Abstract
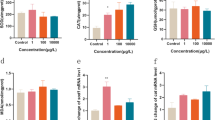
Fish are exposed to a wide variety of environmental stressors, such as chemicals and acute changes in temperature. Oxytetracycline (OTC) has been used as an antibiotic for many kinds of bacterial diseases in cultured fish, but excessive doses of OTC are known to cause side effects in fish and can have negative effects on their environment. In the present study, we examined stress-related biomarker expression in response to excessive doses of dietary OTC in coho salmon (Oncorhynchus kisutch). Fish received OTC (100 mg/kg body weight/day) orally for 2 weeks. The percentage of liver to body weight (hepatosomatic index; HSI) and plasma biochemical parameter, alanine aminotransferase (ALT) activity, of the group fed a diet containing OTC were observed to be significantly higher than those of the control group. The total glutathione (tGSH) levels in the liver of OTC-fed fish were four fold higher than those in control fish and double the control levels in muscle and stomach. Plasma tGSH levels in OTC-fed fish were also higher than those in control fish. Expression levels of heat shock protein 70 in the liver, muscle, and stomach decreased by OTC administration. Accordingly, OTC-induced stress might increase the metabolic turnover of GSH due to consumption by scavenging oxidants generated by stress. These results concerning the changing patterns of stress-related biomarkers indicate that excessive doses of OTC fed to coho salmon induce oxidative stress, which might enhance oxidation in the body and result in damage to tissues, especially in the liver. The present results also suggest that tissue-specific damage caused by OTC might already exist in fish.




Similar content being viewed by others
References
Adams SM (2005) Assessing cause and effect of multiple stressors on marine systems. Mar Pollut Bull 51:649–657
Allen RG, Tresini M (2000) Oxidative stress and gene regulation. Free Radic Biol Med 28:463–499
Ameur WB, De Lapuente J, Megdiche YE, Barhoumi B, Trabelsi S, Camps L, Serret J, Ramos-Lopez D, Gonzalez-Linares J, Driss MR, Borras M (2012) Oxidative stress, genotoxicity and histopathology biomarker responses in mullet (Mugil cephalus) and sea bass (Dicentrarchus labrax) liver from Bizerte Lagoon (Tunisia). Mar Pollut Bull 64:241–251
Arrigo A-P (1999) Gene expression and the thiol redox state. Free Radic Biol Med 27:936–944
Barton BA, Iwama GK (1991) Physiological change in fish from stress in aquaculture with emphasis on the response and effects of corticosteroids. Annu Rev Fish Dis 1:3–26
Basu N, Nakano T, Grau EG, Iwama GK (2001) The effects of cortisol on heat shock protein 70 levels in two fish species. Gen Comp Endocrinol 124:97–105
Basu N, Todgham AE, Ackerman PA, Bibeau MR, Nakano K, Shulte PM, Iwama GK (2002) Heat shock protein genes and their functional significance in fish. Gene 295:173–183
Bruno DW (1989) An investigation into oxytetracycline residues in Atlantic salmon, Salmo salar L. J Fish Dis 12:77–86
Bureau of Food Safety and Consumer Affairs, Ministry of Agriculture, Forestry and Fisheries of Japan (2015): the guideline for usage of drugs for fisheries. Tokyo, pp. 8–12
Burridge L, Weis JS, Cabello F, Pizarro J, Bostick K (2010) Chemical use in aquaculture: a review of current practices and possible environmental effects. Aquaculture 306:7–23
Donaldson EM, Fagerlund UHM, Higgs DA, McBride JR (1979) Hormonal enhancement of growth. In: Hoar WS, Randall DJ, Brett JR (eds) Bioenergetics and growth. Fish physiology 8. Academic, New York, pp 455–597
Droge W (2002) Free radicals in the physiological control of cell function. Physiol Rev 82:47–95
Elia AC, Ciccotelli V, Pacini N, Dorr AJM, Gili M, Natali M, Gasco L, Prearo M, Abete MC (2014) Transferability of oxytetracycline (OTC) from feed to carp muscle and evaluation of the antibiotic effects on antioxidant systems in liver and kidney. Fish Physiol Biochem 40:1055–1068
Feder ME, Hofmann GE (1999) Heat-shock proteins, molecular chaperones, and the stress response. Annu Rev Physiol 61:243–282
Forbes VE, Palmqvist A, Bach L (2006) The use and misuse of biomarkers in ecotoxicology. Environ Toxicol Chem 25:272–280
