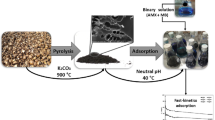
Abstract
In this study, spent tea powder waste was chemically treated for the synthesis of adsorbent using two activating agents, i.e., sulfuric acid and phosphoric acid, to obtain sulfuric acid activated carbon (SAC) and phosphoric acid activated carbon (PAC). The performance of PAC and SAC for the sorption of tetracycline (TCY) and sulfadiazine (SDZ) antibiotics from mono-component (SDZ/TCY) and multi-component (SDZ + TCY) adsorption systems was investigated. Synergistic and antagonistic effects were studied in removing target pollutants in SDZ + TCY systems. Kinetic and equilibrium studies were modeled by different kinetic and isotherm models. The adsorption capacity was assessed using Langmuir’s competitive model in a \({multi-component \ system}\). Pseudo-first-order kinetic and Langmuir isotherm models best fit the experimental kinetic and equilibrium data to remove antibiotics. The Langmuir’s maximum adsorption capacity (qm) of PAC for the removal of SDZ and TCY in a \({mono-component \ system}\) was found to be 16.75 and 10.87 mg/g, and qm of SAC for the removal of SDZ and TCY was found to be 24.69 and 23.20 mg/g, respectively. In SDZ + TCY multi-component system, adsorption of TCY was synergistic in nature for both PAC and SAC. Sorption of SDZ displayed an antagonistic effect in the SDZ + TCY system for both SAC and PAC. In conclusion, the activated carbons synthesized from spent tea waste could be effectively adopted for the simultaneous adsorption of SDZ and TCY from multi-component systems.





Similar content being viewed by others
Data availability
Included in the manuscript.
References
Al-Qodah Z, Shawabkah R (2009) Production and characterization of granular activated carbon from activated sludge. Brazilian J Chem Eng 26:127–136. https://doi.org/10.1590/S0104-66322009000100012
Ben Y, Hu M, Zhang X et al (2020) Efficient detection and assessment of human exposure to trace antibiotic residues in drinking water. Water Res 175. https://doi.org/10.1016/j.watres.2020.115699
Bourbigot S, Le Bras M, Delobel R et al (1995) Carbonization mechanisms resulting from intumescence-part II. Association with an ethylene terpolymer and the ammonium polyphosphate-pentaerythritol fire retardant system. Carbon N Y 33:283–294. https://doi.org/10.1016/0008-6223(94)00131-I
Brigante M, Schulz PC (2011) Remotion of the antibiotic tetracycline by titania and titania-silica composed materials. J Hazard Mater 192:1597–1608. https://doi.org/10.1016/j.jhazmat.2011.06.082
Cai HM, Chen GJ, Peng CY et al (2015) Removal of fluoride from drinking water using tea waste loaded with Al/Fe oxides: A novel, safe and efficient biosorbent. Applied Surface Science 328:34–44. https://doi.org/10.1016/j.apsusc.2014.11.164
Caicedo-Salcedo OD, Vargas-Delgadillo DP, Giraldo L, Moreno-Piraján JC (2019) Data of preparation and characterization of activated carbon using two activant agents and mango seed as precursor material. Data Br 27. https://doi.org/10.1016/j.dib.2019.104769
Dai Y, Li J, Shan D (2020) Adsorption of tetracycline in aqueous solution by biochar derived from waste Auricularia auricula dregs. Chemosphere 238. https://doi.org/10.1016/j.chemosphere.2019.124432
Debnath B, Majumdar M, Bhowmik M et al (2020) The effective adsorption of tetracycline onto zirconia nanoparticles synthesized by novel microbial green technology. J Environ Manage 261. https://doi.org/10.1016/j.jenvman.2020.110235
