Abstract
Orthostatic hypotension (OH) is highly prevalent in older adults and associated with dizziness, falls, lower physical and cognitive function, cardiovascular disease, and mortality. OH is currently diagnosed in a clinical setting with single-time point cuff measurements. Continuous blood pressure (BP) devices can measure OH dynamics but cannot be used for daily life monitoring. Near-infrared spectroscopy (NIRS) has potential diagnostic value in measuring cerebral oxygenation continuously over a longer time period, but this needs further validation. This study aimed to compare NIRS-measured (cerebral) oxygenation with continuous BP and transcranial Doppler-measured cerebral blood velocity (CBv) during postural changes. This cross-sectional study included 41 participants between 20 and 88 years old. BP, CBv, and cerebral (long channels) and superficial (short channels) oxygenated hemoglobin (O2Hb) were measured continuously during various postural changes. Pearson correlations between BP, CBv, and O2Hb were calculated over curves and specific characteristics (maximum drop amplitude and recovery). BP and O2Hb only showed good curve-based correlations (0.58–0.75) in the initial 30 s after standing up. Early (30–40 s) and 1-min BP recovery associated significantly with O2Hb, but no consistent associations were found for maximum drop amplitude and late (60–175 s) recovery values. Associations between CBv and O2Hb were poor, but stronger for long-channel than short-channel measurements. BP associated well with NIRS-measured O2Hb in the first 30 s after postural change. Stronger associations for CBv with long-channel O2Hb suggest that long-channel NIRS specifically reflects cerebral blood flow during postural transitions, necessary to better understand the consequences of OH such as intolerance symptoms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Orthostatic hypotension (OH) is classically defined as a prolonged blood pressure (BP) drop upon standing of at least 20 mmHg systolic and/or 10 mmHg diastolic that occurs within the first 3 min after standing up [1]. OH is a prevalent condition in older adults, varying from 6% in the general population [2] up to 22% in people aged 60 years or older [3]. OH can have detrimental clinical outcomes, such as falls [4] and worse physical performance [5]. People who have OH also have higher risks of cognitive impairment [6], cardiovascular disease [7], and mortality [8]. Advancing current diagnosis and monitoring requires looking beyond the classic definition of OH, towards capturing the dynamics of posture-related changes in BP and their effects on cerebral perfusion pressure, cerebral blood flow (CBF), and cerebral oxygenation under daily life conditions [9,10,11,12]. Current clinical tools to measure OH are mainly intermittent and in some specialized centers non-invasive continuous BP measurements, but these are not suited for diagnosing or monitoring OH in daily life conditions. Another limitation is that measurements restricted to BP do not capture the effects of postural change on cerebral perfusion, while these are probably responsible for most OH-related symptoms [13]. Non-invasive, ambulant measurements of oxygenated (O2Hb), deoxygenated hemoglobin (HHb) and total hemoglobin (tHb) in cerebral frontal lobe tissue using near-infrared spectroscopy (NIRS) may be an alternative, but need further validation [14].
Previous work has shown how O2Hb and HHb are affected by postural changes from supine, sitting, or squatting to standing. Upon standing, the relative O2Hb concentration drops, followed by recovery towards a new equilibrium, which is somewhat lower than the baseline sitting or supine O2Hb. These effects were observed in healthy young and older adults. Simultaneously, HHb increases after standing up and recovers to a value above baseline [15,16,17,18]. Within the first 30 s of standing, Pearson correlation coefficients between O2Hb and BP measured by volume-clamp photoplethysmography ranged from 0.66 to 0.94 depending on the type of postural change [17]. Associations between O2Hb and BP in the remaining of the 3 min after standing have not been described.
NIRS can encompass different inter-optode distances of <1 cm (short channels) and 3–4 cm (long channels). Long-channel optodes are placed at such a distance that light is assumed to travel deep enough to penetrate through cerebral tissue. Short channels, however, are assumed to be restricted to measuring extracranial tissue and are therefore often used in functional NIRS (fNIRS) research to distinguish intra- from extracranial oxygenation responses [19, 20]. When studying OH, short-channel NIRS could in theory be used to reflect tissue perfusion changes due to changes in BP, unaffected by autoregulation, whereas long-channel NIRS would also reflect CBF changes which are modified by autoregulation [21]. To explore this, cerebral hemodynamic responses to standing can be tracked specifically by transcranial Doppler (TCD), which uses ultrasound to measure cerebral blood velocity (CBv) changes, most often in the middle cerebral artery (MCAv). These measurements reflect changes in CBF, provided that the artery diameter remains stable [22]. Until now, no studies have compared the oxygenation responses measured by long and short NIRS channels to postural BP changes.
The present study aimed to compare (cerebral) oxygenation obtained by NIRS with BP and MCAv during transitions from supine, sitting, or squatting to standing, stratified by age. Here, we recruited young and older adults not with the aim to investigate age-related changes in BP dynamics, but to elicit a wider range in postural BP responses. It was hypothesized that (a) BP would associate with O2Hb; (b) MCAv would associate with O2Hb, with the associations in (a) and (b) expected to be strongest in the early phase after standing, when BP changes are most pronounced and the short time window in which they occur limits compensation by cerebral autoregulation; and (c) associations between BP and short-channel O2Hb would be higher than for long-channel O2Hb, because cerebral autoregulation reduces the influence of BP on cerebral oxygenation but not on extracranial oxygenation, and that MCAv would show a stronger association with long-channel O2Hb than with short-channel O2Hb, reflecting changes in CBF.
