humic acids. J Soils Sediments 14:1169–1178
Beyer L, Blume HP, Elsner DC, Willnow A (1995) Soil organic matter composition and microbial activity in urban soils. Sci Total Environ 168:267–278
Beyer L, Kahle P, Kretschmer H, Wu Q (2001) Soil organic matter composition of man-impacted urban sites in North Germany. J Plant Nutr Soil Sci 164:359–364
Brown S, Miltner E, Cogger C (2012) Carbon sequestration potential in urban soils. In: Carbon Sequestration in Urban Ecosystems, pp 173–196
Burghardt W, Schneider T (2018) Bulk density and content, density and stock of carbon, nitrogen and heavy metals in vegetable patches and lawns of allotments gardens in the northwestern Ruhr area, Germany. J Soils Sediments 18:407–417
Burghardt W, Morel JL, Zhang GL (2015) Development of the soil research about urban, industrial, traffic, mining and military areas (SUITMA). Soil Sci Plant Nutr Supplementary Issue: Soils of Anthropized Environments 61(Supplement 1):3–21
Central Statistical Office (GUS) (2016) Area and population in the territorial profile in 2016. Warsaw
Central Statistical Office (GUS) (2017) Population in Poland. Size and structure by territorial division as of June 30, 2017. Warsaw
Charzyński P, Bednarek R, Hulisz P, Zawadzka A (2013a) Soils within Toruń area. In: Charzyński P, Hulisz P, Bednarek R (eds) Technogenic soils of Poland. Polish Society of Soil Science, Toruń, pp 17–29 (in Polish)
Charzyński P, Bednarek R, Różański SZ, Mendyk Ł, Morawski B (2013b) Lawn soils of Toruń and Bydgoszcz. In: Charzyński P, Hulisz P, Bednarek R (eds) Technogenic soils of Poland. Polish Society of Soil Science, Toruń, pp 55–80 (in Polish)
Chen Y, Senesi N, Schnitzer M (1977) Information provided on humic substances by E4/E6 ratios. Soil Sci Soc Am J 41:352–358
Chojnicki J (1993a) Brown soils developed from superficial formations of the Błonie-Sochaczew plain. Roczniki Gleboznawcze-Soil Sci Annu 44(1/2):93–106 (in Polish)
Chojnicki J (1993b) Lessivés soils developed from superficial formations of the Błonie-Sochaczew plain. Roczniki Gleboznawcze-Soil Sci Annu 44(3/4):135–151 (in Polish)
Churkina G, Brown DG, Keoleian G (2010) Carbon stored in human settlements: the conterminous United States. Glob Chang Biol 16:135–143
Dębska B, Banach-Szott M, Rosa E, Pakuła J (2012) The effect of soil pollution by selected PAHs on the fractional composition of organic matter. Roczniki Gleboznawcze-Soil Sci Annu 63(1):13–17 (in Polish)
Dziadowiec H, Gonet SS (1999) Methodical guide-book for soil organic matter studies. Soil Science Society of Poland, Warszawa, pp 52–54 (in Polish)
Edmondson JL, Davies ZG, McHugh N, Gaston KJ, Leake JR (2012) Organic carbon hidden in urban ecosystems. Sci Rep 2:963. https://doi.org/10.1038/srep00963
Edmondson JL, Davies ZG, McCormack SA, Gaston KJ, Leake JR (2014) Land-cover effects on soil organic carbon stocks in a European city. Sci Total Environ 472:444–452
Ellerbrock RH, Kaiser M (2005) Stability and composition of different soluble soil organic matter fractions—evidence from δ13C and FTIR signatures. Geoderma 128:28–37
Ferris J, Norman C, Sempik J (2001) People, land and sustainability: community gardens and the social dimension of sustainable development. Soc Policy Adm 35:559–568
Fic M, Kregiel J, Mierzwicki K, Katrynski W (2004) Program Ochrony Środowiska dla Miasta Pruszków. Falenty (in Polish)
Giovanela M, Crespo JS, Antunes M, Adamatti DS, Fernandes AN, Barison A, da Silva CWP, Guégan R, Motelica-Heino M, Sierra MMD (2010) Chemical and spectroscopic characterization of humic acids extracted from the bottom sediments of a Brazilian subtropical microbasin. J Mol Struct 981:111–119
Goddard MA, Dougill AJH, Benton TG (2010) Scaling up from gardens: biodiversity conservation in urban environments. Trends Ecol Evol 25:90–98
Gorbov SN, Bezuglova OS, Tischenko SA, Gorovtsov AV (2018) Organic matter and elemental composition of humic acids in soils of urban areas: the case of Rostov agglomeration. In: Vasenev et al (eds) Megacities 2050: environmental consequences of urbanization, Springer Geography. https://doi.org/10.1007/978-3-319-70557-6_10