Griffiths HR, Dias IHK, Willetts RS, Devitt A (2014) Redox regulation of protein damage in plasma. Redox Biol 2:430–435
Guardiola FA, Cerezuela R, Meseguer J, Esteban MA (2012) Modulation of immune parameters and expression if genes of gilthead seabream (Sparus aurata L.) by dietary administration of oxytetracycline. Aquaculture 334–337:51–57
Hagger JA, Jones MB, Lowe D, Leonard DRP, Owen R, Galloway TS (2008) Application of biomarkers for improving risk assessments of chemicals under the Water Framework Directive: a case study. Mar Pollut Bull 56:1111–1118
He X, Nie X, Yang Y, Liu X, Pan D, Cheng Z, Liang X (2012) Multi-biomarker responses in fishes from two typical marine aquaculture regions of South China. Mar Pollut Bull 64:2317–2324
Ho E, Galougahi KK, Liu C-C, Bhindi R, Figtree GA (2013) Biological markers of oxidative stress: applications to cardiovascular research and practice. Redox Biol 1:483–491
Hori TSF, Avilez IM, Iwama GK, Johnson SC, Moraes G, Afonso LOB (2008) Impairment of the stress response in matrinxa juveniles (Brycon amazonicus) exposed to low concentrations of phenol. Comp Biochem Physiol 147C:416–423
Ikeda Y, Ozaki H, Sezaki K (1986) Chemical composition of plasma, blood atlas of fishes. Midori Shobo, Tokyo, pp 304–323
Iwama GK, Thomas PT, Forsyth RB, Vijayan MM (1998) Heat shock protein expression in fish. Rev Fish Biol Fish 8:35–56
Iwama GK, Afonso LOB, Todgham AE, Ackerman PA, Nakano K (2004) Are hsps suitable for indicating stressed states in fish? J Exp Biol 207:15–19
Iwama GK, Afonso LOB, Vijayan MM (2006) Stress in fishes. In: Evans DH, Claiborne JB (eds) The physiology of fishes (3rd Ed.). CRC Press, Boca Raton, pp 319–342
Ji LL (1995) Oxidative stress during exercise: implication of antioxidant nutrients. Free Radic Biol Med 18:1079–1086
Kameda M, Nakano T, Yamaguchi T, Sato M, Afonso LOB, Iwama GK, Devlin RH (2008) Effects of heat shock on growth hormone receptor expression in coho salmon. Proc 5th World Fish Cong, Yokohama, Oct. 20–25, 3f-16
Lushchak VI (2011) Environmentally induced oxidative stress in aquatic animals. Aquat Toxicol 101:13–30
Malvisi J, Della Rocca G, Anfossi P, Giorgetti G (1996) Tissue distribution and residue depletion of oxytetracycline in sea bream (Sparus aurata) and sea bass (Dicentrarchus labrax) after oral administration. Aquaculture 147:159–168
Mcdonald DG, Milligan CL (1992) Chemical properties of the blood. In: Hoar WS, Randall DJ, Farrell AP (eds) The cardiovascular system. Fish physiology 12B. Academic, New York, pp 55–133
Moreland JJ, Arrieta DE, Washburn BS (2000) Effects of estrogen on the stress response in CuSO4-exposed PLHC-1 cells. Mar Environ Res 50:509–512
Moriyama S, Ayson FG, Kawauchi H (2000) Growth regulation by insulin-like growth factor-I in fish. Biosci Biotech Biochem 64:1553–1562
Mujahid A, Yoshiki Y, Akiba Y, Toyomizu M (2005) Superoxide radical production in chicken skeletal muscle induced by acute heat stress. Poult Sci 84:307–314
Nakano T (2007) Microorganisms. In: Nakagawa H, Sato M, Gatlin DM III (eds) Dietary supplements for the health and quality of cultured fish. CAB International, Oxfordshire, pp 86–108
Nakano T (2011) Stress in fish. Yoshoku (Aquaculture Magazine) 48:64–67
Nakano T (2015) Rising to the challenge of reconstructing the coastal fisheries environment following the massive tsunami in Japan: the national 10-year “Tohoku Ecosystem-Associated Marine Sciences (TEAMS)”. In: Ceccaldi HJ, Hénocque Y, Koike Y, Komatsu T, Stora G, Tusseau-Vuillemin M-H (eds) Marine productivity: perturbations and resilience of socio-ecosystems. Springer International Publishing AG, Cham, Switzerland, pp 63–68
Nakano T, Takeuchi M (1997) Relationship between fish and reactive oxygen species. Yoshoku (Aquaculture Magazine) 34:69–73
Nakano T, Sato M, Takeuchi M (1992a) Partial purification and properties of glutathione peroxidase from carp hepatopancreas. Comp Biochem Physiol 102B:31–35
Nakano T, Sato M, Takeuchi M (1992b) Glutathione peroxidase of fish. J Food Sci 57:1116–1119
Nakano T, Tosa M, Takeuchi M (1995) Improvement of biochemical features in fish health by red yeast and synthetic astaxanthin. J Agric Food Chem 43:1570–1573
