Abstract
Experiencing decline in both cognition and mobility is associated with a substantially higher dementia risk than cognitive decline only. Metabolites associated with both cognitive and mobility declines may be early predictors of dementia and reveal specific pathways to dementia. We analyzed data from 2450 participants initially free of dementia who had 613 metabolites measured in plasma in 1998–1999 (mean age = 75.2 ± 2.9 years old, 37.8% Black, 50% women) from the Health, Aging and Body Composition study. Dementia diagnosis was determined by race-specific decline in 3MS scores, medication use, and hospital records through 2014. Cognition and mobility were repeatedly measured using 3MS and a 20-m walking test up to 10 years, respectively. We examined metabolite associations with changes in 3MS (n = 2046) and gait speed (n = 2019) using multivariable linear regression adjusted for age, sex, race, and baseline performance and examined metabolite associations with dementia risk using Cox regression. During a mean follow-up of 9.3 years, 534 (21.8%) participants developed dementia. On average, 3MS declined 0.47/year and gait declined 0.04 m/sec/year. After covariate adjustment, 75 metabolites were associated with cognitive decline, and 111 metabolites were associated with gait decline (FDR-adjusted p < 0.05). Twenty-six metabolites were associated with both cognitive and gait declines. Eighteen of 26 metabolites were associated with dementia risk (p < 0.05), notably amino acids, glycerophospholipids (lysoPCs, PCs, PEs), and sphingolipids. Results remained similar after adjusting for cardiovascular disease or apolipoprotein E ɛ4 carrier status. During aging, metabolomic profiles of cognitive decline and mobility decline show distinct and shared signatures. Shared metabolomic profiles suggest that inflammation and deficits in mitochondria and the urea cycle in addition to the central nervous system may play key roles in both cognitive and mobility declines and predict dementia. Future studies are warranted to investigate longitudinal metabolite changes and metabolomic markers with dementia pathologies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Declines in mobility and cognition are common in aging, and both are critical to one’s quality of life and independence in late life. Biomarkers associated with cognitive decline and mobility decline may provide clues about the mechanisms underlying these aging phenotypes. Dual decline in mobility and cognition is associated with a substantially higher dementia risk than cognitive decline only [1,2,3]. Shared biomarkers may be important early predictors of dementia and may explain the biological underpinnings of the connection between dual decline and high dementia risk [4].
There is growing evidence that the study of blood-based omics data, such as metabolomics, can contribute to understanding mechanisms underlying aging phenotypes. Metabolomics quantifies small molecules that in aggregate suggest that specific biological processes and pathways contribute to the development of a specific phenotype. Specific metabolites and pathways have been related to cognition and gait speed, but data are mostly from cross-sectional studies. Evidence to date suggests that sphingolipids, sphingolipid metabolism pathway, biosynthesis of the unsaturated fatty acids and amino acids, and the kynurenine pathway may contribute to both cognition and mobility impairments in aging [5,6,7,8,9,10,11,12]. These markers and pathways are related to apoptosis and proliferation, inflammation, protein synthesis, neurotransmitters, and mitochondrial dysfunction. However, studies often examined metabolomic markers of cognition and mobility separately. Only a few recent studies examined metabolomic associations with mobility and cognitive decline simultaneously, but how these metabolites were related to dementia risk are not further explored [13, 14].
In this longitudinal study of community-dwelling older adults initially free of dementia, we aimed to identify metabolomic markers of cognitive decline and mobility decline over a decade of follow-up and test the hypothesis that a group of shared metabolomic markers exist that predict future dementia.
