Abstract
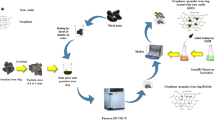
Sugar molasses from agricultural waste could be a sustainable carbon source for the synthesis of graphene adsorbent introduced in this work. The sugar molasses was successfully converted to graphene-like material and subsequently coated on the sand as graphene sand composite (GSC), as proven by XRD, XPS, Raman spectroscopy, and SEM with EDX mapping analyses. The adsorption performance of GSC was evaluated against the removal of Tetracycline (TC) and methylene blue (MB) pollutants from an aqueous solution in a fixed bed column continuous-flow adsorption setup. The effect of different process conditions: bed height (4–12 cm), influent flow rate (3–7 mL/min), and contaminants’ concentration (50–150 ppm) was investigated. The results revealed that column performance was improved by increasing the bed depth and lowering the flow rate and concentration of the pollutants. The best removal efficiency was obtained when the bed height was 12 cm, the influent flow rate of 3 mL/min, and the pollutants’ initial concentration was 50 mg/L. Thomas, Adams-Bohart, and Yoon-Nelson models were attempted to fit the breakthrough curves. Regeneration of the GSC indicated the decline of breakthrough time from 240–280 to 180 min, reflecting the decrease in adsorptive sites due to the incomplete regeneration process. Overall, sugar molasses was shown to be a low-cost precursor for synthesizing valuable graphene material in the form of GSC, which can reduce the problem for industrial waste management of sugar molasses, and the GSC could be used as an adsorbent for environmental application.















Similar content being viewed by others
References
Abbasi M, Safari E, Baghdadi M, Janmohammadi M (2021) Enhanced adsorption of heavy metals in groundwater using sand columns enriched with graphene oxide: lab-scale experiments and process modeling. J Water Process Eng 40:101961. https://doi.org/10.1016/j.jwpe.2021.101961
Abd Ali ZT (2021) Green synthesis of graphene-coated sand (GCS) using low-grade dates for evaluation and modeling of the pH-dependent permeable barrier for remediation of groundwater contaminated with copper. Sep Sci Technol 56:14–25. https://doi.org/10.1080/01496395.2019.1708937
Abu Khadra MR, Eid MH, Allam AA et al (2020) Evaluation of different forms of Egyptian diatomite for the removal of ammonium ions from Lake Qarun: a realistic study to avoid eutrophication. Environ Pollut 266:115277. https://doi.org/10.1016/j.envpol.2020.115277
Ang WL, Mohammad AW (2019) Chapter 9 - Integrated and hybrid process technology. In: Sustainable Water and Wastewater Processing (Book). Elsevier, pp 279–328. https://doi.org/10.1016/B978-0-12-816170-8.00009-0
Aryee AA, Mpatani FM, Kani AN et al (2021) A review on functionalized adsorbents based on peanut husk for the sequestration of pollutants in wastewater: modification methods and adsorption study. J Clean Prod 310:127502. https://doi.org/10.1016/J.JCLEPRO.2021.127502
Chen S, Yue Q, Gao B et al (2012) Adsorption of hexavalent chromium from aqueous solution by modified corn stalk: a fixed-bed column study. Bioresour Technol 113:114–120. https://doi.org/10.1016/J.BIORTECH.2011.11.110
Chu KH (2010) Fixed bed sorption: Setting the record straight on the Bohart-Adams and Thomas models. J Hazard Mater 177:1006–1012. https://doi.org/10.1016/J.JHAZMAT.2010.01.019
Chua SF, Nouri A, Ang WL et al (2021) The emergence of multifunctional adsorbents and their role in environmental remediation. J Environ Chem Eng 9:104793. https://doi.org/10.1016/J.JECE.2020.104793
Cruz MAP, Guimarães LCM, da Costa Júnior EF et al (2019) Adsorption of crystal violet from aqueous solution in continuous flow system using bone char. Chem Eng Commun 207:372–381. https://doi.org/10.1080/00986445.2019.1596899
Dileepkumar N, Ganesh S, Gopinathan S et al (2020) Green synthesis of graphene sand composites for the removal of hazardous chemicals from water. AIP Conf Proc 2283:020017. https://doi.org/10.1063/5.0025108
Du Z, Zheng T, Wang P (2018) Experimental and modelling studies on fixed bed adsorption for Cu(II) removal from aqueous solution by carboxyl modified jute fiber. Powder Technol 338:952–959. https://doi.org/10.1016/j.powtec.2018.06.015
Egbosiuba TC, Abdulkareem AS (2021) Highly efficient as-synthesized and oxidized multi-walled carbon nanotubes for copper(II) and zinc(II) ion adsorption in a batch and fixed-bed process. J Mater Res Technol 15:2848–2872. https://doi.org/10.1016/J.JMRT.2021.09.094
