Abstract
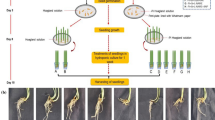
Aluminum (Al) toxicity in acidic soils is a major problem in rice crop production, especially in the acid sulfate soil (pH < 4.0). Selecting Al-tolerant varieties of rice with low toxicity is one of the most appropriate strategies to overcome this problem. In the present study, we investigated the Al content in different rice genotypes, IR64 (high yielding), RD35 (local acidic-tolerant), and Azucena (AZU, positive-check Al-tolerant), and their physiological and morphological adaptations under a wide range Al (10, 25, 50 mM [Al2(SO4)3]) treatments in the greenhouse conditions. Under 50-mM Al treatment, Al levels in the root tissues of rice seedlings cvs. AZU and IR64 were increased by 2.74- and 2.10-fold over control. Interestingly, Al contents in the roots of cv. RD35 were also exhibited by 2.04-fold over control. Similarly, Al contents in the leaves trend to increase in relation to a degree of Al treatments, leading to increase leaf temperature, chlorophyll degradation, limited CO2 assimilation, and negative effect on root traits under 50 mM Al were evidently observed. Therefore, leaf temperature was considered a sensitive parameter regulated by high concentration of Al (50 mM), leading to increase in crop water stress index (CWSI > 0.6) and decrease in stomata conductance. Net photosynthetic rate (Pn) and transpiration rate (E) in rice seedlings of cv. RD35 subjected to 50 mM Al were significantly dropped by 74.76% and 47.71% over the control, respectively, resulting in reduced growth performances in terms of root length (26.57% reduction) and shoot fresh weight (46.15% reduction). An enrichment of Al in the root tissues without toxicity in rice cv. AZU may further help in discovering the Al homeostasis. In summary, Al enrichment in rice genotypes grown under Al-treatments was evidently observed in the root, leading to the limited root growth, root length, and root dry weight, especially in cv. RD35. Al restriction in the root tissues of cv. AZU (Al-tolerant) may play a key role as defense mechanisms to avoid translocation to other organs and the stomata closure was an alternative key factor to limit H2O transpiration.




Similar content being viewed by others
Data availability
Data are available under request to the corresponding author.
Abbreviations
- Al:
-
aluminum
- AZU:
-
Azucena
- CRD:
-
completely randomized design
- CWSI:
-
crop water stress index
- E :
-
transpiration rate
- EC:
-
electroconductivity
- F v/F m :
-
maximum quantum yield of PSII
- g s :
-
stomatal conductance
- ICP–OES:
-
inductively coupled plasma–optical emission spectroscope
- IRGA:
-
infrared gas analyzer
- ΦPSII :
-
photon yield of PSII
- P n :
-
net photosynthetic rate
- PPFD:
-
photosynthetic photon flux density
- ROS:
-
reactive oxygen species
- T c :
-
canopy leaf temperature
- T dry :
-
temperature dry reference
- T wet :
-
temperature wet reference
References
Ali S, Zeng F, Qiu L, Zhang G (2011) The effect of chromium and aluminum on growth, root morphology, photosynthetic parameters and transpiration of the two barley cultivars. Biol Plant 55:291–296. https://doi.org/10.1007/s10535-011-0041-7
Alvim MN, Ramos FT, Oliveira DC, Isaias RMS, Franca MGC (2012) Aluminum localization and toxicity symptoms related to root growth inhibition in rice (Oryza sativa L.) seedlings. J Biosci 37:1079–1088