Methods
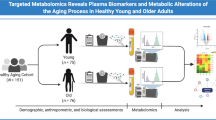
Study populations
Study participants were drawn from the Health, Aging, and Body Composition (Health ABC) study. The Health ABC study is a study focusing on functional decline that began in 1997–1998 with annual follow-up visits, enrolling 3075 participants aged 70–79 years at study entry from two study sites, Pittsburgh PA, and Memphis, TN. Inclusion criteria at study entry included no reported difficulty walking a quarter mile, climbing up ten steps, not reporting disability in basic activities of daily living, no use of a walking aid, no history of cancer treatment in the past 3 years, and no plan to move out of the study area within 3 years after recruitment. Plasma metabolomics were assayed by the Broad Institute (Cambridge, MA) using plasma samples collected at year 2 visit (1998–1999) from 2469 participants. We excluded 19 individuals with prevalent dementia from the sample for metabolomics, leaving 2450 participants in this analysis. Of these participants, we examined metabolomic profiles of cognitive decline among those who had repeated measures of 3MS (n = 2046) and metabolomic profiles of mobility decline in those with repeated measures of usual gait speed (n = 2019) since year 1 up to year 10 visits. The institutional review boards of the University of Pittsburgh, the University of Tennessee, the University of California-San Francisco Coordinating Center, and the National Institute on Aging approved the Health ABC study. All participants provided written informed consent at each visit.
Plasma metabolomics
Plasma metabolomics were assayed using liquid chromatography-mass spectrometry (LC–MS). Blood samples at the year 2 visit in 1998–1999 were drawn after overnight fasting and were stored at − 80 ℃ until metabolite profiling in 2019 − 2021. Details of the metabolite profiling were previously published [15, 16]. Four complimentary LC–MS methods were used, measuring (1) lipids (C8-positive); (2) a variety of amino acids, acylcarnitines, dipeptides, and other cationic polar metabolites (hydrophilic interaction liquid chromatography [HILIC]-positive); (3) fatty acids, eicosanoids, and bile acids (C18-negative); and (4) sugars, organic acids, nucleotides, and anionic polar metabolites (HILIC-negative). Because four LC–MS methods were used to quantify metabolites, metabolites could be measured in more than one platform. In this analysis, we analyzed all 613 available metabolites including 520 unique metabolites. Missing values of the metabolites considered below the limit of detection were imputed using half of the minimum. Metabolite values were then log-transformed and computed as standard Z scores for analysis.
Cognitive function and mobility
Global cognitive status was measured using the Modified Mini-Mental State Exam (3MS) at years 1, 3, 5, 8, and 10. Mobility was measured using a 20-m walk test at years 1, 2, 3, 4, 5, 6, 8, and 10.
Dementia diagnosis
Based on a previously developed algorithm in the Health ABC study, dementia was determined by hospital records, the use of prescribed medications for dementia (galantamine, rivastigmine, memantine, donepezil, or tacrine), or race-specific decline in 3MS greater than 1.5 SDs from the baseline mean [17].
Statistical analysis
To determine metabolomic markers of cognitive decline and gait decline, we examined the associations between each metabolite and slopes of 3MS and slopes of gait speed, separately, using multivariable linear regression, adjusted for age, sex, race, and baseline 3MS and baseline gait speed, respectively. Slopes of 3MS and gait speed were first computed using linear mixed-effects model using all available data since year 1 up to year 10 visits. To understand whether metabolomic markers of baseline performance and change over time share similarities, we also examined the associations between each metabolite and baseline 3MS and baseline gait speed using multivariable linear regression, adjusted for age, sex, and race. The significance for metabolite associations with gait and cognition was set at FDR-adjusted p < 0.05.
For metabolites that were significantly associated with both gait decline and cognitive decline, we further examined their associations with dementia risk using Cox regression, adjusted for age, sex, and race, and additionally adjusted for the prevalence of cardiovascular disease at metabolomics assessment and apolipoprotein E (APOE) ε4 carrier status in those with available data (n = 2328). Significance for specific metabolite associations with dementia risk was set at p < 0.05.
To understand whether metabolomic markers of cognition were correlated with metabolomic markers of gait speed, we created metabolite scores using the least absolute shrinkage and selection operator (LASSO) models and tested the associations between LASSO metabolite scores. We also performed pathway analysis when there was sufficient information on the human metabolomic database (HDMB) identification numbers matching the current database via MetaboAnalyst.ca. Significance for associations between LASSO metabolite scores and pathway analysis was reported at p < 0.05. All analyses were performed using R studio 4.3.1 (Boston, MA).