Methods
Study design and participants
In a cross-sectional study at the geriatric department of the Radboudumc in Nijmegen, the Netherlands, between August 2021 and February 2022, 41 participants were included. To ensure a diversity of postural BP responses, both younger (18-35 years) and older adults (≥65 years) were included, with three times as many older adults, of whom at least 30% were recruited from the geriatric outpatient clinic. Participants were also recruited through flyers and advertisements at Radboud University. Exclusion criteria were being physically unable to perform orthostatic maneuvers or unable to understand written and oral instructions. Ethical approval was obtained from the medical ethics committee (CMO Arnhem-Nijmegen). All participants signed written informed consent. The study was performed in accordance with the declaration of Helsinki.
Data collection
Information about age, height, weight, comorbidity, medication use (type and number of medications), alcohol use (units per week), smoking habits (yes/no), history of falls in the last year, and OH symptoms was obtained from all participants. Intake of caffeine, alcohol, medication, and food before the measurements was noted. Older participants (≥65 years) filled in The Older Persons and Informal Caregiver Survey-Short Form (TOPICS-SF) questionnaire on activities of daily living (ADL) and comorbidities [23], and completed the Montreal Cognitive Assessment (MoCA) as a cognitive screening tool [24]. All participants performed a maximum grip strength, grip work (sustained grip strength), and 5-times chair-stand test, as markers of global physical fitness.
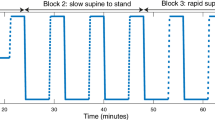
All participants performed three supine-stand maneuvers and three sit-stand maneuvers. Participants were instructed to lie, sit, and stand still and not to talk during measurements. Instructions were given to perform the transitions as fast as possible. Supine and sitting resting periods lasted 5 min. After each postural transition, participants remained standing for 3 min. Three slower supine-stand maneuvers, where the transition was performed in approximately 10 s while the researcher counted from one to ten, and three squat-stand maneuvers, consisting of 1 min of squatting before standing, were performed additionally by participants between 18 and 35 years.
BP was measured continuously using volume-clamp photoplethysmography on the digital artery of the left middle finger (Finapres NOVA, Finapres Medical Systems, Enschede, the Netherlands) [25]. The hand was placed in a sling to prevent hydrostatic pressure artefacts.
(Cerebral) oxygenation was measured using two NIRS sensors bilaterally attached to the forehead approximately 2 cm above the eyebrows (PortaLite MkII, Artinis Medical Systems, Elst, the Netherlands), covered with a black bandana to prevent ambient light interference. Sensors consisted of three light-emitting diodes and two detectors, placed at inter-optode distances of 2.9, 3.5, and 4.1 cm for the three long channels and 0.70, 0.80, and 0.74 cm for the three short channels. The control unit was placed in a belt around the waist.
MCAv was measured using TCD through the temporal window (DWL Doppler Box, Compumedics Germany GmbH, Singen, Germany). TCD probes were fixed with a headband worn over the bandana. Heart rate was recorded using a three-lead electrocardiogram (ECG), and end-tidal carbon dioxide (etCO2) levels were measured using capnography, as an estimation for arterial partial pressure of CO2.
Data acquisition and processing
(Cerebral) oxygenation signals were acquired in Oxysoft (version 3.4, Artinis Medical Systems, Elst, the Netherlands) at a sampling frequency of 100 Hz, while BP, MCAv, ECG, and etCO2 were obtained in Acqknowledge at 200 Hz (version 5.0, BioPac Systems Inc., Goleta, USA). Synchronization between both acquisition systems was achieved by analogue pulses (PortaSync MkII, Artinis Medical Systems, Elst, the Netherlands). Data were processed in MATLAB (2022a, MathWorks Inc., Natick, USA), using custom-written semi-automatic scripts [26]. Movement artefacts during supine-stand and sit-stand transitions were corrected in O2Hb and HHb signals during baseline and after 20 s of standing for each trial, using the movement artefact removal algorithm described by Scholkmann et al. [27, 28]. Between 2 s before standing up and 20 s after standing up (the period of the initial drop in O2Hb), movement artefacts could not be distinguished from postural oxygenation changes. Therefore, this part of the signal was not included in the movement artefact removal algorithm. Settings used were a threshold for artefact detection of three standard deviations, a moving window length for standard deviation calculation of one heartbeat (defined as sixty divided by the average heart rate during baseline or standing), and a 1-s local regression smoothing window for artefact correction. After this, the quality of all acquired signals was assessed visually, and signals not meeting the criteria were discarded. BP and MCAv signals were excluded when peaks and troughs were not distinguishable. ECG signals were excluded when QRS complexes were not visible. NIRS channels were excluded according to criteria described previously [29, 30]. These were an undistinguishable heartbeat during 10 s around the moment of standing up, flatlining, ambient light warnings, and an average tissue saturation index (TSI) fit factor (representing the agreement between different optodes) below 98%. Additionally, channels were excluded when movement artefacts were present during standing up, outside the correction window, or when the heartbeat amplitude was irregular (see Supplementary Table s1 and Fig. s2 for a full description of artefact types). Heart rate, systolic BP (SBP), diastolic BP (DBP), systolic MCAv (S-MCAv), diastolic MCAv (D-MCAv), and etCO2 were obtained over time by peak and trough detection. Mean MCAv, further referred to as MCAv, was calculated by two times D-MCAv and one time S-MCAv divided by three. tHb was calculated as the sum of O2Hb and HHb. All signals were resampled at 10 Hz and filtered using a 5-s moving average filter [31]. When bilateral MCAv and (cerebral) oxygenation signals were available and of sufficient quality, these were averaged. Otherwise, only the available signal was used for further analysis. For NIRS measurements, all available long channels were averaged, just like all available short channels.