Arunakumara KKIU, Walpola BC, Yoon MH (2013) Aluminum toxicity and tolerance mechanism in cereals and legumes—a review. J Korean Soc Appl Biol Chem 56:1–9. https://doi.org/10.1007/s13765-012-2314-z
Awasthi JP, Saha B, Panigrahi J, Yanase E, Koyama H, Panda SK (2019) Redox balance, metabolic fingerprint and physiological characterization in contrasting North East Indian rice for aluminum stress tolerance. Sci Rep 9:1–21. https://doi.org/10.1038/s41598-019-45158-3
Bhoomika K, Pyngrope S, Dubey RS (2013) Differential responses of antioxidant enzymes to aluminum toxicity in two rice (Oryza sativa L.) cultivars with marked presence and elevated activity of Fe-SOD and enhanced activities of Mn-SOD and catalase in aluminum tolerant cultivar. Plant Growth Regul 71:235–252. https://doi.org/10.1007/s10725-013-9824-5
Bojórquez-Quintal E, Escalante-Magaña C, Echevarría-Machado I, Martínez-Estévez M (2017) Aluminum, a friend or foe of higher plants in acid soils. Front Plant Sci 8:1767. https://doi.org/10.3389/fpls.2017.01767
Cao Y, Lou Y, Han Y, Shi J, Wang Y, Wang W, Ming F (2011) Al toxicity leads to enhanced cell division and changed photosynthesis in Oryza rufipogon L. Mol Biol Rep 38:4839–4846. https://doi.org/10.1007/s11033-010-0618-9
Cha-um S, Supaibulwatana K, Kirdmanee C (2006) Water relation, photosynthetic ability and growth of Thai jasmine rice (Oryza sativa L. ssp. indica cv. KDML 105) to salt stress by application of exogenous glycinebetaine and choline. J Agron Crop Sci 192:25–36. https://doi.org/10.1111/j.1439-037X.2006.00186.x
Chen RF, Shen RF (2008) Root phosphate exudation and pH shift in the rhizosphere are not responsible for aluminum resistance in rice. Acta Physiol Plant 30:817–824. https://doi.org/10.1007/s11738-008-0186-y
de Macêdo CE, Jan VV (2008) Effect of aluminum stress on mineral nutrition in rice cultivars differing in aluminum sensitivity. Rev Bras Eng Agr Amb 12:363–369. https://doi.org/10.1590/S1415-43662008000400005
de Macêdo CEC, Jan VVS, Kinet JM, Lutts S (2009) Effects of aluminium on root growth and apical root cells in rice (Oryza sativa L.) cultivars. Reliability of screening tests to detect Al resistance at the seedling stage. Acta Physiol Plant 31:1255–1262. https://doi.org/10.1007/s11738-009-0362-8
Famoso AN, Clark RT, Shaff JE, Craft E, McCouch SR, Kochian LV (2010) Development of a novel aluminum tolerance phenotyping platform used for comparisons of cereal aluminum tolerance and investigations into rice aluminum tolerance mechanisms. Plant Physiol 153:1678–1691. https://doi.org/10.1104/pp.110.156794
Fonseca Júnior EM, Cambraia J, Ribeiro C, Oliva MA, Oliveira JA, da Matta FM (2014) The effects of aluminium on the photosynthetic apparatus of two rice cultivars. Exp Agric 50:343–352. https://doi.org/10.1017/S0014479713000471
Frankowski M (2016) Aluminum uptake and migration from the soil compartment into Betula pendula for two different environments: a polluted and environmentally protected area of Poland. Environ Sci Pollut Res 23:1398–1407. https://doi.org/10.1007/s11356-015-5367-9
Guo TR, Yao PC, Zhang ZD, Wang JJ, Mei WANG (2012) Involvement of antioxidative defense system in rice seedlings exposed to aluminum toxicity and phosphorus deficiency. Rice Sci 19:207–212. https://doi.org/10.1016/S1672-6308(12)60042-0
Hidayatun N, Diaz MGQ, Ismail AM (2018) Exploring aluminum tolerance at seedling stage in rice (Oryza sativa Linn.) by using modified Magnavaca nutrient solution. Buletin Plasma Nutfah 23:81–90. https://doi.org/10.21082/blpn.v23n2.2017.p81-90