Results
Participants’ characteristics at the time of metabolomics assessment are presented in Table 1. The participants, including 50% women, were in their 70 s with a relatively narrow age range between 69.4 and 82.0 years; 37.8% were Black, 76% had greater than high school education, and 28.9% were APOE ε4 carriers. During a mean follow-up of 9.3 years, 534 (21.8%) participants developed dementia. The incidence rate was 23.4 per 1000 person-years.
Metabolites with cognition
Over the 10-year follow-up, the average decline of 3MS was 0.47 points per year (Table 1). Of the 613 metabolites examined, 70 metabolites were associated with baseline 3MS, and 75 metabolites were associated with change in 3MS up to 10 years (FDR-adjusted p < 0.05) (Table 2, Supplementary Table 1). Fifteen metabolites were associated with both baseline 3MS and decline in 3MS and showed consistent directions. Of these, nine metabolites were positively associated with baseline 3MS and change in 3MS, and six metabolites were negatively associated with baseline 3MS and change in 3MS. These 15 metabolites included amino acids (alpha-N-Phenylacetylglutamine via both C18n and HILICp platforms, homocitrulline, N-Methyl-proline, tryptophan), carbohydrates (P-Cresol glucuronide, glyceric acid), phospholipids (LPC (22:5), SM (d18:1/14:0 via C8p and HILICp platforms), pyrimidines (uracil, uridine), fatty acids (myristoleic acid), prenol lipids (maslinic acid), and steroids (CE(14:0) (FDR-adjusted p < 0.05). Some metabolites were implicated in pathways of pyrimidine metabolism (uracil, uridine), glycerolipid metabolism (glyceric acid), and pantothenate and CoA biosynthesis (uracil) (p < 0.05). The LASSO metabolite score of baseline 3MS was associated with the LASSO metabolite score of changes in 3MS (β = 0.308, p < 0.001) (n = 2046) (Supplementary Fig. 1).
Metabolites with gait speed
Over the 10-year follow-up, the average decline in gait speed was 0.04 m/sec per year. There were 197 metabolites associated with baseline gait speed, and 111 metabolites were associated with change in gait speed up to 10 years (FDR-adjusted p < 0.05) (Table 2, Supplementary Table 1). There were 57 metabolites associated with both baseline gait speed and gait decline and showed consistent directions for cross-sectional and longitudinal associations. Of these, 30 metabolites were positively associated with baseline gait speed and change in gait speed, and 27 were negatively associated with baseline gait speed and change in gait speed. These 57 metabolites mainly included glycerophospholipids, amino acids, carbohydrates, fatty acids, and triacylglycerols. Of these 57, metabolites from glycerophospholipids (lysoPCs, PCs), triacylglycerols (TGs), some amino acids (homoarginine, tryptophan), and pyrimidines (uracil) were all positively associated with gait speed or change in gait speed (i.e., upregulated), and metabolites from carbohydrates (such as glucuronic acid, threitol) and fatty acids (such as CAR, adrenic acid, eicosenoic acid) and some amino acids (such as alpha-N-phenylacetylglutamine) were all negatively associated with gait speed or change in gait speed (i.e., downregulated). Some of these metabolites were implicated in pantothenate and CoA biosynthesis, citrate cycle (TCA cycle), beta-alanine metabolism, and pyrimidine metabolism (p < 0.05). The LASSO metabolite score of baseline gait speed was associated with the LASSO metabolite score of change in gait speed (β = 0.470, p < 0.001) (n = 2019) (Supplementary Fig. 2).
Shared metabolites of cognitive decline and gait decline with dementia risk
There were 26 metabolites associated with both change in 3MS and change in gait speed in consistent directions (FDR-adjusted p < 0.05) (Table 2) (Fig. 1). Most metabolites were positively associated with change in 3MS and change in gait speed, including glycerophospholipids (lysoPCs, PCs), sphingolipids (SM (d18:1/14:0), amino acids (tryptophan), and pyrimidines (uracil). Five metabolites were negatively associated with change in 3MS and change in gait speed, including fatty acids (CAR (4:0), adrenic acid, eicosenoic acid), amino acids (alpha-N-phenylacetylglutamine), and N2,N2-dimethylguanosine (Table 2). The LASSO metabolite score of change in 3MS was associated with the LASSO metabolite score of change in gait speed (β = 0.494, p < 0.001) (n = 1810) (Fig. 1).