Signal phases and characteristics
Curves (see Fig. 1) were divided in an initial response phase (the first 30 s after standing up) and late response phase (from 30 s after standing up until the end of the measurement at 3 min of standing) for all signals (curve-based analysis).
Example of A blood pressure response during standing and B oxygenation response during standing. Different phases (indicated by 1, 2) and characteristics (indicated by a, b, c, d, e) are shown in blue. 1, early recovery phase (0–30 s after standing up); 2, late recovery phase (30–175 s after standing up); and a, baseline, calculated as the average value between 60 and 30 s before standing up; b, initial drop within 30 s, calculated by minimum value minus baseline; c, early recovery, calculated as average value between 30 and 40 s after standing up; d, 1-min recovery, calculated as average value between 50 and 60 s after standing up; e, late recovery, calculated as average value between 60 and 175 s after standing up
Specific signal characteristics of the orthostatic courses of BP (systolic and diastolic), (cerebral) oxygenation (O2Hb and HHb in long and short channels), and MCAv were retrieved (visualized as b-e in Fig. 1) [15, 32]. These characteristics were the maximum drop amplitude, defined as the lowest value within 30 s after standing up, relative to the baseline value, and recovery at specified time points: the mean of 30-40 s (early recovery), the mean of 50–60 s (1-min recovery), and the mean of 60–175 s after standing up (late recovery), relative to the baseline value (characteristic-based analysis).
Statistical analysis
Statistical analyses were performed in MATLAB (R2018a), IBM SPSS Statistics 27, and RStudio (2022.02.1). All continuous variables are presented as mean (standard deviation) for normally distributed data, or median (interquartile range) for otherwise distributed data. Categorical variables are presented as number (percentage). For all analyses, two-sided testing with an alpha level of 0.05 was used.
Curve-based Pearson correlation coefficients between BP and O2Hb were calculated for each postural change of each participant, during the initial and late response phases. Correlation coefficients were averaged over three repeats per type of postural change per participant, averaged over all participants, and classified as excellent (0.75–1), good (0.6–0.75), fair (0.4–0.6), or poor (0–0.4) [33, 34]. Signal characteristics (b–e in Fig. 1) were averaged over three repeats per participant if present and Pearson correlation coefficients between BP and O2Hb characteristics were calculated per type of postural change. The association between BP (both systolic and diastolic) and O2Hb (both long and short channels) considering both the supine-stand and sit-stand postural change was determined for the maximum drop value, the early recovery value, the 1-min recovery value, and the late recovery value (characteristics b–e in Fig. 1) in a multilevel model with O2Hb characteristics b-e as outcome variables. Fixed effects for the respective BP characteristics b-e and a random intercept for participants resulted in the best model fit, as evaluated using a likelihood ratio test.
Curve-based and characteristic-based associations between MCAv and O2Hb were determined similarly to the associations between O2Hb and BP. It was determined whether curve-based associations between BP or MCAv and long-channel O2Hb and between BP or MCAv and short-channel O2Hb were significantly different using paired t-tests. Differences between younger versus older adults in curve-based and characteristic-based associations were tested for significance using Mann-Whitney U tests.
Results
Baseline characteristics
Forty-one participants with a diversity of BP responses upon standing up were included of whom eleven were between 18 and 35 years old, and thirty were 65 years or older. Their baseline characteristics are presented in Table 1. Nine older participants had BP responses that fulfilled classic OH criteria during the supine-stand transitions. For one participant, fast supine-stand transitions were excluded due to technical issues. Supplementary Fig. s1 and s4 and Supplementary Table s2 further elaborate quality assessment, removal of artefacts, and number of transitions available for analyses. Of the 303 transitions, BP signals were present and of sufficient quality in almost all (301), TCD signals in two-third (213), long-channel NIRS in most (292), and short-channel NIRS in almost 80% (239).
Average responses
Figure 2 shows the average BP, MCAv, and (cerebral) oxygenation responses for all participants, during all supine-stand and all sit-stand transitions. During a supine-stand postural change, the average initial SBP/DBP drop amplitude was −23.5/−10.4 (SD 14.1/7.2) mmHg, after which BP recovered to baseline values within 30 s from standing. S-MCAv and D-MCAv, which reflect systolic and diastolic CBF, showed different responses upon standing: whereas D-MCAv showed a drop (−12.6 (SD 8.3) cm/s), S-MCAv did not. This means that while, initially, systolic blood flow was maintained, diastolic flow was reduced, causing a reduction in total CBF during standing. In the early and late recovery phases, S-MCAv decreased and stabilized below baseline. This indicates that CBF upon standing does not fully return to supine values. O2Hb showed a clear drop upon standing, followed by recovery with an overshoot and stabilization to a level remaining below baseline (p < 0.001). The amplitude of the O2Hb drop was larger for short channels, on average −4.8 (SD 3.6) μmol/L, compared to −2.6 (SD 1.8) μmol/L for long channels (p < 0.001). When standing, long-channel HHb increased and stabilized above baseline (p = 0.001), while short-channel HHb showed a small decrease on average (p = 0.010). As tHb was calculated as the sum of O2Hb and HHb, and O2Hb showed more profound changes than HHb, the course of tHb was highly similar to the course of O2Hb (data not shown). In summary, there were reductions in long-channel O2Hb with concomitant increases in HHb in the later phase of standing.