Hou N, You J, Pang J, Xu M, Chen G, Yang Z (2010) The accumulation and transport of abscisic acid in soybean (Glycine max L.) under aluminum stress. Plant Soil 330:127–137. https://doi.org/10.1007/s11104-009-0184-x
Huang CF, Yamaji N, Chen Z, Ma JF (2012) A tonoplast-localized half-size ABC transporter is required for internal detoxification of aluminum in rice. Plant J 69:857–867. https://doi.org/10.1111/j.1365-313X.2011.04837.x
Hussain F, Bronson KF, Peng S (2000) Use of chlorophyll meter sufficiency indices for nitrogen management of irrigated rice in Asia. Agron J 92:875–879. https://doi.org/10.2134/agronj2000.925875x
Inostroza-Blancheteau C, Rengel Z, Alberdi M, de la Luz Mora M, Aquea F, Arce-Johnson P, Reyes-Díaz M (2012) Molecular and physiological strategies to increase aluminum resistance in plants. Mol Biol Rep 39:2069–2079. https://doi.org/10.1007/s11033-011-0954-4
IRRI (2002) Standard evaluation system for rice, 4th edn. International Rice Research Institute, Manila
Jaiswal SK, Naamala J, Dakora FD (2018) Nature and mechanisms of aluminium toxicity, tolerance and amelioration in symbiotic legumes and rhizobia. Biol Fertil Soils 54:309–318. https://doi.org/10.1007/s00374-018-1262-0
Jin M, Liu X, Zhang B (2017) Evaluating heavy-metal stress levels in rice using a theoretical model of canopy-air temperature and leaf area index based on remote sensing. IEEE J Sel Top Appl Earth Obs Remote Sens 10:3232–3242. https://doi.org/10.1109/JSTARS.2017.2669204
Jones HG, Schofield P (2008) Thermal and other remote sensing of plant stress. Gen Appl Plant Physiol 34:19–32
Kang DJ, Seo YJ, Lee BK, Vijarnsorn P, Ishii R (2010) Identification and crop performance of acid sulfate soil-tolerant rice varieties. J Crop Sci Biotechnol 13:75–81. https://doi.org/10.1007/s12892-010-0002-2
Khush GS (2005) What it will take to feed 5.0 billion rice consumers in 2030. Plant Mol Biol 59:1–6. https://doi.org/10.1007/s11103-005-2159-5
Kochian LV, Hoekenga OA, Pineros MA (2004) How do crop plants tolerate acid soils? Mechanisms of aluminum tolerance and phosphorous efficiency. Annu Rev Plant Biol 55:459–493. https://doi.org/10.1146/annurev.arplant.55.031903.141655
Leinonen I, Grant OM, Tagliavia CPP, Chaves MM, Jones HG (2006) Estimating stomatal conductance with thermal imagery. Plant Cell Environ 29:1508–1518. https://doi.org/10.1111/j.1365-3040.2006.01528.x
Leyman B, Geelen D, Quintero FJ, Blatt MR (1999) A tobacco syntaxin with a role in hormonal control of guard cell ion channels. Science 283:537–540. https://doi.org/10.1126/science.283.5401.537
Loggini B, Scartazza A, Brugnoli E, Navari-Izzo F (1999) Antioxidant defense system, pigment composition, and photosynthetic efficiency in two wheat cultivars subjected to drought. Plant Physiol 119:1091–1099. https://doi.org/10.1104/pp.119.3.1091
Ma JF, Ryan PR, Delhaize E (2001) Aluminium tolerance in plants and the complexing role of organic acids. Trends Plant Sci 6:273–278. https://doi.org/10.1016/S1360-1385(01)01961-6
Ma JF, Shen R, Zhao Z, Wissuwa M, Takeuchi Y, Ebitani T, Yano M (2002) Response of rice to Al stress and identification of quantitative trait loci for Al tolerance. Plant Cell Physiol 43:652–659. https://doi.org/10.1093/pcp/pcf081
Mao C, Yi K, Yang L, Zheng B, Wu Y, Liu F, Wu P (2004) Identification of aluminum-regulated genes by cDNA-AFLP in rice (Oryza sativa L.): aluminium-regulated genes for the metabolism of cell wall components. J Exp Bot 55:137–143. https://doi.org/10.1093/jxb/erh030