Metabolites associated with gait decline, cognitive decline, and dementia risk. a Number of metabolites associated with cognitive decline, gait decline, and incident dementia. b Scatter plot of the association between the LASSO metabolite score of cognitive decline (x-axis) and LASSO metabolic score of gait decline (y-axis)
Of these 26 metabolites, 18 were significantly associated with dementia risk (p < 0.05) (Table 2). Most of these metabolites were upregulated; higher concentrations were associated with a reduced risk of dementia, including lysoPC (20:5), sphingomyelins, tryptophan, uracil, CE (20:5), PCs, PEs, and PS. Other metabolites, including adrenic acid, eicosenoic acid, and alpha-N-phenylacetylglutamine, were downregulated; higher concentrations were associated with an increased risk of dementia. Results remained similar after further adjustment for APOE ɛ4 status or prevalence of cardiovascular disease at metabolomics assessment, while adjustment for APOE ɛ4 status attenuated some associations more than adjustment for cardiovascular disease (Fig. 2).
Dot plots of associations between 18 shared metabolomic markers of cognitive decline and gait decline and incident dementia with additional adjustment for apolipoprotein E ɛ4 (APOE ɛ4) status and cardiovascular disease. Legend: M1-covariate adjustment results same as Table 2. M2-additional adjustment for APOE ɛ4 carrier status. M3-additional adjustment for cardiovascular disease
Discussion
In this biracial cohort of community-dwelling older adults, we identified metabolomic markers of longitudinal cognitive decline and gait speed decline over a decade. Metabolomic profiles of cognitive decline and mobility decline show both unique and shared biomarkers. Some of the shared markers predicted dementia risk which provide additional insights into mechanisms underlying dual cognitive and mobility decline in the development of dementia.
Among all the 613 metabolites examined, we identified 75 metabolites associated with cognitive decline after adjustment for baseline cognitive function. The metabolomic profiles are mainly derived from amino acids, fatty acids, glycerophosphocholines, and phosphosphingolipids. We confirmed some of these metabolites which were previously reported in either cross-sectional or longitudinal studies of cognitive function, such as alpha-N-phenylacetylglutamine, tryptophan, and PC (38:6) [5]. Alpha-N-phenylacetylglutamine is the main amino acid from the phenylacetate degradation in the urea cycle and high levels of which are implicated in urea cycle disorders, digestive disorders, and deficits in the nervous system. Tryptophan is an essential alpha-amino acid that cannot be synthesized de novo. It is one of the precursors to the neurotransmitter serotonin and of the nicotinamide adenine dinucleotide (NAD +) and nicotinamide adenine dinucleotide phosphate (NADP +), which are essential co-factors in bioenergetic and biosynthesis pathways. PC (38:6) is a glycerophospholipid that includes palmitic and docosahexaenoic fatty acids, which are particularly abundant in fish oils. PC (38:6) has been identified in vascular disorders, digestive system disorders, and other conditions. We identified additional metabolites related to cognitive decline which have not been reported before. For instance, other amino acids (such as homocitrulline), fatty acids (such as adrenic acid, arachidic acid, eicosenoic acid, oleic acid, and stearic acid), other glycerophosphocholines (such as lysoPCs, PCs, LPEs, and PEs), phosphosphingolipids (SM (d18:1/14:0) and S1P), pyrimidines (such as uracil and uridine), maslinic acid, serotonin, carboxyibuprofen, and sphingosine 1-phosphate were also associated with cognitive decline. Uridine and uracil are essential components of DNA and RNA, important to maintain genetic integrity and active transcription of enzyme and signaling molecules [18]. Adrenic acid is a polyunsaturated fatty acid and is involved in alpha-linolenic acid and linoleic acid metabolism. It is abundant in human brains during early development, and some evidence suggests that oxidation of adrenic acid may contribute to cerebral white matter damage [19]. Oleic acid is classified as a monounsaturated omega-9 fatty acid and is the most abundant fatty acid in human adipose tissue. Maslinic acid is a pentacyclic triterpene and shown to have anti-inflammatory, antioxidant, and neuroprotective effects [20]. It is abundant in olives [21, 22] and can also be found in other vegetables and fruits, such as loquat leaves [23], red dates [24, 25], and plantains [26]. SM (d18:1/14:0) is a sphingosine backbone with a myristic acid chain. It is especially found in myelin sheaths surrounded