Responses of A + E blood pressure (BP), systolic in red (solid line), diastolic in blue (dashed line), and heart rate (HR) in green (dashed-dotted line); B + F cerebral blood velocity (MCAv), systolic in red (solid line), and diastolic in blue (dashed line); C + G cerebral oxygenation measured with long channels, oxygenated hemoglobin in red and deoxygenated hemoglobin in blue (dashed line); and D + H cerebral oxygenation measured with short channels, oxygenated hemoglobin in red and deoxygenated hemoglobin in blue (dashed line). All signals are shown from 1 min before standing up to 175 s after standing up, during a supine-stand transition (A–D) or sit-stand transition (E–H). These responses were averaged over all supine-stand or sit-stand transitions and all participants. Standing up is indicated by a vertical black dashed line. Shaded areas show standard deviations for all signals
For a sit-stand maneuver, the mean SBP/DBP drop amplitude upon standing was −20.8/−14.1 (SD 10.7/5.4) mmHg, after which BP recovered to baseline values within 20–30 s after standing up. Similar to the supine-stand test, S-MCAv did not show an initial drop upon standing, while D-MCAv did, on average −5.1 (SD 4.0) cm/s. O2Hb showed an initial drop in both long and short channels. Compared to the supine-stand test with an O2Hb drop of −2.6 (SD 1.8) μmol/L, this drop was smaller with −1.7 (SD 1.2) μmol/L in long channels (p = 0.008). Long-channel O2Hb did not return to the sitting baseline value (p < 0.001), in contrast to short-channel O2Hb. Long- and short-channel HHb both stabilized above baseline (p = 0.006 and p = 0.013 respectively). In summary, also after a sit-stand transition, TCD and NIRS signals indicate a small reduction in CBF upon standing after stabilization following the initial drop in BP and CBF.
The effects of aging on the responses to a postural change are shown in Supplementary Fig. s5 and s6. For both supine-stand and sit-stand transitions, younger adults show a larger drop in O2Hb immediately after standing compared to older adults, followed by a faster recovery. Younger adults also had a larger increase in heart rate upon standing.
For slow supine-stand and squat-stand in young adults, results are shown in Supplementary Fig. s3, showing that both short- and long-channel O2Hb followed BP changes and stabilized below baseline. The effects of postural changes on etCO2 are shown in Supplementary Fig. s4. During standing up from sitting or supine position, etCO2 levels decreased by 0.19–0.59%, both in the early and late recovery phase. This corresponds to a decrease of 1.4–4.2 mmHg [35]. During squatting, etCO2 levels increased, which continued in the early recovery phase, followed by a decrease in the late recovery phase.
Associations between oxygenation and blood pressure
As shown in Table 2, curve-based Pearson correlation coefficients between BP and O2Hb were good (0.58–0.71) during the initial response but were poor (0.28–0.37) during the late response. All BP characteristics (maximum drop, early recovery, 1-min recovery, late recovery) showed poor associations with respective O2Hb characteristics (Table 3). These associations were all positive, indicating that a drop in BP led to a drop in O2Hb, for the supine-stand transition, and mostly positive for the sit-stand transition. Early and 1-min recovery BP values (both SBP and DBP) associated significantly with O2Hb values (Table 4). At maximum drop and late recovery, less consistent associations between BP and O2Hb characteristics were observed. They were only significant for maximum drop amplitude of DBP with long-channel O2Hb and late recovery of SBP with long-channel O2Hb. Sub-analyses (Supplementary Table s5) revealed higher Pearson correlation coefficients between BP and O2Hb for younger compared to older participants, which was significant for most associations.
Associations between oxygenation and cerebral blood velocity
Table 2 shows that curve-based Pearson correlation coefficients between MCAv and O2Hb were lower than between BP and O2Hb. They were positive but poor during both the initial and late response. All MCAv characteristics showed positive poor associations with respective long-channel O2Hb characteristics, but much less so for short-channel O2Hb where only the maximum drop amplitude showed a positive association (Table 3). For sit-stand transitions, less consistent Pearson correlation coefficients between MCAv and O2Hb characteristics were found.
Long- versus short-channel oxygenation
Curve- and characteristic-based associations (Tables 2 and 3) between BP and long-channel O2Hb and between BP and short-channel O2Hb were comparable. However, MCAv consistently showed stronger associations with long-channel O2Hb than with short-channel O2Hb, especially after 30 s of standing. Random effects (Table 4) showed higher between-subject variance in O2Hb characteristics for the short compared to the long channels.
Discussion
Main results
This cross-sectional study in 41 participants between 20 and 88 years old addressed the validity of measuring changes in (cerebral) oxygenation with NIRS to reflect the changes in BP and MCAv during postural changes. Good curve-based associations (0.6–0.8) between dynamic BP and O2Hb were found, albeit only for the initial response within the first 30 s after postural change. Early (30–40 s) and 1-min BP recovery were significantly associated with O2Hb, but there were no consistent associations for maximum drop amplitude and late recovery values. MCAv and O2Hb associated poorly, but MCAv associated consistently better with long-channel O2Hb than with short-channel O2Hb. Taken together, this indicates that in the early phase after standing (30 s), changes in O2Hb reflect changes in BP. Between 30 s and 1 min, O2Hb changes still indicate changes in BP, but based on the O2Hb signal alone, the magnitude of BP changes cannot be inferred. Long-channel NIRS signals correlate better with CBF (measured as changes in MCAv using TCD) than short-channel signals, indicating that short-channel NIRS measures extracranial (skin) tissue, but long-channel NIRS measures also cerebral tissue.