El-sayed MEA (2020) Nanoadsorbents for water and wastewater remediation. Sci Total Environ 739:139903. https://doi.org/10.1016/j.scitotenv.2020.139903
Graimed BH, Abd Ali ZT (2022) Batch and continuous study of one-step sustainable green graphene sand hybrid synthesized from Date-syrup for remediation of contaminated groundwater. Alexandria Eng J 61:8777–8796. https://doi.org/10.1016/j.aej.2022.02.018
Gupta SS, Sreeprasad TS, Maliyekkal SM et al (2012) Graphene from sugar and its application in water purification. ACS Appl Mater Interfaces 4:4156–4163. https://doi.org/10.1021/am300889u
Khan S, Achazhiyath Edathil A, Banat F (2019) Sustainable synthesis of graphene-based adsorbent using date syrup. Sci Rep 9:1–14. https://doi.org/10.1038/s41598-019-54597-x
Khoshnam M, Farahbakhsh J, Zargar M et al (2021) α-Fe2O3/graphene oxide powder and thin film nanocomposites as peculiar photocatalysts for dye removal from wastewater. Sci Rep 11:1–15. https://doi.org/10.1038/s41598-021-99849-x
Kong Y, Zhuang Y, Shi B (2020) Tetracycline removal by double-metal-crosslinked alginate/graphene hydrogels through an enhanced Fenton reaction. J Hazard Mater 382:121060. https://doi.org/10.1016/j.jhazmat.2019.121060
Kumar P, Pérez JAE, Cledon M et al (2020a) Removal of microcystin-LR and other water pollutants using sand coated with bio-optimized carbon submicron particles: graphene oxide and reduced graphene oxide. Chem Eng J 397:125398. https://doi.org/10.1016/j.cej.2020.125398
Kumar P, Rehab H, Hegde K et al (2020b) Physical and biological removal of Microcystin-LR and other water contaminants in a biofilter using Manganese Dioxide coated sand and Graphene sand composites. Sci Total Environ 703:135052. https://doi.org/10.1016/j.scitotenv.2019.135052
Kumarasinghe AR, Perera WPRT, Bandara J et al (2021) Multiple coated graphite oxide - sand composites for fluoride removal in water. J Environ Chem Eng 9:104962. https://doi.org/10.1016/j.jece.2020.104962
Kumari U, Mishra A, Siddiqi H, Meikap BC (2021) Effective defluoridation of industrial wastewater by using acid modified alumina in fixed-bed adsorption column: experimental and breakthrough curves analysis. J Clean Prod 279:123645. https://doi.org/10.1016/j.jclepro.2020.123645
Lin X, Huang Q, Qi G et al (2017) Estimation of fixed-bed column parameters and mathematical modeling of breakthrough behaviors for adsorption of levulinic acid from aqueous solution using SY-01 resin. Sep Purif Technol 174:222–231. https://doi.org/10.1016/j.seppur.2016.10.016
Maged A, Iqbal J, Kharbish S et al (2020) Tuning tetracycline removal from aqueous solution onto activated 2:1 layered clay mineral: characterization, sorption and mechanistic studies. J Hazard Mater 384:121320. https://doi.org/10.1016/J.JHAZMAT.2019.121320
Mahmoud MA (2021) Batch and column modelling of Cd (II) separation from aqueous phase using chitosan nitrogen foam carbon. Environ Nanotechnol Monit Manag 16:100494. https://doi.org/10.1016/j.enmm.2021.100494
Mahmoudi E, Ang WL, Ng CY et al (2019) Distinguishing characteristics and usability of graphene oxide based on different sources of graphite feedstock. J Colloid Interface Sci 542:429–440. https://doi.org/10.1016/J.JCIS.2019.02.023
Mahmoudi E, Azizkhani S, Mohammad AW et al (2020) Simultaneous removal of Congo red and cadmium(II) from aqueous solutions using graphene oxide–silica composite as a multifunctional adsorbent. J Environ Sci 98:151–160. https://doi.org/10.1016/J.JES.2020.05.013
Mohammad-Rezaei R, Jaymand M (2019) Graphene quantum dots coated on quartz sand as efficient and low-cost adsorbent for removal of Hg2+ and Pb2+ from aqueous solutions. Environ Prog Sustain Energy 38:S24–S31. https://doi.org/10.1002/ep.12911
Mondal J, Srivastava SK (2020) MnO2 nanoflowers and their reduced graphene oxide nanocomposites for electromagnetic interference shielding. ACS Appl Nano Mater 3:11048–11059. https://doi.org/10.1021/acsanm.0c02247
Nizam NUM, Hanafiah MM, Mahmoudi E et al (2022) Effective adsorptive removal of dyes and heavy metal using graphene oxide based pre-treated with NaOH / H2SO4 rubber seed shells synthetic graphite precursor: equilibrium isotherm, kinetics and thermodynamic studies. Sep Purif Technol 289:120730. https://doi.org/10.1016/J.SEPPUR.2022.120730
Nouri A, Chua SF, Mahmoudi E, Wibawa Sakti SC, Ang WL (2021) CHAPTER 5 - Polymer-based Nanocomposites for Environmental Remediation. In: Functional Hybrid Nanomaterials for Environmental Remediation (Book). Royal Society of Chemistry, pp 1–32. https://doi.org/10.1039/9781839165283-00101
