Abstract
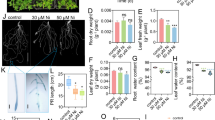
To improve the potential of cadmium phytoremediation, distant hybridization between tobacco (Nicotiana tabacum L. var. 78–04), a high-biomass crop, and Perilla frutescens var. frutescens, a wild Cd-hyperaccumulator, was carried out, developing a new variety N. tabacum L. var. ZSY. Seedlings at the six-leaf stage were grown in hydroponics and treated with 0 (control), 10 µM, 180 µM, and 360 µM CdCl2 for 7 days; then, the differences in Cd tolerance and accumulation and physiological and metabolic responses were evaluated among “ZSY” and its parents. At high Cd dose, the growth of “ZSY,” such as fresh weight, plant height, and root length, was evidently better than “78–04.” In contrast to P. frutescens and “78–04,” “ZSY” could accumulate more Cd in shoots than roots. Under the same treatment, “ZSY” accumulated greater amounts of Cd in both shoots (195–1523 mg kg−1) and roots (140–1281 mg kg−1) than “78–04” (shoots: 35–89 mg kg−1, roots: 39–252 mg kg−1), followed by P. frutescens (shoots: 156–454 mg kg−1, roots: 103–761 mg kg−1). BCF and TF values of “ZSY” reached 38–195 and 1.2–1.4, which were far higher than those of “78–04” (BCF: 2.2–35.3, TF: 0.35–0.9). Perilla frutescens was found with BCF and TF of 11–156 and 0.5–1.5. Cd stress obviously promoted the production of ROS and MDA in seedlings but reduced chlorophyll contents, especially in “78–04.” As a response to Cd stress, “ZSY” had higher SOD and CAT activities when compared to P. frutescens and “78–04,” while “78–04” produced more POD and proline than those of P. frutescens and “ZSY.” Cd stress could affect the production and accumulation of alkaloids and phenolic compounds in root (endodermis and cortex) and mesophyll. At high Cd doses, P. frutescens and “ZSY” had more alkaloids in tissues than “78–04.” Phenolic compounds in “78–04” were more obviously inhibited compared with P. frutescens and “ZSY.” These secondary metabolites may play an important role in eliminating oxidative damage and enhancing Cd tolerance and accumulation in “ZSY” and P. frutescens. Results indicated that distant hybridization could be one of effective methods for introducing excellent genes from metal-hyperaccumulators into high biomass species, creating plants with superior phytoremediation potential.
Graphical abstract









Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Alkorta I, Hernandez-Allica J, Becerril JM, Amezaga I, Albizu I, Garbisu C (2004) Recent findings on the phytoremediation of soils contaminated with environmentally toxic heavy metals and metalloids such as zinc, cadmium, lead, and arsenic. Rev Environ Sci Bio-Technol 3:71–90
Argyropoulou C, Akoumianaki-Ioannidou A, Christodoulakis NS, Fasseas C (2010) Leaf anatomy and histochemistry of Lippia citriodora (Verbenaceae). Aust J Bot 58:398–409
Assunção AG, Pieper B, Vromans J, Lindhout P, Aarts MG, Schat H (2006) Construction of a genetic linkage map of Thlaspi caerulescens and quantitative trait loci analysis of zinc accumulation. New Phytol 1:21–32
Bhargava A, Carmona FF, Bhargava M, Srivastava S (2012) Approaches for enhanced phytoextraction of heavy metals. J Environ Manage 105:103–120
Boojar MMA, Goodarzi F (2007) The copper tolerance strategies and the role of antioxidative enzymes in three plant species grown on copper mine. Chemosphere 67(11):2138–2147
Boudet AM (2007) Evolution and current status of research in phenolic compounds. Phytochemistry 68:2722–2735
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chen SH, Chen X, Dou WH, Wang L, Yin HB, Guo SL (2016) GST (phi) gene from Macrophyte Lemna minor is involved in cadmium exposure responses. Chin J Oceanol Limnol 34:342–351 (in Chinese)
Chen Q, Lu XY, Guo XR, Pan YJ, Yu BF, Tang ZH, Guo QX (2018) Differential responses to Cd stress induced by exogenous application of Cu, Zn or Ca in the medicinal plant Catharanthus roseus. Ecotoxicol Environ Saf 157:266–275