(Cerebral) oxygenation compared to blood pressure responses to postural change
Long-channel O2Hb and HHb responses to postural change found in this study are consistent with previous studies: in young as well as older healthy adults, O2Hb and HHb did not return to baseline values, while BP did, even after 5 min of standing [15, 16, 36,37,38]. This is also reflected in the good curve-based Pearson correlation values, which showed high similarity between BP and O2Hb dynamics in the early recovery phase after standing up, confirming previous research [17], but this association disappeared in the late recovery phase. Supine-stand transitions showed slightly higher correlation coefficients between BP and O2Hb than sit-stand transitions. Standing up from a supine position leads to a more profound drop in O2Hb than standing up from a sitting position, probably because cerebral oxygenation is already lower in sitting compared to supine position [15, 16]. In this study, the larger O2Hb drop after a supine-stand transition was not accompanied by a larger BP drop, contrary to existing literature [39]. A lower cerebral oxygenation during sitting and standing could be explained by the differences in hydrostatic pressure gradient between the heart and brain in sitting or standing versus supine position, leading to a small but sustained reduction in cerebral perfusion pressure and thus cerebral oxygen delivery [21, 40, 41]. Because the cerebral metabolic demand and therefore oxygen consumption remain stable, this results in standing O2Hb values below and HHb values above baseline.
In the late recovery phase, cerebral autoregulation has had time to react to BP changes, resulting in a new equilibrium wherein there is no longer an influence of BP on CBF. O2Hb and BP were significantly associated for early and 1-min recovery values, but did not strongly associate characteristic-based, and thus between participants, suggesting that subject-specific characteristics may be an important source of variance. This was supported by the fact that a random, participant-dependent, intercept improved the multilevel model fit, indicating that an average BP drop is accompanied by a subject-specific oxygenation drop. Age may play a role, as younger participants had a better curve-based association between O2Hb and BP in the initial phase than older participants.
(Cerebral) oxygenation measured with long channels and short channels
O2Hb and MCAv both dropped upon standing. Compared to associations between BP and O2Hb, MCAv and O2Hb associated only poorly. This indicates that not only cerebral effects have been reflected in O2Hb, but also extracranial blood flow. However, both curve-based and characteristic-based associations were consistently higher between MCAv and long-channel O2Hb than between MCAv and short-channel O2Hb, while BP associated similarly well with both long- and short-channel O2Hb. This corresponds to our hypothesis and underpins the potential value of discerning between long and short channels, in which long-channel signals are expected to better reflect the effects of cerebral autoregulation, and thus associate better with MCAv, than short-channel signals which are thought to reflect superficial, extracerebral blood flow changes. In further support of this, in the later phase (>30 s) after postural change, when compensation by cerebral autoregulation has taken place, the difference between long and short channels became more pronounced.
Although the combination of long and short channels is promising, short channels showed a few disadvantages in our study. Compared to long-channel oxygenation, short-channel O2Hb and HHb responses were more heterogeneous, both during a supine-stand and a sit-stand transition, and less robust than long-channel measurements. This was expressed in the number of repetitions that had to be discarded due to bad signal quality, like movement artefacts, and irregular heartbeat amplitudes that were not present in long channels.
Importance of cerebral oxygenation measurements
Our results indicate that there is no linear relation between O2Hb and BP, and that linear methods as used in this study are not sufficient for an absolute BP estimation using oxygenation signals, as characteristic-based associations between O2Hb and BP were all poor. It is however questionable whether absolute BP estimation should be targeted, because orthostatic intolerance is not only determined by absolute BP values. Orthostatic intolerance symptoms (light-headedness, dizziness, blurred vision) as well as negative health outcomes related to OH such as falls and fractures all originate from cerebral hypoperfusion [4, 21, 42, 43]. During postural changes, the reduction in CBF is not solely dependent on BP, but also on the efficiency of cerebral autoregulation, on the magnitude of changes in etCO2, and on the hydrostatic gradient between the heart and brain [21, 44,45,46]. Measuring cerebral oxygenation, which reflects cerebral perfusion, may therefore have a more direct association with symptoms and outcomes than measuring BP. Previous research by the TILDA group, linking low cerebral oxygenation to depression, suggests this mediating role [47, 48]. However, it is good to realize that cerebral hypoperfusion is not always symptomatic. Orthostatic cerebral oxygenation in the same cohort was not associated with orthostatic intolerance symptoms [49]. Orthostatic intolerance symptoms were defined as the presence of any symptoms of dizziness, light-headedness, or unsteadiness, in which symptom severity was not considered. In the present study, more than 50% of young participants reported symptoms, although it is unlikely that they suffer from orthostatic intolerance impairing daily life or mediating negative health outcomes. Moreover, recent research suggests that patients who have OH are often not recognizing accompanying symptoms themselves, making them at risk of falls [50]. Therefore, cerebral oxygenation in relation to orthostatic intolerance is a topic for further research, especially in the home setting where falls occur. Eventually, this may aid in reconsidering the OH definition, from BP-based to cerebral oxygenation-based.
Strengths and limitations
Strengths of this study were the multimodal measurements, use of different postural changes, each repeated three times, and a heterogeneous cohort with a broad age range, the latter two leading to a large variety of BP responses. This study had a few limitations. First, since the PortaLite MkII was still being improved during this study, some channels had to be excluded due to noise which was present in one sensor. Second, different repetitions were excluded due to artefacts, which were classified arbitrarily. However, to ensure consistency, classification rules were set, as described in the Supplementary Information, and classification was performed by two researchers independently. Third, the TCD headband was placed over the NIRS sensors, because of limited space on the forehead. This complicated MCAv recordings, and therefore led to TCD measurements being unavailable in one-third of all subjects. This however reflects reality, as TCD is less often measurable in older adults, and supports the use of NIRS for cerebral perfusion monitoring, as sensor attachment is less complex and time-consuming than for TCD. Fourth, we did not specifically target to include participants with OH, and therefore, only a limited number with OH were included, especially a small number of participants with orthostatic intolerance symptoms. Therefore, assessment of the relation between O2Hb after postural change and orthostatic intolerance was not possible. Finally, future work is needed to investigate the effects of dynamic cerebral autoregulation on the relationship between BP and CBF and cerebral oxygenation during postural changes. Such studies should expand beyond linear statistics and investigate the non-linear dynamics of these signals, both in the time and frequency domain [51].
