Abstract
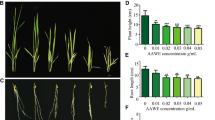
Oryza rufipogon L. (O. rufipogon) or a common wild rice, showed considerable aluminum (Al) tolerance. In this study, we examined the physiologic and genetic response of wild rice short term and long term to Al toxicity, respectively. In the short term study, morin staining, DAPI staining and aniline blue staining were used to detect Al distribution, cell division and callose production in the roots of O. rufipogon. The results indicated cell division could be enhanced by Al within low concentration range. In the long term study, we chose Oryza sativa L (O. sativa) (the close sib of O. rufipogon) as a reference. It showed that O. rufipogon grew better than O. sativa when treated with Al of 1.4 mmol/l concentration and also experienced a short period of root growth stimulation. This study gave some basic data to explain the mechanisms Oryza rufipogon L. developed to deal with Al and lay a good foundation to further study. SSH (suppression subtractive hybridization) proved that transcripts of the small subunit of Rubisco and a Photosystem I P700 apoprotein were enhanced under long term Al treatment in wild rice. Further investigation via the assessment of the content of chlorophyll a, b indicated that the content of chlorophyll a, b in the leaves of O. rufipogon generally rose after Al treatment for 15 days. This indicated that intake of Al can affect photosynthesis of plant.







Similar content being viewed by others
Abbreviations
- RRE:
-
Relative root elongation
References
Bot AJ, Nachtergaele FO, Young A (2000) Land resource potential and constraints at regional and country levels. In Food and Agricultural Organization of the United Nations, Rome
Degenhardt J, Larsen PB, Howell SH, Kochian LV (1998) Aluminum resistance in the Arabidopsis mutant alr-104 is caused by an aluminum-induced increase in rhizosphere pH. Plant Physiol 117:19–27
Delhaize E, Craig S, Beaton CD, Bennet RJ, Jagadish VC, Randall PJ (1993) Aluminum tolerance in wheat (Triticum aestivum L.) (I. uptake and distribution of aluminum in root apices). Plant Physiol 103:685–693
Delhaize E, Ryan PR (1995) Aluminum toxicity and tolerance in plants. Plant Physiol 107:315–321
Eticha D, Stass A, Horst WJ (2005) Localization of aluminum in the maize root apex: can morin detect cell wall-bound aluminum. J Exp Bot 56:1351–1357
Hoekenga OA, Vision TJ, Shaff JE, Monforte AJ, Lee GP, Howell SH, Kochian LV (2003) Identification and characterization of aluminum tolerance loci in Arabidopsis (Landsberg erecta x Columbia) by quantitative trait locus mapping. A physiologically simple but genetically complex trait. Plant Physiol 132:936–948
Ishikawa S, Wagatsuma T, Sasaki R, Ofei-Manu P (2000) Comparison of the amount of citric and malic acids in Al media of seven plant species and two cultivars each in five plant species. Soil Sci Plant Nutr 46:751
Kollmeier M, Felle HH, Horst WJ (2000) Genotypical differences in aluminum resistance of maize are expressed in the distal part of the transition zone is reduced basipetal auxin flow involved in inhibition of root elongation by aluminum. Plant Physiol 122:945–956
Koyama H, Takita E, Kawamura A, Hara T, Shibata D (1999) Over expression of mitochondrial citrate synthase gene improves the growth of carrot cells in Al-phosphate medium. Plant Cell Physiol 40:482–488
Larsen PB, Tai CY, Kochian LV, Howell SH (1996) Arabidopsis mutants with increased sensitivity to aluminum. Plant Physiol 110:743–751
Ma JF (2000) Role of organic acids in detoxification of aluminum in higher plants. Plant Cell Physiol 41:383–390
Ma JF, Hiradate S, Matsumoto H (1998) High aluminum resistance in buckwheat Ii. Oxalic acid detoxifies aluminum internally. Plant Physiol 117:753–759
Ma JF, Shen R, Zhao Z, Wissuwa M, Takeuchi Y, Ebitani T, Yano M (2002) Response of rice to Al stress and identification of quantitative trait loci for Al tolerance. Plant Cell Physiol 43:652–659
Matsumoto H (2000) Cell biology of aluminum toxicity and tolerance in higher plants. Int Rev Cytol 200:1–46
Miyasaka SC, Buta JG, Howell RK, Foy CD (1991) Mechanism of aluminum tolerance in Snapbeans: root exudation of citric acid. Plant Physiol 96:737–743
Li RQ, Wang JB (2002) The cellular and physiological bases of metal ions tolerance in plants. In: Plant Cytol Physiol Adversity. WuHan University Press, pp 285–319
Raman H, Zhang K, Cakir M, Appels R, Garvin DF, Maron LG, Kochian LV, Moroni JS, Raman R, Imtiaz M, Drake-Brockman F, Waters I, Martin P, Sasaki T, Yamamoto Y, Matsumoto H, Hebb DM, Delhaize E, Ryan PR (2005) Molecular characterization and mapping of ALMT1 the aluminum-tolerance gene of bread wheat (Triticum aestivum L.). Genome 48:781–791
Ryan PR, Skerrett M, Findlay GP, Delhaize E, Tyerman SD (1997) Aluminum activates an anion channel in the apical cells of wheat roots. Proc Natl Acad Sci USA 94:6547–6552
Yoshida S (1976) Laboratory manual for physiological studies of rice, 3rd ed. The Philippines: International Rice Research Institute, Manila, pp 17–19, 27–33.