by nerve cell axons. Serotonin is a major neurotransmitter involved in various physiological processes, including digestion, mood, and sleep. Ibuprofen is commonly used to treat pain and reduce inflammation. Higher levels of carboxyibuprofen being associated with greater cognitive decline may indicate the presence of inflammation. Sphingosine 1-phosphate, which delays cellular senescence, is reportedly associated with gait performance [8]. We now found it was also associated with cognitive decline. Taken together, metabolomic profiles of cognitive decline point to the dysregulation of the urea cycle, protein synthesis, and central nervous system (CNS). Among the 75 metabolites associated with cognitive decline, 15 were also associated with baseline cognition and showed consistent directions of upregulation and downregulation.
Nearly half of the metabolomic profiles of gait decline are glycerophosphocholines and glycerophosphoethanolamines, including lysoPCs, PCs, and PEs. Specific lysoPCs, such lysoPC (16:0), (16:1), (18:0), (18:1), and (18:2), are consistent with previous findings [5, 14, 27]. These metabolites are important precursors of cardiolipin which is the main structure of the mitochondrial inner member and have been associated with skeletal muscle mitochondrial function [28]. We also confirmed other metabolites, such as N2,N2-dimethylguanosine, N-formylmethionine, and hexose [5, 29]. N2,N2-dimethylguanosine is a methylated purine metabolite derived from tRNA degradation and has been involved in renal dysfunction [30]. N-formylmethionine plays an effective role in initiating protein synthesis. The process of methionine entering the ribosome occurs in the mitochondria of eukaryotic cells, Escherichia coli, and other bacteria [31]. Recent data from critically ill patients have shown that circulating N-formylmethionine is related to metabolic shift and high mortality which may involve impaired mitochondrial oxidation, elevated branch chain amino acid metabolism, and triggering the pentose phosphate pathway [32]. Multiple sugars are classified under the label “Hexose” including glucose, galactose, and fructose, which may suggest that high levels of sugar intake or circulating glucose contribute to gait decline. The remaining profiles are mainly from amino acids, carbohydrates, and sphingomyelins. Some of these are shared with metabolomic profiles of cognitive decline, such as tryptophan, alpha-N-phenylacetylglutamine, PC (38:6), SM (d18:1/14:0), and maslinic acid. We found additional metabolites associated with gait decline which were not reported nor investigated before. Some of the metabolites may suggest the important roles of metabolic disorders and vascular problems in gait decline. For instance, metformin and warfarin are used to treat diabetes and prevent blood clots, respectively. The negative associations of these metabolites with gait decline may indicate the presence of diabetes and heart disease. Threitol, PE (P-36:0)/PE (O-36:1), and SM (d18:1/14:0), are reportedly involved in diabetes. Sulfamethoxazole is an antibacterial and antibiotic agent and showed a positive association with gait decline. The role of sulfamethoxazole in physical function or frailty is unclear in humans, while some animal data shows that antibiotics increases locomotion [33].
The metabolomic profiles of cognitive decline and gait decline share 24 metabolites from amino acids, carbohydrates, carboxylic acids, glycerophosphocholines, and phosphosphingolipids. These shared profiles are in line with recently published data on metabolomic changes of dual decline in cognition and mobility which suggests deficits in mitochondrial function, compromised immunity, and increased burden in cardiovascular system and kidney [13, 14]. Specific lysoPCs and maslinic acid may highlight the importance of mitochondria function and inflammation. Alpha-N-phenylacetylglutamine may suggest the important role of the urea cycle. N2,N2-dimethylguanosine and uracil may indicate the importance of RNA synthesis and degradation. Other metabolites, such as SM (d18:1/14:0), adrenic acid, and tryptophan, may suggest the key role of the CNS in both cognitive decline and gait decline. The LASSO metabolite scores of cognitive decline and gait decline show a moderate correlation, suggesting substantial overlapping metabolomic markers predicting both cognitive decline and gait decline. We further identified that the majority of shared metabolomic markers predicted dementia risk, and further adjustment for the APOE e4 genotype did not substantially alter these associations. These findings provided additional insights into the connection between dual decline and high risk of dementia.