Gao Y, Li Y, Zhang L et al (2012) Adsorption and removal of tetracycline antibiotics from aqueous solution by graphene oxide. J Colloid Interface Sci 368:540–546. https://doi.org/10.1016/j.jcis.2011.11.015
Goze B, Evirgen OA, Acikel YS (2016) Investigation of antagonistic and synergistic interactions on simultaneous adsorption of crystal violet and Cu(II) using chitin and chitosan. Desalin Water Treat 57:4059–4072. https://doi.org/10.1080/19443994.2014.989914
Hamdaoui O, Naffrechoux E (2007) Modeling of adsorption isotherms of phenol and chlorophenols onto granular activated carbon. Part I. Two-parameter models and equations allowing determination of thermodynamic parameters. J Hazard Mater 147:381–394. https://doi.org/10.1016/j.jhazmat.2007.01.021
He X, Li J, Meng Q et al (2020) Enhanced adsorption capacity of sulfadiazine on tea waste biochar from aqueous solutions by the two-step sintering method without corrosive activator. J Environ Chem Eng 9. https://doi.org/10.1016/j.jece.2020.104898
Hoslett J, Ghazal H, Katsou E, Jouhara H (2021) The removal of tetracycline from water using biochar produced from agricultural discarded material. Sci Total Environ 751. https://doi.org/10.1016/j.scitotenv.2020.141755
Jiang H, Duan Y, Liu Y, et al (2020) Removal of tetracycline from wastewater by activated peroxymonosulfate using calcined kaolin. Chinese J Environ Eng 14:2494–2505. https://doi.org/10.12030/j.cjee.202003163
Kang J, Liu H, Zheng YM et al (2010) Systematic study of synergistic and antagonistic effects on adsorption of tetracycline and copper onto a chitosan. J Colloid Interface Sci 344:117–125. https://doi.org/10.1016/j.jcis.2009.11.049
Kumar PS, Ramalingam S, Kirupha SD et al (2011) Adsorption behavior of nickel(II) onto cashew nut shell: equilibrium, thermodynamics, kinetics, mechanism and process design. Chem Eng J 167:122–131. https://doi.org/10.1016/j.cej.2010.12.010
Lamshöft M, Sukul P, Zühlke S, Spiteller M (2007) Metabolism of 14C-labelled and non-labelled sulfadiazine after administration to pigs. Anal Bioanal Chem 388:1733–1745. https://doi.org/10.1007/s00216-007-1368-y
Lataye DH, Mishra IM, Mall ID (2011) Removal of 4-picoline from aqueous solution by adsorption onto bagasse fly ash and rice husk ash: equilibrium, thermodynamic, and desorption study. J Environ Eng 137:1048–1057. https://doi.org/10.1061/(ASCE)EE.1943-7870.0000423
Liu JL, Wong MH (2013) Pharmaceuticals and personal care products (PPCPs): a review on environmental contamination in China. Environ Int 59:208–224. https://doi.org/10.1016/j.envint.2013.06.012
Liu WJ, Zeng FX, Jiang H, Zhang XS (2011) Preparation of high adsorption capacity bio-chars from waste biomass. Bioresour Technol 102:8247–8252. https://doi.org/10.1016/j.biortech.2011.06.014
Liu X, Liu Y, Lu S et al (2018) Performance and mechanism into TiO2/Zeolite composites for sulfadiazine adsorption and photodegradation. Chem Eng J 350:131–147. https://doi.org/10.1016/j.cej.2018.05.141
Luo X, Zhang Z, Zhou P et al (2015) Synergic adsorption of acid blue 80 and heavy metal ions (Cu2+/Ni2+) onto activated carbon and its mechanisms. J Ind Eng Chem 27:164–174. https://doi.org/10.1016/j.jiec.2014.12.031
Mall ID, Srivastava VC, Agarwal NK (2006a) Removal of orange-G and methyl violet dyes by adsorption onto bagasse fly ash - kinetic study and equilibrium isotherm analyses. Dye Pigment 69:210–223. https://doi.org/10.1016/j.dyepig.2005.03.013
Mall ID, Srivastava VC, Kumar GVA, Mishra IM (2006b) Characterization and utilization of mesoporous fertilizer plant waste carbon for adsorptive removal of dyes from aqueous solution. Colloids Surfaces A Physicochem Eng Asp 278:175–187. https://doi.org/10.1016/j.colsurfa.2005.12.017