Conclusion
In conclusion, our results indicate that O2Hb measured with NIRS can capture postural BP dynamics. The larger the change in O2Hb, for example in the initial response phase or a supine-stand instead of sit-stand transition, the better O2Hb is reflecting BP changes. Small changes, as present in the late response phase after postural change, were not directly transferred from BP to oxygenation. Long-channel O2Hb probably captured part of the cerebral response, contrary to short-channel O2Hb, and is therefore promising for measuring orthostatic intolerance, as these symptoms are not always directly related to OH. As such, future research should focus on measuring (cerebral) oxygenation in a daily life setting, to investigate NIRS as an ambulant monitoring tool for OH and orthostatic intolerance.
Change history
31 May 2023
A Correction to this paper has been published: https://doi.org/10.1007/s11357-023-00837-y
References
Freeman R, et al. Consensus statement on the definition of orthostatic hypotension, neurally mediated syncope and the postural tachycardia syndrome. Clin Auton Res. 2011;21(2):69–72. https://doi.org/10.1007/s10286-011-0119-5.
Joseph A, Wanono R, Flamant M, Vidal-Petiot E. Orthostatic hypotension: a review. Néphrologie & Thérapeutique. 2017;13:S55–67. https://doi.org/10.1016/j.nephro.2017.01.003.
Saedon NI, Pin Tan M, Frith J. The prevalence of orthostatic hypotension: a systematic review and meta-analysis. J Gerontol A Biol Sci Med Sci. 2020;75(1):117–22. https://doi.org/10.1093/gerona/gly188.
Mol A, et al. Orthostatic hypotension and falls in older adults: a systematic review and meta-analysis. J Am Med Dir Assoc. 2019;20(5):589–597.e5. https://doi.org/10.1016/j.jamda.2018.11.003.
Mol A, Reijnierse EM, Bui Hoang PTS, van Wezel RJA, Meskers CGM, Maier AB. Orthostatic hypotension and physical functioning in older adults: a systematic review and meta-analysis. Ageing Res Rev. 2018;48:122–44. https://doi.org/10.1016/j.arr.2018.10.007.
Iseli R, Nguyen VTV, Sharmin S, Reijnierse EM, Lim WK, Maier AB. Orthostatic hypotension and cognition in older adults: a systematic review and meta-analysis. Exp Gerontol. 2019;120:40–9. https://doi.org/10.1016/j.exger.2019.02.017.
Verwoert GC, et al. Orthostatic hypotension and risk of cardiovascular disease in elderly people: the Rotterdam study. J Am Geriatr Soc. 2008;56(10):1816–20. https://doi.org/10.1111/j.1532-5415.2008.01946.x.
Frith J, Bashir AS, Newton JL. The duration of the orthostatic blood pressure drop is predictive of death. QJM: An Int J of Med. 2015;109(4):231–5. https://doi.org/10.1093/qjmed/hcv126.
Claassen JAHR. Orthostatische hypotensie bij de oudere patient. Ned Tijdschr Geneeskd. 2018;162:D1943.
Finucane C, et al. Age-related normative changes in phasic orthostatic blood pressure in a large population study. Circulation. 2014;130(20):1780–9. https://doi.org/10.1161/CIRCULATIONAHA.114.009831.
Moloney D, et al. Clinical clustering of eight orthostatic haemodynamic patterns in The Irish Longitudinal Study on Ageing (TILDA). Age and Ageing. 2020;50(3):854–60. https://doi.org/10.1093/ageing/afaa174.
Harms MPM, et al. Systemic and cerebral circulatory adjustment within the first 60 s after active standing: an integrative physiological view. Autonomic Neurosci. 2021;231:102756. https://doi.org/10.1016/j.autneu.2020.102756.
van Wijnen VK, Harms MP, Wieling W. Orthostatic hypotension in the first minute after standing up. Hypertension. 2018;71(5):816–8. https://doi.org/10.1161/HYPERTENSIONAHA.118.10609.
Scholkmann F, et al. A review on continuous wave functional near-infrared spectroscopy and imaging instrumentation and methodology. Neuroimage. 2014;85(Part 1):6–27. https://doi.org/10.1016/j.neuroimage.2013.05.004.
Mol A, Claassen J, Maier A, Wezel R, Meskers C. Determinants of orthostatic cerebral oxygenation assessed using near-infrared spectroscopy. Autonomic Neurosci. 2022;238:102942. https://doi.org/10.1016/j.autneu.2022.102942.
Mol A, Woltering JHH, Colier W, Maier AB, Meskers CGM, van Wezel RJA. Sensitivity and reliability of cerebral oxygenation responses to postural changes measured with near-infrared spectroscopy. Eur J Appl Physiol. 2019;119(5):1117–25. https://doi.org/10.1007/s00421-019-04101-0.
Mol A, Maier AB, van Wezel RJA, Meskers CGM. Multimodal Monitoring of Cardiovascular Responses to Postural Changes. Front Physiol. 2020;11:168. https://doi.org/10.3389/fphys.2020.00168.
Mehagnoul-Schipper DJ, Vloet LCM, Colier WNJM, Hoefnagels WHL, Jansen RWMM. Cerebral oxygenation declines in healthy elderly subjects in response to assuming the upright position. Stroke. 2000;31(7):1615–20. https://doi.org/10.1161/01.STR.31.7.1615.
Kirilina E, Yu N, Jelzow A, Wabnitz H, Jacobs AM, Tachtsidis I. Identifying and quantifying main components of physiological noise in functional near infrared spectroscopy on the prefrontal cortex. Front Physiol. 2013;7:864. https://doi.org/10.3389/fnhum.2013.00864.