This study has novel aspects, and we extended prior literature from several aspects. First, metabolomic markers covered more than 600 metabolites, some of which were measured on more than one platform. This provides a more comprehensive look than examining only a few metabolites, and finding the same metabolites in more than one platform adds robustness to the findings. Second, multiple repeated assessments of cognitive function and gait speed over a decade allow us to more accurately model functional decline than only one-time assessment, two, or a few repeated measures. Third, the study population is diverse and includes nearly 40% Black participants. Last but not the least, we extended prior research by identifying shared metabolomic markers with dementia risk. One potential limitation is the dementia diagnosis based on cognitive decline, hospital records, and medication use. The ascertainment may be less rigorous compared to using cognitive adjudication conferences conducted in other studies.
In conclusion, among older adults initially free of dementia, metabolomic profiles of cognitive decline and mobility decline show distinct and shared signatures. Metabolites key to the urea cycle, CNS especially neurotransmitters and myelin health, protein synthesis, and inflammation are associated with cognitive decline; and metabolites important for mitochondrial function, urea cycle, carbohydrate metabolism, diabetes, and metabolic disorders are associated with gait decline. Shared metabolomic profiles which suggest mitochondrial function, inflammation, and urea cycle in addition to CNS may play key roles in both cognitive and mobility declines and predict dementia risk. These findings warrant validation in other aging populations. Future studies are warranted to understand these shared metabolomic markers with pathologies underlying dementia and subtypes and to investigate longitudinal relationships of metabolomic changes with functional decline.
References
Collyer TA, Murray AM, Woods RL, et al. Association of dual decline in cognition and gait speed with risk of dementia in older adults. JAMA Netw Open. 2022;5(5):e2214647. https://doi.org/10.1001/jamanetworkopen.2022.14647.
Tian Q, Resnick SM, Mielke MM, et al. Association of dual decline in memory and gait speed with risk for dementia among adults older than 60 years: a multicohort individual-level meta-analysis. JAMA Netw Open. 2020;3(2):e1921636. https://doi.org/10.1001/jamanetworkopen.2019.21636.
Montero-Odasso M, Speechley M, Muir-Hunter SW, et al. Motor and cognitive trajectories before dementia: results from gait and brain study. J Am Geriatr Soc. 2018;66(9):1676–83. https://doi.org/10.1111/jgs.15341.
Tian Q, Montero-Odasso M, Buchman AS, et al. Dual cognitive and mobility impairments and future dementia—setting a research agenda. Alzheimers Dement. 2023;19(4):1579–86. https://doi.org/10.1002/alz.12905.
Tian Q, Mitchell BA, Corkum AE, Moaddel R, Ferrucci L. Metabolites associated with memory and gait: a systematic review. Metabolites. 2022;12(4)https://doi.org/10.3390/metabo12040356
Huo Z, Yu L, Yang J, Zhu Y, Bennett DA, Zhao J. Brain and blood metabolome for Alzheimer’s dementia: findings from a targeted metabolomics analysis. Neurobiol Aging. 2020;86:123–33. https://doi.org/10.1016/j.neurobiolaging.2019.10.014.
Murphy RA, Moore S, Playdon M, et al. Metabolites associated with risk of developing mobility disability in the health, aging and body composition study. J Gerontol A Biol Sci Med Sci. 2019;74(1):73–80. https://doi.org/10.1093/gerona/glx233.