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence – a practical guide. J Exp Bot 51:659–668. https://doi.org/10.1093/jexbot/51.345.659
Merlot S, Mustilli AC, Genty B, North H, Lefebvre V, Sotta B, Vavasseur A, Giraudat J (2002) Use of infrared thermal imaging to isolate Arabidopsis mutants defective in stomatal regulation. Plant J 30:601–609. https://doi.org/10.1046/j.1365-313X.2002.01322.x
Muhammad N, Cai S, Shah JM, Zhang G (2016) The combined treatment of Mn and Al alleviates the toxicity of Al or Mn stress alone in barley. Acta Physiol Plant 38:277. https://doi.org/10.1007/s11738-016-2296-2
Munde NA, Jadhao KR, Samal KC, Pradhan SK, Rout GR (2016) Allele mining in Indica rice (Oryza sativa L.) for ATP binding cassette (ABC) transporter gene family for aluminum tolerance. Indian J Plant Physiol 21:161–170. https://doi.org/10.1007/s40502-016-0217-4
Pan JW, Zhu MY, Chen H (2001) Aluminum-induced cell death in root-tip cells of barley. Environ Exp Bot 46:71–79. https://doi.org/10.1016/S0098-8472(01)00083-1
Panda D, Sahoo RS, Behera PK, Barik J, Nayak JK (2020) Leaf photochemical activity and antioxidant protection in selected hill rice genotypes of Koraput, India in relation to aluminum (Al3+) stress. J Stress Physiol Biochem 16:13–21
Pandey P, Srivastava RK, Dubey RS (2014) Water deficit and aluminum tolerance are associated with a high antioxidative enzyme capacity in Indica rice seedlings. Protoplasma 251:147–160. https://doi.org/10.1007/s00709-013-0533-8
Pipatsitee P, Eiumnoh A, Praseartkul P, Taota K, Kongpugdee S, Sakulleerungroj K, Cha-um S (2018) Application of infrared thermography to assess cassava physiology under water deficit condition. Plant Prod Sci 21:398–406. https://doi.org/10.1080/1343943X.2018.1530943
Poirier-Pocovi M, Volder A, Bailey BN (2020) Modeling of reference temperatures for calculating crop water stress indices from infrared thermography. Agric Water Manag 233:106070. https://doi.org/10.1016/j.agwat.2020.106070
Ramachandran V, Patel DU, Kumar SC (2004) Influence of aluminium on mineral nutrients of rice seedlings grown in solution culture. Indian J Plant Physiol 9:348–353
Ribeiro C, Cambraia J, Peixoto PHP, Fonseca Júnior ÉMD (2012) Antioxidant system response induced by aluminum in two rice cultivars. Braz J Plant Physiol 24:107–116. https://doi.org/10.1590/S1677-04202012000200004
Roselló M, Poschenrieder C, Gunse B, Barceló J, Llugany M (2015) Differential activation of genes related to aluminium tolerance in two contrasting rice cultivars. J Inorg Biochem 152:160–166. https://doi.org/10.1016/j.jinorgbio.2015.08.021
Roy B, Bhadra S (2014) Effects of toxic levels of aluminium on seedling parameters of rice under hydroponic culture. Rice Sci 21:217–223. https://doi.org/10.1016/S1672-6308(13)60182-1
Roy B, Mandal AB (2005) Towards development of Al-toxicity tolerant lines in indica rice by exploiting somaclonal variation. Euphytica 145:221–227. https://doi.org/10.1007/s10681-005-0055-3
Sade H, Meriga B, Surapu V, Gadi J, Sunita MSL, Suravajhala P, Kishor PK (2016) Toxicity and tolerance of aluminum in plants: tailoring plants to suit to acid soils. Biometals 29:187–210. https://doi.org/10.1007/s10534-016-9910-z