Nakano T, Kanmuri T, Sato M, Takeuchi M (1999a) Effect of astaxanthin rich red yeast (Phaffia rhodozyma) on oxidative stress in rainbow trout. Biochim Biophys Acta 1426:119–125
Nakano T, Miura Y, Wazawa M, Sato M, Takeuchi M (1999b) Red yeast Phaffia rhodozyma reduces susceptibility of liver homogenate to lipid peroxidation in rainbow trout. Fish Sci 65:961–962
Nakano T, Wazawa M, Yamaguchi T, Sato M, Iwama GK (2004) Positive biological actions of astaxanthin in rainbow trout. Mar Biotechnol 6:S100–S105
Nakano T, Masuda M, Suzuki T, Ohshima H (2012a) Inhibition by polyphenolic phytochemicals and sulfurous compounds of the formation of 8-chloroguanosine mediated by hypochlorous acid, human myeloperoxidase, and activated human neutrophils. Biosci Biotechnol Biochem 76:2208–2213
Nakano T, Shiba Y, Yamaguchi T, Sato M, Ohshima H (2012b) Natural marine products inhibit formation of 8-chloroguanosine mediated by hypochlorous acid, myeloperoxidase and activated neutrophils. Free Radic Biol Med 53:S95
Nakano T, Afonso LO, Beckman BR, Iwama GK, Devlin RH (2013) Acute physiological stress down-regulates mRNA expressions of growth-related genes in coho salmon. PLoS ONE 8:e71421
Nakano T, Kameda M, Shoji Y, Hayashi S, Yamaguchi T, Sato M (2014) Effect of severe environmental thermal stress on redox state in salmon. Redox Biol 2:772–776
Nakano T, Kameda M, Yamaguchi T, Sato M, Afonso LOB, Beckman BR, Iwama GK, Devlin RH (2015) Effect of thermal stressors on growth-related gene expressions in cultured fish. In: Ceccaldi HJ, Hénocque Y, Koike Y, Komatsu T, Stora G, Tusseau-Vuillemin M-H (eds) Marine productivity: perturbations and resilience of socio-ecosystems. Springer International Publishing AG, Cham, Switzerland, pp 147–157
Namdari R, Abedini S, Law FCP (1996) Tissue distribution and elimination of oxytetracycline in seawater chinook and coho salmon following medicated-feed treatment. Aquaculture 144:27–38
Nemcsok J, Benedeczky I (1995) Pesticide metabolism and the adverse effects of metabolites on fishes. In: Hochachka PW, Mommsen TP (eds) Environmental and ecological biochemistry. Biochemistry and molecular biology of fishes. Elsevier, Amsterdam, pp 313–348
Neuhaus-Steinmetz U, Xu C, Fracella F, Oberheitmann B, Richter-Landsberg C, Rensing L (1994) Heat shock response and cytotoxicity in C6 rat glioma cells: structure-activity relationship of different alcohols. Moll Pharmacol 45:36–41
Niki E (1988) Ascorbic acid, glutathione. In: Nakano M, Asada K, Oyanagui Y (eds) Reactive oxygen. Kyoritsu Shuppan, Tokyo, pp 321–326
Nogawa H, Yagisawa I (1994) Optimum environmental condition for rearing juvenile chum salmon (Oncorhynchus keta): a review. Sci Rep Hokkaido Salm Hatch 48:31–39
Ozaki H (1971) Liver weight. Fish physiology. Midori Shobo, Tokyo, pp 97–120
Ozaki H (1978) Diagnosis of fish health by blood analysis. In: Ishikawa T, Hanyu I, Hibiya K (eds) Respiration and circulation of fish. Koseisha Koseikaku, Tokyo, pp 63–80
Ozaki H (1980) Antibiotics 2. The pharmacology of fishes. Midori Shobo, Tokyo, pp 73–110
Pari L, Gnanasoundari M (2006) Influence of naringenin on oxytetracycline mediated oxidative damage in rat liver. Basic Clin Pharmacol Toxicol 98:456–461
Peng J, Jones LG, Watson K (2000) Stress proteins as biomarkers of oxidative stress: effects of antioxidant supplements. Free Radic Biol Med 28:1598–1606
Petrenko I, Titov VI, Vladimirov IA (1995) Generation of active forms of oxygen by antibiotics of the tetracycline series during tetracycline catalysis of oxidation of ferrous ion. Antibiot Khimioter 40:3–8
Pickering AD (1993) Growth and stress in fish production. Aquaculture 111:51–63
Prunet P, Overli O, Douxfils J, Bernardini G, Kestemont P, Baron D (2012) Fish welfare and genomics. Fish Physiol Biochem 38:43–60
Quinlan GJ, Gutteridge MC (1988) Hydroxyl radical generation by the tetracycline antibiotics with free radical damage to DNA, lipids and carbohydrate in the presence of iron and copper salts. Free Radic Biol Med 5:341–348
Regoli F, Giuliani ME (2014) Oxidative pathways of chemical toxicity and oxidative stress biomarkers in marine organisms. Mar Environ Res 93:106–117