Deng X, Yan X, Wei H, Zhang H, Shen Z (2010) Cadmium-induced oxidative damage and protective effects of N-acetyl-L-cysteine against cadmium toxicity in Solanum nigrum L. J Hazard Mater 180(1–3):722–729
Deniau AX, Pieper B, Ten Bookum WM, Lindhout P, Aarts MGM, Schat H (2006) QTL analysis of cadmium and zinc accumulation in the heavy metal hyperaccumulator Thlaspi caerulescens. Theor Appl Genet 113:907–920
Eapen S, D’Souza SF (2005) Prospects of genetic engineering of plants for phytoremediation of toxic metals. Biotechnol Adv 23(2):97–114
Emamverdian A, Ding YL, Mokhberdoran F, Xie YF (2015) Heavy metal stress and some mechanisms of plant defense response. Sci World J. https://doi.org/10.1155/2015/756120
Gallego SM, Pena LB, Barcia RA, Azpilicueta CE, Iannone MF, Rosales EP, Zawoznik MS, Groppa MD, Benavides MP (2012) Unravelling cadmium toxicity and tolerance in plants: Insight into regulatory mechanisms. Environ Exp Bot 83:33–46
Gomez AA, Mercado MI, Belizán MME, Ponessa G, Vattuone MA, Diego A, Sampietro DA (2019) In situ histochemical localization of alkaloids in leaves and pods of Prosopis ruscifolia. Flora 256:1–6
Gorinova N, Nedkovska M, Todorovska E, Simova-Stoilova L, Stoyanova Z, Georgieva K, Demirevska-Kepova K, Atanassov A, Herzig R (2007) Improved phytoaccumulation of cadmium by genetically modified tobacco plants (Nicotiana tabacum L.). Physiological and biochemical response of the transformants to cadmium toxicity. Environ Pollut 145(1):161–170
Grispen VMJ, Hakvoort HWJ, Bliek T, Verkleij JAC, Schat H (2011) Combined expression of the Arabidopsis metallothionein MT2b and the heavy metal transporting ATPase HMA4 enhances cadmium tolerance and the root to shoot translocation of cadmium and zinc in tobacco. Environ Exp Bot 72:71–76
Guo B, Liang YC, Zhu YG, Zhao FJ (2007) Role of salicylic acid in alleviating oxidative damage in rice roots (Oryza sativa) subjected to cadmium stress. Environ Pollut 147(3):743–749
Han T, Liang YP, Wu ZN, Zhang L, Liu ZW, Li QF, Chen XJ, Guo WL, Jiang LN, Pan FF, Ge SD, Mi ZR, Liu ZC, Huang H, Li XZ, Zhou JG, Li Y, Wang JL, Zhang Z, Tang YY, Yang LR, Wu MD (2019) Effects of tetracycline on growth, oxidative stress response, and metabolite pattern of ryegrass. J Hazard Mater 380:120885
Hassanzadeh M, Ebadi A, Panahyan-e-Kivi M, Eshghi AG, Jamaati-e-Somarin S, Saeidi M, Zabihi-e-Mahmoodabad R (2009) Evaluation of drought stress on relative water content and chlorophyll content of sesame (Sesamum indicum L) genotypes at early flowering stage. Res J Environ Sci 3(3):345–350
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplast. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Hou TY, Netala VR, Zhang HJ, Xing Y, Li HZ, Zhang ZJ (2022) Perilla frutescens: a rich source of pharmacological active compounds. Molecules 27:3578
Iannone MF, Groppa MD, Benavides MP (2015) Cadmium induces different biochemical responses in wild type and catalase-deficient tobacco plants. Environ Exp Bot 109:201–211
Janošević D, Budimir S, Alimpić A, Marin P, Sheef NA, Giweli A, Duletić-Laušević S (2016) Micromorphology and histochemistry of leaf trichomes of Salvia aegyptiaca (Lamiaceae). Arch Biol Sci 68(2):291–301
Joo JH, Wang SY, Chen JG, Jones AM, Fedoroff NV (2005) Different signaling and cell death roles of heterotrimeric G protein α and β subunits in the Arabidopsis oxidative stress response to ozone. Plant Cell 17:957–970
Katoh A, Ohki H, Inai K, Hashimoto T (2005) Molecular regulation of nicotine biosynthesis. Plant Biotechnol 22:389–392
Khator K, Saxena I, Shekhawat GS (2021) Nitric oxide induced Cd tolerance and phytoremediation potential of B. juncea by the modulation of antioxidant defense system and ROS detoxification. Biometals 34:15–32
Kim KH, Lee YH, Kim D, Park YH, Lee JY, Hwang YS, Kim YH (2004) Agrobacterium-mediated genetic transformation of Perilla frutescens. Plant Cell Rep 6:386–390
Liang HM, Lin TH, Chiou JM, Yeh KC (2009) Model evaluation of the phytoextraction potential of heavy metal hyperaccumulators and non-hyperaccumulators. Environ Pollut 157(6):1945–1952