Silva IR, Smyth TJ, Moxley DF, Carter TE, Allen NS, Rufty TW (2000) Aluminum accumulation at nuclei of cells in the root tip. Fluorescence detection using lumogallion and confocal laser scanning microscopy. Plant Physiol 123:543–552
Silva IR, Smyth TJ, Moxley DF, Carter TE, Allen NS, Rufty TW (2001) Differential aluminum tolerance in soybean: an evaluation of the role of organic acids. Physiol Plant 112:200–210
Sivaguru M, Baluska F, Volkmann D, Felle HH, Horst WJ (1999) Impacts of aluminum on the cytoskeleton of the maize root apex. Short-term effects on the distal part of the transition zone. Plant Physiol 119:1073–1082
Sivaguru M, Ezaki B, He ZH, Tong H, Osawa H, Baluska F, Volkmann D, Matsumoto H (2003) Aluminum-induced gene expression and protein localization of a cell wall-associated receptor kinase in Arabidopsis. Plant Physiol 132:2256–2266
Smith FW, Ealing PM, Dong B, Delhaize E (1997) The cloning of two Arabidopsis genes belonging to a phosphate transporter family. Plant J 11:83–92
Wenzl P, Patino GM, Chaves AL, Mayer JE, Rao IM (2001) The high level of aluminum resistance in signalgrass is not associated with known mechanisms of external aluminum detoxification in root apices. Plant Physiol 125:1473–1484
Yamaguchi M, Sasaki T, Sivaguru M, Yamamoto Y, Osawa H, Ahn SJ, Matsumoto H (2005) Evidence for the plasma membrane localization of Al-activated malate transporter (ALMT1). Plant Cell Physiol 46:812–816
Yang JL, Li YY, Zhang YJ, Zhang SS, Wu YR, Wu P, Zheng SJ (2008) Cell wall polysaccharides are specifically involved in the exclusion of aluminum from the rice root apex. Plant Physiol 146:602–611
Zhao FG, Li H, Luo QY (2004). Aluminum toxicity and mechanism of aluminum tolerance in plants, Plant Physiol Ecol Adversity 154-174
Zheng JS, Ma JF, Matsumoto H (1998) High aluminum resistance in buckwheat. I. Al-induced specific secretion of oxalic acid from root tips. Plant Physiol 117:745–751
Zheng K, Pan JW, Ye L, Fu Y, Peng HZ, Wan BY, Gu Q, Bian HW, Han N, Wang JH, Kang B, Pan JH, Shao HH, Wang WZ, Zhu MY (2007) Programmed cell death-involved aluminum toxicity in yeast alleviated by antiapoptotic members with decreased calcium signals. Plant Physiol 143:38–49
Acknowledgments
This study was supported by the National High Technology Research and Development Program of China (2008AA10Z116), the Ministry of Agriculture of China (2008ZX08009-001) and the Youth Science and Technology Phosphor Foundation of Shanghai (08QH14003), and partly by the Science and Technology Foundation of Shanghai (083391910400).
Author information
Authors and Affiliations
Corresponding author
Additional information
Yingping Cao and Yuxia Lou contributed equally to this work.
Rights and permissions
About this article
Cite this article
Cao, Y., Lou, Y., Han, Y. et al. Al toxicity leads to enhanced cell division and changed photosynthesis in Oryza rufipogon L.. Mol Biol Rep 38, 4839–4846 (2011). https://doi.org/10.1007/s11033-010-0618-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-010-0618-9