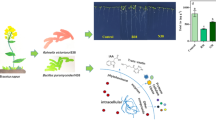
Abstract
In this study, the addition of γ-aminobutyric acid (GABA), Bacillus pumilus, or both, was found to enhance rice growth and yield while significantly decreasing arsenic (As) accumulation in Oryza sativa rice tissues. GABA emerged as a regulator of iron (Fe) homeostasis, acting as a signaling modulator that influenced phytosiderophore secretions in the plant. Meanwhile, B. pumilus directly increased Fe levels through siderophore production, promoting the development of Fe-rich rice plants. Subsequently, Fe competed with As uptake at the root surface, leading to decreased As levels and translocation to the grains. Furthermore, the addition of GABA and B. pumilus optimized rice indole-3 acetic acid (IAA) contents, thereby adjusting cell metabolite balance under As stress. This adjustment results in low malondialdehyde (MDA) contents in the leaves and roots during the early and late vegetative phases, effectively reducing oxidative stress. When added to As-contaminated soil, GABA and B. pumilus effectively maintained endogenous GABA levels and exhibited low ROS generation, similar to normal soil. Concurrently, GABA and B. pumilus significantly downregulated the activity of OsLsi1, OsLsi2, and OsABCC1 in roots, reducing As uptake through roots, shoots, and grains, respectively. These findings suggest that GABA and B. pumilus additions impede As translocation through grains, ultimately enhancing rice productivity under As stress.





Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Álvarez-Robles MJ, Bernal MP, Sánchez-Guerrero A et al (2020) Major As species, lipid peroxidation and protein carbonylation in rice plants exposed to increasing As(V) concentrations. Heliyon 6:e04703. https://doi.org/10.1016/j.heliyon.2020.e04703
Ashraf U, Mahmood S, Anjum SA et al (2022) Exogenous gamma-aminobutyric acid application induced modulations in the performance of aromatic rice under lead toxicity. Front Plant Sci 13:933694. https://doi.org/10.3389/fpls.2022.933694
Aung MS, Masuda H (2020) How does rice defend against excess iron?: physiological and molecular mechanisms. Front Plant Sci 11:1–8. https://doi.org/10.3389/fpls.2020.01102
Borgarrd OK (1983) The influence of iron oxides on phosphate adsorption by soil. J Soil Sci 34:337–341. https://doi.org/10.1111/J.1365-2389.1983.TB01039.X
Bouche N, Fromm H (2004) GABA in plants: just a metabolite? TRENS Plant Sci 9:110–115. https://doi.org/10.1016/j.tplants.2004.01.006
Briat JF, Curie C, Gaymard F (2007) Iron utilization and metabolism in plants. Curr Opin Plant Biol 10:276–282. https://doi.org/10.1016/j.pbi.2007.04.003
Chakraborty U, Chakraborty BN, Roychowdhury P (2013) Plant growth promoting activity of Bacillus pumilus in tea (Camellia sinensis) and its biocontrol potential against Poria hypobrunnea. Indian Phytopathol 66:387–396
Chen WQ, Shi YL, Wu SL, Zhu YG (2016) Anthropogenic arsenic cycles: a research framework and features. J Clean Prod 139:328–336. https://doi.org/10.1016/j.jclepro.2016.08.050
Crichton RR, Wilmet S, Legssyer R, Ward RJ (2002) Molecular and cellular mechanisms of iron homeostasis and toxicity in mammalian cells. J Inorg Biochem 91:9–18. https://doi.org/10.1016/s0162-0134(02)00461-0
Dobrzyński J, Jakubowska Z, Dybek B (2022) Potential of Bacillus pumilus to directly promote plant growth. Front Microbiol 13:1–6. https://doi.org/10.3389/fmicb.2022.1069053
Dolphen R, Thiravetyan P (2019) Reducing arsenic in rice grains by leonardite and arsenic-resistant endophytic bacteria. Chemosphere 223:448–454. https://doi.org/10.1016/j.chemosphere.2019.02.054