Wennberg AMV, Schafer MJ, LeBrasseur NK, et al. Plasma sphingolipids are associated with gait parameters in the Mayo clinic study of aging. J Gerontol A Biol Sci Med Sci. 2018;73(7):960–5. https://doi.org/10.1093/gerona/glx139.
Varma VR, Oommen AM, Varma S, et al. Brain and blood metabolite signatures of pathology and progression in Alzheimer disease: a targeted metabolomics study. PLoS Med. 2018;15(1): e1002482. https://doi.org/10.1371/journal.pmed.1002482.
Frison E, Boirie Y, Peuchant E, Tabue-Teguo M, Barberger-Gateau P, Feart C. Plasma fatty acid biomarkers are associated with gait speed in community-dwelling older adults: the three-city-Bordeaux study. Clin Nutr. 2017;36(2):416–22. https://doi.org/10.1016/j.clnu.2015.12.008.
Bressler J, Yu B, Mosley TH, et al. Metabolomics and cognition in African American adults in midlife: the atherosclerosis risk in communities study. Transl Psychiatry. 2017;7(7):e1173. https://doi.org/10.1038/tp.2017.118.
Simpson BN, Kim M, Chuang YF, et al. Blood metabolite markers of cognitive performance and brain function in aging. J Cereb Blood Flow Metab. 2016;36(7):1212–23. https://doi.org/10.1177/0271678X15611678.
Sultana M, Camicioli R, Dixon RA, et al. A metabolomics analysis of a novel phenotype of older adults at higher risk of dementia. J Alzheimers Dis. 2023;https://doi.org/10.3233/JAD-230683
Tian Q, Shardell MD, Kuo PL, et al. Plasma metabolomic signatures of dual decline in memory and gait in older adults. Geroscience. 2023;45(4):2659–67. https://doi.org/10.1007/s11357-023-00792-8.
Murthy VL, Nayor M, Carnethon M, et al. Circulating metabolite profile in young adulthood identifies long-term diabetes susceptibility: the coronary artery risk development in young adults (CARDIA) study. Diabetologia. 2022;65(4):657–74. https://doi.org/10.1007/s00125-021-05641-x.
Shah RV, Steffen LM, Nayor M, et al. Dietary metabolic signatures and cardiometabolic risk. Eur Heart J. 2023;44(7):557–69. https://doi.org/10.1093/eurheartj/ehac446.
Hong CH, Falvey C, Harris TB, et al. Anemia and risk of dementia in older adults: findings from the Health ABC study. Neurology. 2013;81(6):528–33. https://doi.org/10.1212/WNL.0b013e31829e701d.
Dobolyi A, Juhasz G, Kovacs Z, Kardos J. Uridine function in the central nervous system. Curr Top Med Chem. 2011;11(8):1058–67. https://doi.org/10.2174/156802611795347618.
Skinner ER, Watt C, Besson JA, Best PV. Differences in the fatty acid composition of the grey and white matter of different regions of the brains of patients with Alzheimer’s disease and control subjects. Brain. 1993;116(Pt 3):717–25. https://doi.org/10.1093/brain/116.3.717.
He Y, Wang Y, Yang K, et al. Maslinic acid: a new compound for the treatment of multiple organ diseases. Molecules 2022;27(24)https://doi.org/10.3390/molecules27248732
García A, Brenes M, Dobarganes MC, Romero C, Ruiz-Méndez MV. Enrichment of pomace olive oil in triterpenic acids during storage of “Alpeorujo” olive paste. Eur J Lipid Sci Technol. 2008;110(12):1136–41. https://doi.org/10.1002/ejlt.200800070.
Stiti N, Triki S, Hartmann MA. Formation of triterpenoids throughout Olea europaea fruit ontogeny. Lipids. 2007;42(1):55–67. https://doi.org/10.1007/s11745-006-3002-8.
Ho HY, Lin, W. C., Kitanaka, S., Chang, C. T., & Wu, J. B. (2008). Analysis of bioactive triterpenes in Eriobotrya japonica Lindl by high-performance liquid chromatography. J Food Drug Anal. 2008;16(6):41–45. https://doi.org/10.38212/2224-6614.2314
Guo S, Duan JA, Tang Y, et al. Simultaneous qualitative and quantitative analysis of triterpenic acids, saponins and flavonoids in the leaves of two Ziziphus species by HPLC-PDA-MS/ELSD. J Pharm Biomed Anal. 2011;56(2):264–70. https://doi.org/10.1016/j.jpba.2011.05.025.