Plata-Gryl M, Momotko M, Makowiec S, Boczkaj G (2022) Characterization of diatomaceous earth coated with nitrated asphaltenes as superior adsorbent for removal of VOCs from gas phase in fixed bed column. Chem Eng J 427:130653. https://doi.org/10.1016/j.cej.2021.130653
Prathibha C, Biswas A, Chunduri LAA et al (2020) Zr(IV) functionalized graphene oxide anchored sand as potential and economic adsorbent for fluoride removal from water. Diam Relat Mater 109:108081. https://doi.org/10.1016/j.diamond.2020.108081
Raizada P, Kumari J, Shandilya P et al (2017) Magnetically retrievable Bi2WO6/Fe3O4 immobilized on graphene sand composite for investigation of photocatalytic mineralization of oxytetracycline and ampicillin. Process Saf Environ Prot 106:104–116. https://doi.org/10.1016/j.psep.2016.12.012
Ranjan Rout D, Mohan Jena H (2021) Synthesis of novel reduced graphene oxide decorated β-cyclodextrin epichlorohydrin composite and its application for Cr(VI) removal: batch and fixed-bed studies. Sep Purif Technol 278:119630. https://doi.org/10.1016/J.SEPPUR.2021.119630
Rodrigues Sousa H, Santos Silva L, Abreu Sousa PA et al (2019) Evaluation of methylene blue removal by plasma activated palygorskites. J Mater Res Technol 8:5432–5442. https://doi.org/10.1016/J.JMRT.2019.09.011
Sharma R, Baik JH, Perera CJ, Strano MS (2010) Anomalously large reactivity of single graphene layers and edges toward electron transfer chemistries. Nano Lett 10:398–405. https://doi.org/10.1021/NL902741X
Singh P, Priya B, Shandilya P et al (2019) Photocatalytic mineralization of antibiotics using 60%WO3/BiOCl stacked to graphene sand composite and chitosan. Arab J Chem 12:4627–4645. https://doi.org/10.1016/j.arabjc.2016.08.005
Sonal S, Acharya S, Mishra BK (2022) Mesoporous carbon structure impregnated with 2D engineered zirconium: a sustainable adsorbent for the removal of dyes from the aqueous solution. J Environ Manage 314:115009. https://doi.org/10.1016/J.JENVMAN.2022.115009
Tee GT, Gok XY, Yong WF (2022) Adsorption of pollutants in wastewater via biosorbents, nanoparticles and magnetic biosorbents: a review. Environ Res 212:113248. https://doi.org/10.1016/J.ENVRES.2022.113248
Teow YH, Tajudin S’A, Ho KC, Mohammad AW (2020) Synthesis and characterization of graphene shell composite from oil palm frond juice for the treatment of dye-containing wastewater. J Water Process Eng 35:101185. https://doi.org/10.1016/j.jwpe.2020.101185
Tripathi AD, Yadav A, Jha A, Srivastava SK (2012) Utilizing of sugar refinery waste (cane molasses) for production of bio-plastic under submerged fermentation process. J Polym Environ 20:446–453. https://doi.org/10.1007/s10924-011-0394-1
Wang J, Li X, Tang L et al (2021a) Synthesis approaches to magnetic graphene oxide and its application in water treatment: a review. Water Air Soil Pollut 232:1–16. https://doi.org/10.1007/s11270-021-05281-2
Wang J, Liu X, Yang M et al (2021b) Removal of tetracycline using modified wheat straw from solution in batch and column modes. J Mol Liq 338:116698. https://doi.org/10.1016/j.molliq.2021.116698
Yanyan L, Kurniawan TA, Zhu M et al (2018) Removal of acetaminophen from synthetic wastewater in a fixed-bed column adsorption using low-cost coconut shell waste pretreated with NaOH, HNO3, ozone, and/or chitosan. J Environ Manage 226:365–376. https://doi.org/10.1016/J.JENVMAN.2018.08.032
Zhao C, Pei S, Ma J et al (2020) Influence of graphene oxide nanosheets on the cotransport of Cu-tetracycline multi-pollutants in saturated porous media. Environ Sci Pollut Res 27:10846–10856. https://doi.org/10.1007/S11356-020-07622-W
Zhou Q, Hu X (2017) Systemic stress and recovery patterns of rice roots in response to graphene oxide nanosheets. Environ Sci Technol 51:2022–2030. https://doi.org/10.1021/ACS.EST.6B05591/SUPPL_FILE/ES6B05591_SI_003.XLS
Funding
The work was funded by the ASEAN University Network/Southeast Asia Engineering Education Development Network (AUN/SEED-Net), Japan International Cooperation Agency (JICA).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Alireza Nouri and Ebrahim Mahmoudi. The first draft of the manuscript was written by Alireza Nouri and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nouri, A., Mahmoudi, E., Ang, W.L. et al. Sugar molasses as a sustainable precursor for the synthesis of graphene sand composite adsorbent for tetracycline and methylene blue removal. Environ Sci Pollut Res 30, 98817–98831 (2023). https://doi.org/10.1007/s11356-022-21996-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-21996-z