Du H, Liu H, Xiong L (2013) Endogenous auxin and jasmonic acid levels are differentially modulated by abiotic stresses in rice. Front Plant Sci 4:397. https://doi.org/10.3389/fpls.2013.00397
Fabiani A, Versari A, Parpinello GP et al (2002) High-performance liquid chromatographic analysis of free amino acids in fruit juices using derivatization with 9-fluorenylmethyl-chloroformate. J Chromatogr Sci 40:14–18. https://doi.org/10.1093/chromsci/40.1.14
FAO (2020) http://www.fao.org/news/story/en/item/1269721/icode/. Retrieved 15 Sept 2020
Feng Y, Bayaer E, Qi Y (2022) Advances in the biological functions of auxin transporters in rice. Agriculture 12:989. https://doi.org/10.3390/agriculture12070989
Finnegan PM, Chen W (2012) Arsenic toxicity: the effects on plant metabolism. Front Physiol 3:1–18. https://doi.org/10.3389/fphys.2012.00182
Hasanuzzaman M, Borhannuddin Bhuyan MHM, Zulfiqar F, Raza A et al (2020) Reactive oxygen species and antioxidant defence in plants under abiotic stress: revisiting the crucial role of a universal defence regulator. Antioxidans (bassel) 9:681. https://doi.org/10.3390/antiox9080681
Huang JH, Hu KN, Decker B (2011) Organic arsenic in the soil environment: speciation, occurrence, transformation, and adsorption behavior. Water Air Soil Pollut 219:401–415. https://doi.org/10.1007/s11270-010-0716-2
Kathiresan A, Tung P, Chinnappa CC, Reid DM (1997) Gamma-aminobutyric acid stimulates ethylene biosynthesis in sunflower. Plant Physiol 115:129–135. https://doi.org/10.1104/pp.115.1.129
Kim J, Rees DC (1992) Structural models for the metal centers in the nitrogenous molybdenum-iron protein. Science 257:1677–1682. https://doi.org/10.1126/science.1529354
Kinnersley AM, Turano FJ (2000) Gamma aminobutyric acid (GABA) and plant responses to stress. Crit Rev Plant Sci 19:479–509. https://doi.org/10.1080/07352680091139277
Krishnamurthy A, Rathinasabapathi B (2013) Auxin and its transport play a role in plant tolerance to arsenite-induced oxidative stress in Arabidopsis thaliana. Plant Cell Environ 36:1838–1849
Kumar S, Trivedi PK (2018) Glutathione S-transferases: role in combating abiotic stresses including arsenic detoxification in plants. Front Plant Sci 9:751. https://doi.org/10.3389/fpls.2018.00751
Kumar A, Singh RP, Singh PK et al (2014) Selenium ameliorates arsenic induced oxidative stress through modulation of antioxidant enzymes and thiols in rice (Oryza sativa L). Ecotoxicology 23:1153–1163. https://doi.org/10.1007/s10646-014-1257-z
Kumar N, Dubey AK, Jaiswal PK, Sahu N, Behera SK, Tripathi RD, Mallick S (2016) Selenite supplementation reduces arsenate uptake greater than phosphate but compromises the phosphate level and physiological performance in hydroponically grown Oryza sativa L. Environ Toxicol Chem 35:163–172. https://doi.org/10.1002/etc.3171
Kumar N, Dubey AK, Upadhyay AK et al (2017) GABA accretion reduces Lsi-1 and Lsi-2 gene expressions and modulates physiological responses in Oryza sativa to provide tolerance towards arsenic. Sci Rep 7:8786. https://doi.org/10.1038/s41598-017-09428-2
Li L, Dou N, Zhang H, Wu C (2021) The versatile GABA in plants. Plant Signal Behav 16:e1862565. https://doi.org/10.1080/15592324.2020.186256
Li W, Liu J, Ashraf U, Li G, Li Y, Lu W, Gao L, Han F, Hu J (2016) Exogenous γ-aminobutyric acid (GABA) application improved early growth, net photosynthesis, and associated physio-biochemical events in maize. Front Plant Sci 7:919. https://doi.org/10.3389/fpls.2016.00
Ma JF, Yamaji N (2006) Silicon uptake and accumulation in higher plants. Trends Plant Sci 11:392–397. https://doi.org/10.1016/j.tplants.2006.06.007