Manjunath SV, Kumar M (2018) Evaluation of single-component and multi-component adsorption of metronidazole, phosphate and nitrate on activated carbon from Prosopıs julıflora. Chem Eng J 346:525–534. https://doi.org/10.1016/j.cej.2018.04.013
Manjunath SV, Kumar M (2021a) Removal and recovery of nutrients using low-cost adsorbents from single-component and multicomponent adsorption systems. In: Guo W, Ngo HH, Surampalli RY, Zhang TC (eds) Sustainable Resource Management : Technologies for Recovery and Reuse ofEnergy andWaste Materials, 1st edn. Wiley-VCH, pp 397–435
Manjunath SV, Kumar SM, Ngo HH, Guo W (2017) Metronidazole removal in powder-activated carbon and concrete-containing graphene adsorption systems: estimation of kinetic, equilibrium and thermodynamic parameters and optimization of adsorption by a central composite design. J Environ Sci Heal Part A 52:1269–1283. https://doi.org/10.1080/10934529.2017.1357406
Manjunath SV, Baghel RS, Kumar M (2019) Performance evaluation of cement-carbon composite for adsorptive removal of acidic and basic dyes from single and multi-component systems. Environ Technol Innov 16:100478. https://doi.org/10.1016/j.eti.2019.100478
Manjunath SV, Singh Baghel R, Kumar M (2020) Antagonistic and synergistic analysis of antibiotic adsorption on prosopis juliflora activated carbon in multicomponent systems. Chem Eng J 122713. https://doi.org/10.1016/j.cej.2019.122713
Manjunath S V, Kumar M (2021b) Removal and recovery of nutrients using low-cost adsorbents from single-component and multicomponent adsorption systems
Martins AC, Pezoti O, Cazetta AL et al (2015) Removal of tetracycline by NaOH-activated carbon produced from macadamia nut shells: kinetic and equilibrium studies. Chem Eng J 260:291–299. https://doi.org/10.1016/j.cej.2014.09.017
Meng Q, Zhang Y, Meng D et al (2020) Removal of sulfadiazine from aqueous solution by in-situ activated biochar derived from cotton shell. Environ Res 191. https://doi.org/10.1016/j.envres.2020.110104
Mohammad YS, Shaibu-Imodagbe EM, Igboro SB et al (2015) Effect of phosphoric acid modification on characteristics of rice husk activated carbon. Iran J Energy Environ 6:20–25. https://doi.org/10.5829/idosi.ijee.2015.06.01.05
Mohtashami SA, AsasianKolur N, Kaghazchi T et al (2018) Optimization of sugarcane bagasse activation to achieve adsorbent with high affinity towards phenol. Turkish J Chem 42:1720–1735. https://doi.org/10.3906/kim-1806-71
Nguyen CH, Fu CC, Kao DY et al (2020) Adsorption removal of tetracycline from water using poly(vinylidene fluoride)/polyaniline-montmorillonite mixed matrix membranes. J Taiwan Inst Chem Eng 112:259–270. https://doi.org/10.1016/j.jtice.2020.06.007
Padmavathy KS, Madhu G, Haseena PV (2016) A study on effects of pH, adsorbent dosage, time, initial concentration and adsorption isotherm study for the removal of hexavalent chromium (Cr (VI)) from wastewater by magnetite nanoparticles. Procedia Technol 24:585–594. https://doi.org/10.1016/j.protcy.2016.05.127
Peiris C, Gunatilake SR, Mlsna TE et al (2017) Biochar based removal of antibiotic sulfonamides and tetracyclines in aquatic environments: a critical review. Bioresour Technol 246:150–159. https://doi.org/10.1016/j.biortech.2017.07.150
Puziy AM, Poddubnaya OI, Martínez-Alonso A et al (2002) Synthetic carbons activated with phosphoric - acid I. Surface chemistry and ion binding properties. Carbon N Y 40:1493–1505. https://doi.org/10.1016/S0008-6223(01)00317-7