Samad R, Rashid P, Karmoker JL (2020) Effects of aluminium toxicity on some biochemical components of rice (Oryza sativa L.). Dhaka Univ J Biol Sci 29:125–132. https://doi.org/10.3329/dujbs.v29i1.46538
Sharma P, Dubey RS (2007) Involvement of oxidative stress and role of antioxidative defense system in growing rice seedlings exposed to toxic concentrations of aluminum. Plant Cell Rep 26:2027–2038. https://doi.org/10.1007/s00299-007-0416-6
Sivaguru M, Pike S, Gassmann W, Baskin TI (2003) Aluminum rapidly depolymerizes cortical microtubules and depolarizes the plasma membrane: evidence that these responses are mediated by a glutamate receptor. Plant Cell Physiol 44:667–675. https://doi.org/10.1093/pcp/pcg094
Sun P, Tian QY, Chen J, Zhang WH (2010) Aluminium-induced inhibition of root elongation in Arabidopsis is mediated by ethylene and auxin. J Exp Bot 61:347–356. https://doi.org/10.1093/jxb/erp306
Tarantino TB, Barbosa IS, Lima DDC, Pereira MDG, Teixeira LS, Korn MGA (2017) Microwave-assisted digestion using diluted nitric acid for multi-element determination in rice by ICP OES and ICP-MS. Food Anal Methods 10:1007–1015. https://doi.org/10.1007/s12161-016-0658-4
Thakur AK, Singh KJ (2012) Leaf temperature as thermal bio-indicator of heavy metal pollutants. J Agric Sci Technol A 2(1A):131–137
Vardar F, Ünal M (2007) Aluminum toxicity and resistance in higher plants. Adv Mol Biol 1:1–12
Violante A, Cozzolino V, Perelomov L, Caporale AG, Pigna M (2010) Mobility and bioavailability of heavy metals and metalloids in soil environments. J Soil Sci Plant Nutr 10:268–292. https://doi.org/10.4067/S0718-95162010000100005
Wang JW, Kao CH (2007) Protective effect of ascorbic acid and glutathione on AlCl3-inhibited growth of rice roots. Biol Plant 51:493–500. https://doi.org/10.1007/s10535-007-0104-y
Wang S, Ren X, Huang B, Wang G, Zhou P, An Y (2016) Aluminium-induced reduction of plant growth in alfalfa (Medicago sativa) is mediated by interrupting auxin transport and accumulation in roots. Sci Rep 6:1–13. https://doi.org/10.1038/srep30079
Wiriya-Alongkorn W, Spreer W, Ongprasert S, Spohrer K, Pankasemsuk T, Muller J (2013) Detecting drought stress in longan tree using thermal imaging. Maejo Int J Sci Technol 7:166–180
Xia J, Yamaji N, Ma JF (2011) Further characterization of an aluminum influx transporter in rice. Plant Signal Behav 6:160–163. https://doi.org/10.4161/psb.6.1.14319
Xu FJ, Jin CW, Liu WJ, Zhang YS, Lin XY (2011) Pretreatment with H2O2 alleviates aluminum-induced oxidative stress in wheat seedlings. J Integr Plant Biol 53:44–53. https://doi.org/10.1111/j.1744-7909.2010.01008.x
Xu J, Lv Y, Liu X, Dalson T, Yang S, Wu J (2016) Diagnosing crop water stress of rice using infra-red thermal imager under water deficit condition. Int J Agric Biol 18:565–572. https://doi.org/10.17957/IJAB/15.0125
Yang JL, Li YY, Zhang YJ, Zhang SS, Wu YR, Wu P, Zheng SJ (2008) Cell wall polysaccharides are specifically involved in the exclusion of aluminum from the rice root apex. Plant Physiol 146:602–611. https://doi.org/10.1104/pp.107.111989
Yokosho K, Yamaji N, Ma JF (2011) An Al-inducible MATE gene is involved in external detoxification of Al in rice. Plant J 68:1061–1069. https://doi.org/10.1111/j.1365-313X.2011.04757.x
Zeng QL, Chen RF, Zhao XQ, Wang HY, Shen RF (2011) Aluminium uptake and accumulation in the hyperaccumulator Camellia oleifera Abel. Pedosphere 21:358–364. https://doi.org/10.1016/S1002-0160(11)60136-7