Ma JF, Yamaji N, Mitani N et al (2008) Transporters of arsenite in rice and their role in arsenic accumulation in rice grain. Proc Natl Acad Sci USA 105:9931–9935. https://doi.org/10.1073/pnas.0802361105
Mitra A, Chatterjee S, Moogouei R, Gupta DK (2017) Arsenic accumulation in rice and probable mitigation approaches: a review. Agronomy 7:67
Ngo DH, Vo TS (2019) Updated review on pharmaceutical properties of gamma-aminobutyric acid. Molecules 24:2678. https://doi.org/10.3390/molecules24152678
Nishimura M, Yoshida SI, Haramoto M (2016) Effects of white rice containing enriched gamma-aminobutyric acid on blood pressure. J Tradit Complement Med 6:6–71. https://doi.org/10.1016/j.jtcme.2014.11.022
Nozoye T, Nagasaka S, Kobayashi T et al (2011) Phytosiderophore efflux transporters are crucial for iron acquisition in graminaceous plants. J Biol Chem 286:5446–5454. https://doi.org/10.1074/jbc.M110.180026
Pan D, Yi J, Li F et al (2020) Dynamics of gene expression associated with arsenic uptake and transport in rice during the whole growth period. BMC Plant Biol 20:133. https://doi.org/10.1186/s12870-020-02343-1
Paszkowski U, Kroken S, Roux C, Briggs SP (2002) Rice phosphate transporters include an evolutionarily divergent gene specifically activated in arbuscular mycorrhizal symbiosis. Proc Natl Acad Sci 99:13324–13329. https://doi.org/10.1073/pnas.202474599
Pathare V, Srivastava S, Suprasanna P (2013) Evaluation of effects of arsenic on carbon, nitrogen, and sulfur metabolism in two contrasting varieties of Brassica juncea. Acta Physiol Plant 35:3–15. https://doi.org/10.1007/s11738-013-1370-2
Patten CL, Glick BR (2002) Role of Pseudomonas putida indole acetic acid in development of the host plant root system. Appl Environ Microbiol 68:3795–3801. https://doi.org/10.1128/AEM.68.8.3795-3801.2002
Ramesh SA, Tyerman SD, Xu B et al (2015) GABA signalling modulates plant growth by directly regulating the activity of plant-specific anion transporters. Nature Commun 6:78–79. https://doi.org/10.1038/ncomms8879
Ramos-Ruiz R, Martinez F, Knauf-Beiter G (2019) The effects of GABA in plants. Cogent Food Agric 5:1670553. https://doi.org/10.1080/23311932.2019.1670553
Renault H, Amrani AE, Berger A et al (2013) γ-Aminobutyric acid transaminase deficiency impairs central carbon metabolism and leads to cell wall defects during salt stress in Arabidopsis roots. Plant Cell Environ 36:1009–1018. https://doi.org/10.1111/pce.12033
Rout GR, Sahoo S (2015) Role of iron in plant growth and metabolism. Rev Agri Sci 3:1–24. https://doi.org/10.7831/ras.3.1
Rout GR, Das AB, Sahoo S (2014) Screening of iron toxicity in rice genotypes on the basis of morphological, physiological and biochemical analysis. J Exp Biol Agri Sci 2:567–582
Safiuddin M, Shirazi SM, Yussof S (2011) Arsenic contamination of groundwater in Bangladesh: a review. Int J Phys Sci 6:6791–6800. https://doi.org/10.5897/IJPS11.1300
Senoura T, Sakashita E, Kobayashi T et al (2017) The iron-chelate transporter OsYSL9 plays a role in iron distribution in developing rice grains. Plant Mol Biol 95:375–387. https://doi.org/10.1007/s11103-017-0656-y
Singh H, Bhat JA, Singh VP et al (2021) Auxin metabolic network regulates the plant response to metalloids stress. J Hazard Mater 405:124250. https://doi.org/10.1016/j.jhazmat.2020.124250
Song H, Xu X, Wang H et al (2010) Exogenous gamma-aminobutyric acid alleviates oxidative damage caused by aluminium and proton stresses on barley seedlings. J Sci Food Agri 90:1410–1416. https://doi.org/10.1002/jsfa.3951