Brigadoi S, Cooper RJ. How short is short? Optimum source-detector distance for short-separation channels in functional near-infrared spectroscopy. Neurophotonics. 2015;2(2):025005. https://doi.org/10.1117/1.NPh.2.2.025005.
Claassen JA, Thijssen DH, Panerai RB, Faraci FM. Regulation of cerebral blood flow in humans: physiology and clinical implications of autoregulation. Physiol Rev. 2021;101(4):1487–559. https://doi.org/10.1152/physrev.00022.2020.
Skow RJ, et al. On the use and misuse of cerebral hemodynamics terminology using transcranial Doppler ultrasound: a call for standardization. Am J Physiol Heart Circ Physiol. 2022;323(2):H350–7. https://doi.org/10.1152/ajpheart.00107.2022.
Santoso AMM, et al. Development of a patient-reported outcome measure for geriatric care: the older persons and informal caregivers survey short form. Value in Health. 2018;21(10):1198–204. https://doi.org/10.1016/j.jval.2018.02.011.
Nasreddine ZS, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695–9. https://doi.org/10.1111/j.1532-5415.2005.53221.x.
Imholz BP, Wieling W, van Montfrans GA, Wesseling KH. Fifteen years experience with finger arterial pressure monitoring: assessment of the technology. Cardiovasc Res. 1998;38(3):605–16. https://doi.org/10.1016/s0008-6363(98)00067-4.
de Heus RAA, et al. Dynamic regulation of cerebral blood flow in patients with alzheimer disease. Hypertension. 2018;72(1):139–50. https://doi.org/10.1161/hypertensionaha.118.10900.
Scholkmann F, Spichtig S, Muehlemann T, Wolf M. How to detect and reduce movement artifacts in near-infrared imaging using moving standard deviation and spline interpolation. Physiol Meas. 2010;31(5):649–62. https://doi.org/10.1088/0967-3334/31/5/004.
Nieuwhof F, et al. Measuring prefrontal cortical activity during dual task walking in patients with Parkinson's disease: feasibility of using a new portable fNIRS device. Pilot Feasibility Stud. 2016;2:59. https://doi.org/10.1186/s40814-016-0099-2.
O’Connor JD, et al. Functional analysis of continuous, high-resolution measures in aging research: a demonstration using cerebral oxygenation data from the Irish longitudinal study on aging. Front Hum Neurosci. 2020;14:261. https://doi.org/10.3389/fnhum.2020.00261.
Sappia MS, Hakimi N, Colier WN, Horschig JM. Signal quality index: an algorithm for quantitative assessment of functional near infrared spectroscopy signal quality. Biomed Opt Express. 2020;11(11):6732–54. https://doi.org/10.1364/BOE.409317.
van der Velde N, van den Meiracker AH, Stricker BH, van der Cammen TJ. Measuring orthostatic hypotension with the Finometer device: is a blood pressure drop of one heartbeat clinically relevant? Blood Press Monit. 2007;12(3):167–71. https://doi.org/10.1097/MBP.0b013e3280b083bd.
Finucane C, et al. A practical guide to active stand testing and analysis using continuous beat-to-beat non-invasive blood pressure monitoring. Clin Auton Res. 2019;29(4):427–41. https://doi.org/10.1007/s10286-019-00606-y.
Akoglu H. User's guide to correlation coefficients. Turk J Emerg Med. 2018;18(3):91–3. https://doi.org/10.1016/j.tjem.2018.08.001.
Cicchetti DV. Guidelines, criteria, and rules of thumb for evaluating normed and standardized assessment instruments in psychology. Psycho assess. 1994;6(4):284.
Kreit JW. Volume capnography in the intensive care unit: physiological principles, measurements, and calculations. Ann Am Thorac Soc. 2019;16(3):291–300. https://doi.org/10.1513/AnnalsATS.201807-501CME.
Mehagnoul-Schipper DJ, Vloet LC, Colier WN, Hoefnagels WH, Jansen RW. Cerebral oxygenation declines in healthy elderly subjects in response to assuming the upright position. Stroke. 2000;31(7):1615–20. https://doi.org/10.1161/01.str.31.7.1615.
Harms MP, Colier WN, Wieling W, Lenders JW, Secher NH, Van Lieshout JJ. Orthostatic tolerance, cerebral oxygenation, and blood velocity in humans with sympathetic failure. Stroke. 2000;31(7):1608–14. https://doi.org/10.1161/01.str.31.7.1608.
Kim YS, Bogert LW, Immink RV, Harms MP, Colier WN, van Lieshout JJ. Effects of aging on the cerebrovascular orthostatic response. Neurobio of Aging. 2011;32(2):344–53. https://doi.org/10.1016/j.neurobiolaging.2009.02.019.
Shaw BH, et al. Optimal diagnostic thresholds for diagnosis of orthostatic hypotension with a 'sit-to-stand test'. J Hypertens. 2017;35(5):1019–25. https://doi.org/10.1097/HJH.0000000000001265.
Soeding P. Effects of positioning on cerebral oxygenation. Curr Anesthesiol Rep. 2013;3(3):184–96. https://doi.org/10.1007/s40140-013-0020-y.
Garrett ZK, Pearson J, Subudhi AW. Postural effects on cerebral blood flow and autoregulation. Physiol Rep. 2017;5:4. https://doi.org/10.14814/phy2.13150.
Wolters FJ, et al. Cerebral perfusion and the risk of dementia: a population-based study. Circulation. 2017;136(8):719–28. https://doi.org/10.1161/CIRCULATIONAHA.117.027448.