Zhang J, Wei J, Li D, Kong X, Rengel Z, Chen L, Yang Y, Cui X, Chen Q (2017a) The role of the plasma membrane H+-ATPase in plant responses to aluminum toxicity. Front Plant Sci 8:1757. https://doi.org/10.3389/fpls.2017.01757
Zhang B, Liu X, Liu M, Wang D (2017b) Thermal infrared imaging of the variability of canopy-air temperature difference distribution for heavy metal stress levels discrimination in rice. J Appl Remote Sens 11:026036. https://doi.org/10.1117/1.JRS.11.026036
Zhu CQ, Zhang JH, Sun LM, Zhu LF, Abliz B, Hu WJ, Zhong C, Bai ZG, Sajid H, Cao XC, Jin QY (2018) Hydrogen sulfide alleviates aluminum toxicity via decreasing apoplast and symplast Al contents in rice. Front Plant Sci 9:294. https://doi.org/10.3389/fpls.2018.00294
Zhu CQ, Hu WJ, Cao XC, Zhu LF, Bai ZG, Huang J, Liang QD, Jin QY, Zhang JH (2020) Role of salicylic acid in alleviating the inhibition of root elongation by suppressing ethylene emission in rice under Al toxicity conditions. Plant Growth Regul 90:475–487. https://doi.org/10.1007/s10725-019-00554-7
Zioła-Frankowska A, Frankowski M (2018) Speciation analysis of aluminium in plant parts of Betula pendula and in soil. J Environ Sci 65:153–161. https://doi.org/10.1016/j.jes.2017.03.021
Acknowledgements
The authors would like to sincerely thank National Science and Technology Development Agency (NSTDA) for funding support (Grant number P-18-51456). MP is thankful to Thailand Graduate Institute of Science and Technology (TGIST) for partial scholarship.
Funding
This work was supported by the National Science and Technology Development Agency (NSTDA) for funding support (Grant Number P-18-51456) and partially support for the Graduate Scholarship for Suwanna Phukunkamkaew by the Thailand Graduate Institute of Science and Technology (TGIST).
Author information
Authors and Affiliations
Contributions
Conceptualization: Suriyan Cha-um, Sarunyaporn Maksup and Rujira Tisarum. Formal analysis and investigation: Suwanna Phukunkamkaew, Piyanan Pipatsitee, Thapanee Samphumphuang. Writing and original draft preparation: Suwanna Phukunkamkaew and Rujira Tisarum. Writing and review and editing: Suriyan Cha-um and Sarunyaporn Maksup. Funding acquisition: Suriyan Cha-um.
Corresponding author
Ethics declarations
Ethical approval
The authors whose names appear on the submission approve the ethical responsibilities of authors of Environmental Science and Pollution Research.
Consent to participate
The authors voluntarily agree to participate in this research study.
Consent for publication
Consent to publish all authors whose names appear on the submission (1) made substantial contributions to the conception or design of the work or to the acquisition, analysis or interpretation of the data, (2) drafted the work or revised it critically for important intellectual content, (3) approved the version to be published, and (4) agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Competing interests
The authors declare that they have no completing interests.
Additional information
Responsible Editor: Gangrong Shi
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PPTX 997 kb).
Rights and permissions
About this article
Cite this article
Phukunkamkaew, S., Tisarum, R., Pipatsitee, P. et al. Morpho-physiological responses of indica rice (Oryza sativa sub. indica) to aluminum toxicity at seedling stage. Environ Sci Pollut Res 28, 29321–29331 (2021). https://doi.org/10.1007/s11356-021-12804-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-12804-1