Rajak VK, Kumar S, Thombre NV, Mandal A (2018) Synthesis of activated charcoal from saw-dust and characterization for adsorptive separation of oil from oil-in-water emulsion. Chem Eng Commun 205:897–913. https://doi.org/10.1080/00986445.2017.1423288
Rajasekaran SJ, Raghavan V (2020) Facile synthesis of activated carbon derived from Eucalyptus globulus seed as efficient electrode material for supercapacitors. Diam Relat Mater 109. https://doi.org/10.1016/j.diamond.2020.108038
Ratnaji T, Kennedy LJ (2020) Hierarchical porous carbon derived from tea waste for energy storage applications: waste to worth. Diam Relat Mater 110. https://doi.org/10.1016/j.diamond.2020.108100
Rong SP, Sun YB, Zhao ZH (2014) Degradation of sulfadiazine antibiotics by water falling film dielectric barrier discharge. Chinese Chem Lett 25:187–192. https://doi.org/10.1016/j.cclet.2013.11.003
Rouhani M, Ashrafi SD, Taghavi K et al (2022) Evaluation of tetracycline removal by adsorption method using magnetic iron oxide nanoparticles (Fe3O4) and clinoptilolite from aqueous solutions. J Mol Liq 356. https://doi.org/10.1016/j.molliq.2022.119040
Sirimuangjinda A, Atong D, Pechyen C (2013) Comparison on pore development of activated carbon produced from scrap tire by hydrochloric acid and sulfuric acid. Adv Mater Res 626:706–710. https://doi.org/10.4028/www.scientific.net/AMR.626.706
Srivastava VC, Mall ID, Mishra IM (2008) Removal of cadmium(II) and zinc(II) metal ions from binary aqueous solution by rice husk ash. Colloids Surfaces A Physicochem Eng Asp 312:172–184. https://doi.org/10.1016/j.colsurfa.2007.06.048
Srivastava VC, Mall ID, Mishra IM (2009) Equilibrium modeling of ternary adsorption of metal ions onto rice husk ash. J Chem Eng Data 54:705–711. https://doi.org/10.1021/je8003029
Sun J, Wang Q, Zhang J et al (2018) Degradation of sulfadiazine in drinking water by a cathodic electrochemical membrane filtration process. Electrochim Acta 277:77–87. https://doi.org/10.1016/j.electacta.2018.05.005
Tan X, Liu Y, Zeng G et al (2015) Application of biochar for the removal of pollutants from aqueous solutions. Chemosphere 125:70–85. https://doi.org/10.1016/j.chemosphere.2014.12.058
Tanis E, Hanna K, Emmanuel E (2008) Experimental and modeling studies of sorption of tetracycline onto iron oxides-coated quartz. Colloids Surfaces A Physicochem Eng Asp 327:57–63. https://doi.org/10.1016/j.colsurfa.2008.06.013
Temkin MJ, Phyzev V (1940) Recent modifications to Langmuir isotherms. Acta Physico-Chimica USSR 12:217–222
Tovar-Gómez R, del Moreno-Virgen M, R, Moreno-Pérez J, et al (2015) Analysis of synergistic and antagonistic adsorption of heavy metals and acid blue 25 on activated carbon from ternary systems. Chem Eng Res Des 93:755–772. https://doi.org/10.1016/j.cherd.2014.07.012
Turan B, Sarigol G, Demircivi P (2022) Adsorption of tetracycline antibiotics using metal and clay embedded cross-linked chitosan. Mater Chem Phys 279. https://doi.org/10.1016/j.matchemphys.2022.125781
Vieno NM, Härkki H, Tuhkanen T, Kronberg L (2007) Occurrence of pharmaceuticals in river water and their elimination in a pilot-scale drinking water treatment plant. Environ Sci Technol 41:5077–5084. https://doi.org/10.1021/es062720x
Vu TH, Ngo TMV, Duong TTA et al (2020) Removal of tetracycline from aqueous solution using nanocomposite based on polyanion-modified laterite material. J Anal Methods Chem 2020:25–27. https://doi.org/10.1155/2020/6623511