Fitzgibbon-Collins LK, Heckman GA, Bains I, Noguchi M, McIlroy WE, Hughson RL. Older adults' drop in cerebral oxygenation on standing correlates with postural instability and may improve with sitting prior to standing. J Gerontol A Biol Sci Med Sci. 2021;76(6):1124–33. https://doi.org/10.1093/gerona/glaa194.
Novak P. Orthostatic cerebral hypoperfusion syndrome. Front Cell Neurosci. 2016;8:22. https://doi.org/10.3389/fnagi.2016.00022.
Panerai RB. Assessment of cerebral pressure autoregulation in humans - a review of measurement methods. Physiol Meas. 1998;19(3):305. https://doi.org/10.1088/0967-3334/19/3/001.
Lipsitz LA, Mukai S, Hamner J, Gagnon M, Babikian V. Dynamic regulation of middle cerebral artery blood flow velocity in aging and hypertension. Stroke. 2000;31(8):1897–903. https://doi.org/10.1161/01.str.31.8.1897.
Briggs R, et al. The association between frontal lobe perfusion and depressive symptoms in later life. Br J Psychiatry. 2019;214(4):230–6. https://doi.org/10.1192/bjp.2018.288.
McDowell CP, et al. The association between generalized anxiety disorder and resting-state prefrontal cortex oxygenation is modified by self-reported physical activity: results from the irish longitudinal study on ageing. J Gerontol: Series A. 2021;76(8):1391–7. https://doi.org/10.1093/gerona/glab090.
Newman L, O'Connor JD, Nolan H, Reilly RB, Kenny RA. Age and sex related differences in orthostatic cerebral oxygenation: findings from 2764 older adults in the Irish Longitudinal Study on Ageing (TILDA). Exp Gerontol. 2022;167:111903. https://doi.org/10.1016/j.exger.2022.111903.
Freeman R, et al. Symptom recognition is impaired in patients with orthostatic hypotension. Hypertension. 2020;75:1325–32. https://doi.org/10.1161/HYPERTENSIONAHA.119.13619.
Mukli P, et al. Two-Tiered Response of Cardiorespiratory-Cerebrovascular Network to Orthostatic Challenge. Front Physiol. 2021;12:216. https://doi.org/10.3389/fphys.2021.622569.
Acknowledgements
We are grateful to all subjects who participated in this study for their time and effort. We would like to thank Maaike Laagland, from Artinis Medical Systems, for her help with methodological questions and Odette Helsloot, Ricardo Wennekers, and Stella Thissen for their assistance in the data collection and preprocessing.
Funding
This study was funded by the Operational Program European Regional Development Fund (OP ERDF) of the European Union under the “PROHEALTH” project (PROJ-01003).
Author information
Authors and Affiliations
Contributions
Marjolein Klop: methodology, data collection, data analysis, writing original draft; Rianne de Heus: methodology, data analysis, writing—review and editing; Andrea Maier: conceptualization, methodology, writing—review and editing; Anne van Alphen: data collection, data analysis, writing—review and editing; Marianne Floor-Westerdijk: funding, conceptualization, methodology, writing—review and editing; Mathijs Bronkhorst: conceptualization, methodology, writing—review and editing; René Melis: data analysis, writing—review and editing; Carel Meskers: funding, conceptualization, methodology, writing—review and editing; Jurgen Claassen: funding, conceptualization, methodology, writing—review and editing; Richard van Wezel: funding, conceptualization, methodology, writing—review and editing
Corresponding author
Ethics declarations
Ethical approval and consent to participate
All participants gave written informed consent and the study was performed in accordance with the Declaration of Helsinki. Approval to perform this study, which was judged to fall beyond the remits of the Dutch Medical Research Involving Human Subjects Act (WMO), was obtained from the medical ethics committee (CMO Arnhem-Nijmegen).
Conflict of interest
Marianne Floor-Westerdijk and Mathijs Bronkhorst are employees of Artinis Medical Systems and were involved in the development of the NIRS device used in this study. This research was part of a funded project in which Artinis Medical Systems (the manufacturer of the NIRS system) also participated. The authors declare no other competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1:
Fig. s1 Flowchart of available participants, maneuvers, repetitions and signals, that were included in the analyses (available data fulfilling quality criteria). Fig. s2 Examples of different artefacts that were seen in the near-infrared spectroscopy (NIRS) data. Table s1 Description of artefacts that were present in the data. Table s2 Number of excluded channels due to artefacts per type of maneuver and type of near-infrared spectroscopy (NIRS) channel (short or long), specified by type of artefact. Fig. s3 Responses of blood pressure, heart rate, cerebral blood velocity, cerebral oxygenation measured with long channelsand cerebral oxygenation measured with short channels, averaged over all supine-stand or squat-stand transitionsand all young participants. Table s3 Heatmaps of average correlations during a slow supine-stand transition. Table s4 Heatmaps of average correlations during a squat-stand transition. Fig. s4 Responses of end-tidal carbon dioxide (etCO2) during a fast supine-stand, sit-stand, slow supine-stand and squat-stand transition. Table s5 Heatmaps of average correlations during a fast supine-stand transition, showing sub-analyses for younger (18-35 years) and older (>65 years) adults. Fig. s5 Supine-stand responses in older (>65 years) and younger (18-35 years) participants Fig. s6 Sit-stand responses in older (>65 years) and younger (18-35 years) participants.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Klop, M., de Heus, R.A.A., Maier, A.B. et al. Capturing postural blood pressure dynamics with near-infrared spectroscopy-measured cerebral oxygenation. GeroScience 45, 2643–2657 (2023). https://doi.org/10.1007/s11357-023-00791-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-023-00791-9