Liu X, Zhang SR, Zhu R, Xu XX, Fan XW (2009) Effect of cadmium and copper stress on the growth response and accumulation characteristics of Perilla frutescens (L.) Britt. J Agro-Environ Sci 11:2264–2269 (in Chinese)
Liu WX, Shang SH, Feng X, Zhang GP, Wu FB (2015) Modulation of exogenous selenium in cadmium-induced changes in antioxidative metabolism, cadmium uptake, and photosynthetic performance in the 2 tobacco genotypes differing in cadmium tolerance. Environ Toxicol Chem 34(1):92–99
Lugon-Moulin N, Martin F, Krauss MR, Ramey PB, Rossi L (2006) Cadmium concentration in tobacco (Nicotiana tabacum L.) from different countries and its relationship with other elements. Chemosphere 63(7):1074–1086
Manquián-Cerda K, Escudey M, Zúñiga G, Arancibia-Miranda N, Molina M, Cruces E (2016) Effect of cadmium on phenolic compounds, antioxidant enzyme activity and oxidative stress in blueberry (Vaccinium corymbosum L.) plantlets grown in vitro. Ecotoxicol Environ Saf 133:316–326
Martin F, Bovet L, Cordier A, Stanke M, Gunduz I, Peitsch MC, Ivanov NV (2012) Design of a tobacco exon array with application to investigate the differential cadmium accumulation property in two tobacco varieties. BMC Genomics 13(1):1–16
Matias LJ, Mercadante-Simões MO, Royo VA, Ribeiro LM, Santos AC, Fonseca JMS (2016) Structure and histochemistry of medicinal species of Solanum. Rev Bras 26:147–160
Mesjasz-Przybylowicz J, Barnabas A, Przybyiowicz W (2007) Comparison of cytology and distribution of nickel in roots of Ni-hyperaccumulating and non-hyperaccumulating genotypes of Senecio coronatus. Plant Soil 293:61–78
Michalak A (2006) Phenolic compounds and their antioxidant activity in plants growing under heavy metal stress. Pol J Environ Stud 15(4):523–530
Qiu RL, Zhao X, Tang YT, Yu FM, Hu PJ (2009) Antioxidative response to Cd in a newly discovered cadmium hyperaccumulator, Arabis paniculata F. Chemosphere 74(1):6–12
Rasheed R, Ashraf MA, Arshad A, Iqbal M, Hussain I (2020) Interactive effects of chitosan and cadmium on growth, secondary metabolism, oxidative defense, and element uptake in pea (Pisum sativum L.). Arab J Geosci 13:847
Sa KJ, Kim JA, Lee JK (2012) Comparison of seed characteristics between the cultivated and the weedy types of Perilla species. Hortic Environ Biotechnol 4:310–315
Sakihama Y, Cohen MF, Grace SC, Yamasaki H (2002) Plant phenolic antioxidant and prooxidant activities: phenolics induced oxidative damage mediated by metals in plants. Toxicology 177:67–80
Shukla D, Huda KMK, Banu MSA, Gill SS, Tuteja R, Tuteja N (2014) OsACA6, a P-type 2B Ca2+ ATPase functions in cadmium stress tolerance in tobacco by reducing the oxidative stress load. Planta 240(4):809–824
Soukupová J, Cvikrová M, Albrechtová J, Rock BN, Eder J (2000) Histochemical and biochemical approaches to the study of phenolic compounds and peroxidases in needles of Norway spruce (Picea abies). New Phytol 146(3):403–414
Tajik S, Zarinkamar F, Soltani BM, Nazari M (2019) Induction of phenolic and flavonoid compounds in leaves of saffron (Crocus sativus L.) by salicylic acid. Sci Hortic 257:108751
Touati M, Bottega S, Castiglione MR, Sorce C, Béjaoui Z, Spanò C (2019) Modulation of the defence responses against Cd in willow species through a multifaceted analysis. Plant Physiol Biochem 142:125–136
Tozin LRS, Rodrigues TM (2017) Morphology and histochemistry of glandular trichomes in Hyptis villosa Pohl ex Benth. (Lamiaceae) and differential labeling of cytoskeletal elements. Acta Bot Bras 31(3):330–343
Vamerali T, Bandiera M, Mosca G (2010) Field crops for phytoremediation of metal-contaminated land. A review. Environ Chem Lett 8:1–17
Wang P, Liang Z, Zeng J, Li W, Sun X, Miao Z, Tang K (2008a) Generation of tobacco lines with widely different reduction in nicotine levels via RNA silencing approaches. J Biosci 2:177–184
Wang Z, Zhang YX, Huang ZB, Huang L (2008b) Antioxidative response of metal-accumulator and non-accumulator plants under cadmium stress. Plant Soil 310:137–149