Wang H, Wang S, Jiang JQ, Shu J (2019) Removal of sulfadiazine by ferrate(VI) oxidation and montmorillonite adsorption - synergistic effect and degradation pathways. J Environ Chem Eng 7. https://doi.org/10.1016/j.jece.2019.103225
Wang Y, Gong S, Li Y et al (2020) Adsorptive removal of tetracycline by sustainable ceramsite substrate from bentonite/red mud/pine sawdust. Sci Rep 10:1–18. https://doi.org/10.1038/s41598-020-59850-2
Xie D, Zhang H, Jiang M et al (2020) Adsorptive removal of tetracycline from water using Fe(III)-functionalized carbonized humic acid. Chinese J Chem Eng 28:2689–2698. https://doi.org/10.1016/j.cjche.2020.06.039
Yadav MSP, Neghi N, Kumar M, Varghese GK (2018) Photocatalytic-oxidation and photo-persulfate-oxidation of sulfadiazine in a laboratory-scale reactor: analysis of catalyst support, oxidant dosage, removal-rate and degradation pathway. J Environ Manage 222:164–173. https://doi.org/10.1016/j.jenvman.2018.05.052
Yakout SM, Sharaf El-Deen G (2016) Characterization of activated carbon prepared by phosphoric acid activation of olive stones. Arab J Chem 9:S1155–S1162. https://doi.org/10.1016/j.arabjc.2011.12.002
Yang Y, Zheng L, Zhang T et al (2019) Adsorption behavior and mechanism of sulfonamides on phosphonic chelating cellulose under different pH effects. Bioresour Technol 288. https://doi.org/10.1016/j.biortech.2019.121510
Yu JX, Cai XL, Feng LY et al (2015) Synergistic and competitive adsorption of cationic and anionic dyes on polymer modified yeast prepared at room temperature. J Taiwan Inst Chem Eng 57:98–103. https://doi.org/10.1016/j.jtice.2015.05.018
Zhang L, Song X, Liu X et al (2011) Studies on the removal of tetracycline by multi-walled carbon nanotubes. Chem Eng J 178:26–33. https://doi.org/10.1016/j.cej.2011.09.127
Zhang L, Wang Y, Jin SW et al (2017) Adsorption isotherm, kinetic and mechanism of expanded graphite for sulfadiazine antibiotics removal from aqueous solutions. Environ Technol (united Kingdom) 38:2629–2638. https://doi.org/10.1080/09593330.2016.1272637
Zhang L, Xin Z, Fei X et al (2019) Study on adsorption of tetracycline by red mud-based ceramsite. J Water Supply Res Technol - AQUA 68:39–50. https://doi.org/10.2166/aqua.2018.100
Zhao Z, Nie T, Zhou W (2019) Enhanced biochar stabilities and adsorption properties for tetracycline by synthesizing silica-composited biochar. Environ Pollut 254. https://doi.org/10.1016/j.envpol.2019.113015
Zuo X, Qian C, Ma S, Xiong J (2020) Sulfonamide antibiotics sorption by high silica ZSM-5: effect of pH and humic monomers (vanillin and caffeic acid). Chemosphere 248. https://doi.org/10.1016/j.chemosphere.2020.126061
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. Characterization studies—Nayanathara O Sanjeev and Abinaya Sekar.
Experimentation—Nayanathara O Sanjeev and Mandala Siva Priyanka Yadav.
Modeling—Mandala Siva Priyanka Yadav and Manjunath Singanodi Vallabha.
Manuscript preparation—Mandala Siva Priyanka Yadav, Nayanathara O Sanjeev, Manjunath Singanodi Vallabha, and Abinaya Sekar.
Supervision and proofreading—George Kuttiparichel Varghese and Aswathy Erat Valsan.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yadav, M.S.P., Sanjeev, N.O., Vallabha, M.S. et al. Competitive adsorption analysis of antibiotics removal from multi-component systems using chemically activated spent tea waste: effect of operational parameters, kinetics, and equilibrium study. Environ Sci Pollut Res 30, 42697–42712 (2023). https://doi.org/10.1007/s11356-022-22323-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-22323-2