Greinert A (2013) Urban Soils in Zielona Góra. In: Charzyński P, Hulisz P, Technogenic BR (eds) Soils of Poland. Polish Society of Soil Science, Toruń, pp 31–54 (in Polish)
Greinert A (2015) The heterogeneity of urban soils in the light of their properties. J Soils Sediments 15:1725–1737
Greinert A (2017) Functions of soils in the urban environment. In: Levin MJ, Kim KHJ, Morel JL, Burghardt W, Charzynski P, Shaw RK (eds) Soils within Cities. Catena-Schweitzerbart, Stuttgart, pp 43–52
Gryndler M, Soukupová L, Gryndlerová H, Baldrian P, Hršelová H (2010) Local distribution of ectomycorrhizae-associated basidiomycetes in forest soil correlates with the degree of soil organic matter humification and available electrolytes. Folia Microbiol 55:454–460
He Y, Zhang GL (2009) Historical record of black carbon in urban soils and its environmental implications. Environ Pollut 157(10):2684–2688
Hulisz P, Charzyński P, Greinert A (2018) Urban soil resources of medium-sized cities in Poland: a comparative case study of Toruń and Zielona Góra. J Soils Sediments 18:58–372
Kayler ZE, Kaiser M, Gessler A, Ellerbrock RH, Sommer M (2011) Application of δ13C and δ15N isotopic signatures of organic matter fractions sequentially separated from adjacent arable and forest soils to identify carbon stabilization mechanisms. Biogeosciences 8:2895–2906
Klimkowicz-Pawlas A, Smreczak B, Ukalska-Jaruga A (2016) The impact of selected soil organic matter fractions on the PAH accumulation in the agricultural soils from areas of different anthropopressure. Environ Sci Pollut Res Int 24(12):10955–10965
Konen ME, Burras CL, Sandor JA (2003) Organic carbon, texture, and quantitative color measurement relationships for cultivated soils in North Central Iowa. Soil Sci Soc Am J 67:1823–1830
Krupski M, Kabala C, Sady A, Gliński R, Wojcieszak J (2017) Double-and triple-depth digging and Anthrosol formation in a medieval and modern-era city (Wrocław, SW Poland). Geoarchaeological research on past horticultural practices. Catena 153:9–20
Kusińska A (1991) Transformation of organic matter in soils of greens and parks of the Łódź city. Roczniki Gleboznawcze-Soil Sci Ann 42(1/2):101–107 (in Polish)
Kusińska A, Łakomiec I, Usakiewicz T (1983) Changes in the fraction composition of soil humus of green areas in Warsaw. Pol Ecol Stud 9(1–2):45–54 (in Polish)
Łabaz B (2007) Fractional composition of humus in black earths in Wrocław district. Roczniki Gleboznawcze-Soil Sci Ann 58:69–83 (in Polish)
Liu S, Xia X, Zhai Y, Wang R, Liu T, Zhang S (2011) Black carbon (BC) in urban and surrounding rural soils of Beijing, China: spatial distribution and relationship with polycyclic aromatic hydrocarbons (PAHs). Chemosphere 82:223–228
Lorenz K, Lal R (2015) Managing soil carbon stocks to enhance the resilience of urban ecosystems. Carbon Manag 6:35–50
Lorenz K, Preston CM, Kandeler E (2006) Soil organic matter in urban soils: estimation of elemental carbon by thermal oxidation and characterization of organic matter by solid-state 13C nuclear magnetic resonance (NMR) spectroscopy. Geoderma 130:312–323
Markiewicz M, Hulisz P, Charzyński P, Piernik A (2017) Characteristics of soil organic matter of edifisols—an example of techno humus system. Appl Soil Ecol. https://doi.org/10.1016/j.apsoil.2017.04.022
Matteson KC, Ascher JS, Langellotto GA (2008) Bee richness and abundance in New York City urban gardens. Ann Entomol Soc Am 101:140–150
Medyńska-Juraszek A (2016) Biochar as a soil amendment. Soil Sci Annu 67:151–157 (in Polish)
Morel JL, Burghardt W, Kim KHJ (2017) The challenges for soils in the urban environment. In: Levin MJ, Kim KHJ, Morel JL, Burghardt W, Charzynski P, Shaw RK (eds) Soils within Cities. Catena-Schweitzerbart, Stuttgart, pp 1–6
Oktaba L, Kusinska A (2011) Mineral nitrogen in soils of different land use. Ecol Chem Eng A 18:585–592