Reineck M (2010) Influences of the environment on the endocrine and paracrine fish growth hormone-insulin-like growth factor-I system. J Fish Biol 76:1233–1254
Rogstad A, Hormazabal V, Ellingsen OF, Rasmussen KE (1991) Pharmacokinetic study of oxytetracycline in fish. I. Absorption, distribution and accumulation in rainbow trout in freshwater. Aquaculture 96:219–226
Shin MH, Moon YJ, Seo J-E, Lee Y, Kim KH, Chung JH (2008) Reactive oxygen species produced by NADPH oxidase, xanthine oxidase, and mitochondrial electron transport system mediate heat shock-induced MMP-1 and MMP-9 expression. Free Radic Biol Med 44:635–645
Sies H (1999) Glutathione and its role in cellular functions. Free Radic Biol Med 27:916–921
Srikanth K, Pereira E, Duarte AC, Ahmad I (2013) Glutathione and its dependent enzymes’ modulatory responses to toxic metals and metalloids in fish—a review. Environ Sci Pollut Res 20:2133–2149
Topic Popovic N, Howell T, Babish JG, Bowser PR (2012) Cross-sectional study of hepatic CYP1A and CYP3A enzymes in hybrid striped bass, channel catfish and Nile tilapia following oxytetracycline treatment. Res Vet Sci 92:283–291
Ueno R, Kinoshita A, Wakabayashi J (2004) Comparative pharmacokinetics of oxytetracycline in eel and its fate in a close aquatic environment. Aquaculture 235:53–63
Uno K, Aoki T, Ueno R (1992) Pharmacokinetic study of oxytetracycline in cultured rainbow trout, amago salmon and yellowtail. Nippon Suisan Gakk 58:1151–1156
Valavanidis A, Vlahogianni T, Dassenakis M, Scoullos M (2006) Molecular biomarkers of oxidative stress in aquatic organisms in relation to toxic environmental pollutants. Ecotoxicol Environ Saf 64:178–189
Vasseur P, Cossu-Leguille C (2003) Biomarkers and community indices as complementary tools for environmental safety. Environ Int 28:711–717
Yamamoto Y (1981) Determination of toxicity by biochemical method. In: Egami N (ed) Fish as laboratory animals. Soft Science, Tokyo, pp 420–448
Yonar ME (2012) The effect of lycopene on oxytetracycline-induced oxidative stress and immunosuppression in rainbow trout (Oncorhynchus mykiss, W.). Fish Shellfish Immunol 32:994–1001
Yonar ME, Yonar SM, Silici S (2011) Protective effect of propolis against oxidative stress and immunosuppression induced by oxytetracycline in rainbow trout (Oncorhynchus mykiss, W.). Fish Shellfish Immunol 31:318–325
Zhang Q, Li X (2007) Pharmacokinetics and residue elimination of oxytetracycline in grass carp, Ctenopharyngodon idellus. Aquaculture 272:140–145
Zounkova R, Klimesova Z, Nepejchalove L, Hilscherova K, Blaha L (2011) Complex evaluation of ecotoxicity and genotoxicity of antimicrobials oxytetracycline and flumequine used in aquaculture. Environ Toxicol Chem 30:1184–1189
Acknowledgments
The authors wish to thank Mr. T. Mandeville at New Day English Language Services, Sendai, Japan, for editing this manuscript. The authors are grateful to Drs. M. Sato, T. Yamaguchi, and H. Shirakawa at Tohoku University, Japan; S. Lertsiri at Mahidol University; and U. Na-Nakorn and W. Worawattanamateekul at Kasetsart University, Thailand, for valuable discussion and suggestions. The authors wish to thank Miss. A. Yamauchi and Miss. R. Okada at Tohoku University for assistance in laboratory work. The authors also acknowledge Drs. T. Nakano at Osaka University and K. Kubono at ASKA Animal Health Co., Ltd., for their help in the preparation of this paper. This study was supported in part by a Grant-in-Aid for Scientific Research (KAKENHI, grant number 23580277) from the Japan Society for the Promotion of Science (JSPS) to TN.
Conflict of interest
The authors declare that they have no competing interests.
Author contributions
TN and SH conceived and designed the experiments. TN, SH, and NN performed the experiments. TN, SH, and NN analyzed the data. TN wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Nakano, T., Hayashi, S. & Nagamine, N. Effect of excessive doses of oxytetracycline on stress-related biomarker expression in coho salmon. Environ Sci Pollut Res 25, 7121–7128 (2018). https://doi.org/10.1007/s11356-015-4898-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-015-4898-4