Guo S, Duan JA, Tang YP, et al. Characterization of triterpenic acids in fruits of ziziphus species by HPLC-ELSD-MS. J Agric Food Chem. 2010;58(10):6285–9. https://doi.org/10.1021/jf101022p.
Tarvainen M, Suomela J-P, Kallio H, Yang B. Triterpene acids in Plantago major: identification, quantification and comparison of different extraction methods. Chromatographia. 2009;71(3–4):279–84. https://doi.org/10.1365/s10337-009-1439-7.
Gonzalez-Freire M, Moaddel R, Sun K, et al. Targeted metabolomics shows low plasma lysophosphatidylcholine 18:2 predicts greater decline of gait speed in older adults: the Baltimore longitudinal study of aging. J Gerontol A Biol Sci Med Sci. 2019;74(1):62–7. https://doi.org/10.1093/gerona/gly100.
Tian Q, Mitchell BA, Zampino M, Ferrucci L. Longitudinal associations between blood lysophosphatidylcholines and skeletal muscle mitochondrial function. Geroscience. 2022;44(4):2213–21. https://doi.org/10.1007/s11357-022-00548-w.
Nierenberg JL, He J, Li C, et al. Serum metabolites associate with physical performance among middle-aged adults: evidence from the Bogalusa heart study. Aging (Albany NY). 2020;12(12):11914–41. https://doi.org/10.18632/aging.103362.
Grams ME, Tin A, Rebholz CM, et al. Metabolomic alterations associated with cause of CKD. Clin J Am Soc Nephrol. 2017;12(11):1787–94. https://doi.org/10.2215/CJN.02560317.
Lee CS, Kim D, Hwang CS. Where does N-formylmethionine come from? What for? Where is it going? What is the origin of N-formylmethionine in eukaryotic cells? Mol Cells. 2022;45(3):109–11. https://doi.org/10.14348/molcells.2021.5040.
Sigurdsson MI, Kobayashi H, Amrein K, et al. Circulating N-formylmethionine and metabolic shift in critical illness: a multicohort metabolomics study. Crit Care. 2022;26(1):321. https://doi.org/10.1186/s13054-022-04174-y.
Schretter CE, Vielmetter J, Bartos I, et al. A gut microbial factor modulates locomotor behaviour in Drosophila. Nature. 2018;563(7731):402–6. https://doi.org/10.1038/s41586-018-0634-9.
Funding
Open access funding provided by the National Institutes of Health. This work was supported by the National Institute on Aging R01-AG-059729. The Health ABC cohort was supported by National Institute on Aging Contracts N01-AG-6–2101, N01-AG-6–2103, and N01-AG-6–2106; National Institute on Aging Grant R01-AG028050; and National Institute of Nursing Research Grant R01-NR012459. MMM is supported by the National Institute on Aging K01-AG-075143. ABN is supported by the Pittsburgh Pepper Center P30 AG024827. V. Murthy owns stock in General Electric, Cardinal Health, Pfizer, Amgen, Merck, and Johnson & Johnson and stock options in Ionetix. He is a paid consultant for INVIA Medical Imaging Solutions & Siemens Healthineers. V. Murthy has received research support through his institution from Siemens Healthineers. V. Murthy is supported by the Melvyn Rubenfire Professorship in Preventive Cardiology. V. Murthy is also supported by R01AG059729, R01HL136685, and U01DK123013 from the National Institutes of Health and AHA Strategically Focused Research Network 20SFRN35120123.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Ravi Shah, Venkatesh L. Murthy, and Anne B. Newman are co-senior authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Tian, Q., Yao, S., Marron, M.M. et al. Shared plasma metabolomic profiles of cognitive and mobility decline predict future dementia. GeroScience (2024). https://doi.org/10.1007/s11357-024-01228-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11357-024-01228-7