Oktaba L, Paziewski K, Kwasowski W, Kondras M (2014) The effect of urbanization on soil properties and soil organic carbon accumulation in topsoil of Pruszków—a medium-sized city in the Warsaw Metropolitan Area, Poland. Soil Sci Annu 65:10–17
Pisarek I (2000) Physicochemical properties of humic acids extracted from Luvisols, Cambisols and Phaeozems. Roczniki Gleboznawcze - Soil Sci Ann al 51:19–27 (in Polish)
Polak J, Bartoszek M, Żądło M, Kos A, Sułkowski WW (2011) The spectroscopic studies of humic acid extracted from sediment collected at different seasons. Chemosphere 84:1548–1555
Polláková N, Jonczak J, Szlováková T, Kolenčík M (2016) Soil organic matter quantity and quality of land transformed from arable to forest. J Cent Eur Agric 17(3):661–674
Ponomareva and Plotnikova (1980) Humus and soil formation (methods and results). Nauka, Moscow (in Russian)
Pouyat RV, Groffman O, Yesilonis I, Hernandez L (2002) Soil carbon pools and fluxes in urban ecosystems. Environ Pollut 116:107–118
Pouyat RV, Yesilonis ID, Nowak DJ (2006) Carbon storage by urban soils in the United States. J Environ Qual 35:1566–1575
Schmidt MWI, Noack AG (2000) Black carbon in soils and sediments: analysis, distribution, implications, and current challenges. Glob Biogeochem Cycles 14:777–793
Schnitzer M (1978) Soil organic matter. Elsevier Science Publishers BV, Amsterdam
Schnitzer M, Khan SU (1972) Humic substances in the environment. Marcel Dekker Press, New York
Speak AF, Mizgajski A, Borysiak J (2015) Allotment gardens and parks: provision of ecosystem services with an emphasis on biodiversity. Urban For Urban Green 14(4):772–781
Tinoco P, Almendros G, González-Vila FJ, Sanz J, González-Pérez JA (2015) Revisiting molecular characteristics responsive for the aromaticity of soil humic acids. J Soils Sediments 15:781–791
Traversa A, D’Orazio V, Mezzapesa GN, Bonifacio E, Farrag K, Senesi N, Brunetti G (2014) Chemical and spectroscopic characteristics of humic acids and dissolved organic matter along two Alfisol profiles. Chemosphere 111:184–194
Trubetskaya O (2013) Determination of hydrophobicity and optical properties of soil humic acids isolated by different methods. J Geochem Explor 132:84–89
Turski R (1986) Composition of humus compounds in polish soils. Roczniki gleboznawcze-Soil Sci Annu 37(2/3):75–89 (in Polish)
Wang DD, Shi XZ, Wang HJ, Weindorf DC, Yu DS, Sun WX, Ren HY, Zhao YC (2010) Scale effect of climate and soil texture on soil organic carbon in the uplands of northeast China. Pedosphere 20:525–535
Website (2015) http://www.gios.gov.pl/chemizm_gleb/index.php?mod=wyniki&cz=C (in Polish)
West TO, Post WM (2002) Soil carbon sequestration rates by tillage and crop rotation: a global data analysis. Soil Sci Soc Am J 66:930–1946
Zav’yalova NE (2012) Investigation of optical properties of humic acids of sod-podzolic soil. Russ Agric Sci 38(5–6):386–388
Zhou J, Wu F, Yang W, Tan B, Xu Z, Zhang J, Duan F, Liu H, Justine MF (2016) The mass loss and humification of stumps and roots in Masson pine plantations based on log file records. PLoS One 11(8):e0160913. https://doi.org/10.1371/journal.pone.0160913
Acknowledgements
We gratefully acknowledge people who allowed us to collect soil samples at different sites in Pruszków town. We would like to thank Dr. Dariusz Gozdowski, Department of Experimental Design and Bioinformatics, Warsaw University of Life Sciences – SGGW, for his kind help in statistical calculations. The editors and anonymous reviewers are acknowledged for their helpful suggestions, which improved the previous draft of the paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Yona Chen
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Oktaba, L., Odrobińska, D. & Uzarowicz, Ł. The impact of different land uses in urban area on humus quality. J Soils Sediments 18, 2823–2832 (2018). https://doi.org/10.1007/s11368-018-1982-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-018-1982-7