Song WY, Yamaki T, Yamaji N et al (2014) A rice ABC transporter, OsABCC1, reduces arsenic accumulation in the grain. Proc Natl Acad Sci USA 111:15699–15704. https://doi.org/10.1073/pnas.1414968111
Sun L, Bai Y, Zhang X, Zhou C et al (2021) Characterization of three glutamate decarboxylases from Bacillus spp. for efficient γ-aminobutyric acid production. Microb Cell Fact. 20:153. https://doi.org/10.1186/s12934-021-01646-8
Takagi S (1976) Naturally occurring iron-chelating compounds in oat- and rice-root washing. I. Activity measurement and preliminary characterization. Soil Sci Plant Nutr 22:423–433. https://doi.org/10.1080/00380768.1976.10433004
Tognetti VB, Mühlenbock P, Van Breusegem F (2012) Review stress homeostasis – the redox and auxin perspective. Plant Cell Environ 35:321
Tripathi RD, Srivastava S, Mishra S et al (2007) Arsenic hazards: strategies for tolerance and remediation by plants. Trends Biotechnol 25:158–165. https://doi.org/10.1016/j.tibtech.2007.02.003
Williams PN, Villada A, Deacon C (2007) Greatly enhanced arsenic shoot assimilation in rice leads to elevated grain levels compared to wheat and barley. Environ Sci Technol 41:6854–6859. https://doi.org/10.1021/es070627i
Xiaoming Z, Hua-ming L, Hong H et al (2016) Gamma-aminobutyric acid mediates nicotine biosynthesis in tobacco under flooding stress. Plant Divers 38:53–58. https://doi.org/10.1016/j.pld.2016.05.004
Xie W, Ashraf U, Zhong D et al (2019) Application of γ-aminobutyric acid (GABA) and nitrogen regulates aroma biochemistry in fragrant rice. Food Sci Nutr 7:3784–3796. https://doi.org/10.1002/fsn3.1240
Xu XY, McGrath SP, Meharg AA, Zhao FJ (2008) Growing rice aerobically markedly decreases arsenic accumulation. Environ Sci Technol 42:5574–5579. https://doi.org/10.1021/es800324u
Yadav SS, Hunter D, Redden R et al (2015) Impact of climate change on agriculture production, food, and nutritional security. In: Redden R et al (eds) Crop Wild Relatives and Climate Change. New Jersey, USA: Wiley-Blackwell, pp 1–23. https://doi.org/10.1002/9781118854396.ch1
Zhao FJ, Ma JF, Meharg AA, McGrath SP (2009) Arsenic uptake and metabolism in plants. New Phytol 181:777–794. https://doi.org/10.1111/j.1469-8137.2008.02716.x
Zheng YH, Li X, Li YG et al (2012) Contrasting responses of salinity-stressed salt-tolerant and intolerant winter wheat (Triticum aestivum L.) cultivars to ozone pollution. Plant Physiol Biochem 52:169–178. https://doi.org/10.1016/j.plaphy.2012.01.007
Zhu C, Qi Q, Niu H et al (2021) γ-Aminobutyric acid suppresses iron transportation from roots to shoots in rice seedlings by inducing aerenchyma formation. Int J Mol Sci 22:220. https://doi.org/10.3390/ijms22010220
Funding
This project is funded by the National Research Council of Thailand for New Research (NRCT5-TRG63005-02).
Author information
Authors and Affiliations
Contributions
R.D.: conceptualization, data curation, formal analysis, investigation, methodology, validation, writing—original draft; P.T.: resources, supervision, writing—review and editing.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Disclaimer
We declare that we have no financial and personal relationships with other people or organizations that can inappropriately influence our work.
Additional information
Responsible Editor: Gangrong Shi
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dolphen, R., Thiravetyan, P. Exogenous γ-aminobutyric acid and Bacillus pumilus reduce arsenic uptake and toxicity in rice. Environ Sci Pollut Res 31, 10609–10620 (2024). https://doi.org/10.1007/s11356-024-31893-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-024-31893-2