Wang JB, Su LY, Yang JZ, Yuan JG, Ag Y, Qiu Q, Zhang K, Yang ZY (2015) Comparisons of cadmium subcellular distribution and chemical forms between low-Cd and high-Cd accumulation genotypes of watercress (Nasturtium officinale L. R. Br.). Plant Soil 396:325–337
Wei ZZ, Wei KQ (2008) Distant hybridization breeding of tobacco. China Agricultural Science and Technology Press, Beijing (in Chinese)
Wei ZZ, Yan XP, Deng ZF, Yang JX, Wang YT (2005) Research on distant hybridization between tobacco and D. stramonium. Chin Tob Sci 1:1–5 (in Chinese)
Wei KQ, Yang JX, Wei ZZ (2010) Chemical components and medicine ingredients in the hybrid of families between tobacco and medicinal plant Perilla frutescens (L.) Britt. Nat Prod Res Dev 22:801–806 (in Chinese)
Wei KQ, Pang SX, Yang JX, Wei ZZ (2015) Enhancement of cadmium tolerance and accumulation by introducing Perilla frutescens (L.) Britt var. frutescens genes in Nicotiana tabacum L. plants. Environ Sci Pollut Res 22:5405–5416
Xie HL, Chen AP, Zhang FY, Xiong Y, Chen YL, Liu J, Zhou LY, Lin RY (2011) Physiological response of Perilla frutescens (L.) Britt. to cadmium. Chin J Eco-Agric 3:672–675 (in Chinese)
Xie LP, He XY, Shang SH, Zheng WT, Liu WX, Zhang GP, Wu FB (2014) Comparative proteomic analysis of two tobacco (Nicotiana tabacum) genotypes differing in Cd tolerance. Biometals 27:1277–1289
Yang XE, Feng Y, He ZL, Stoffella PJ (2005) Molecular mechanisms of heavy metal hyperaccumulation and phytoremediation. J Trace Elem Med Biol 18(4):339–353
Yılmaza DD, Parlak KU (2011) Changes in proline accumulation and antioxidative enzyme activities in Groenlandia densa under cadmium stress. Ecol Ind 11(2):417–423
Zhan XR, Chen ZH, Chen R, Shen CJ (2022) Environmental and genetic factors involved in plant protection-associated secondary metabolite biosynthesis pathways. Front Plant Sci 13:877304
Zhang DZ, Wang PH, Zhao HX (1990) Determination of the content of free proline in wheat leaves. Plant Physiol Commun 4:62–65
Zhang QH, Yang FJ, Li BX, Zhou GZ (2007) Analysis and evaluation of heavy metals in the soil and plant system in some mining area of southwest China. J Shandong Univ Sci Technol 3:15–18 (in Chinese)
Zhang X, Wu W, Zheng YL, Chen L, Cai QR (2009) Essential oil variations in different Perilla L. accessions: chemotaxonomic implications. Plant Syst Evol 281:1–10
Zhang J, Zhang M, Shohag MJI, Tian SK, Song HY, Feng Y, Yang XE (2016) Enhanced expression of SaHMA3 plays critical roles in Cd hyperaccumulation and hypertolerance in Cd hyperaccumulator Sedum alfredii Hance. Planta 243:577–589
Zheng ZG, Wu M (2004) Cadmium treatment enhances the production of alkaloid secondary metabolites in Catharanthus roseus. Plant Sci 166(2):507–514
Funding
This work was supported by the Shanxi Provincial Key Technologies Research and Development Program (201603D321002).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Keqiang Wei: conceptualization, methodology, supervision, writing, and funding acquisition; Tingting Guo: methodology, experiments and data analysis.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Elena Maestri
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Hybrid tobacco displayed larger biomass, stronger tolerance, and higher accumulation than female parent.
• Introducing Perilla frutescens genes changed Cd distribution and accumulation in tobacco tissues.
• Introducing Perilla frutescens genes alleviated Cd-induced oxidative stress in tobacco.
• Introducing Perilla frutescens genes changed metabolic response of tobacco to Cd.
• Distant hybridization could create plant with superior phytoremediation potential.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wei, K., Guo, T. Enhancing the potential for cadmium phytoremediation by introducing Perilla frutescens genes in tobacco. Environ Sci Pollut Res 30, 70039–70053 (2023). https://doi.org/10.1007/s11356-023-27392-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-27392-5