Abstract
Objective
The objective of this study is to examine the diurnal variability of C-reactive protein (CRP) in obstructive sleep apnea (OSA).
Methods and measurements
Participants included 44 women and men with untreated OSA (mean apnea/hypopnea index = 37.5, SD ± 28) and 23 healthy adults with no OSA. Sleep was monitored with polysomnography in the University of California San Diego General Clinical Research Center. Over a 24-h period, blood was collected every 2 h, and CRP levels were determined.
Results
Adjusting for age, gender, and body mass index, a significant group by time interaction showed that patients with OSA had higher CRP levels during the daytime (8:00 a.m.–8:00 p.m.) versus the nighttime (10:00 p.m. until 6:00 p.m.; p < 0.001). Non-apneics showed no significant change in CRP levels during the 24 h.
Conclusions
The findings indicate that sleep apnea patients have disproportionately elevated CRP levels in the day versus the nighttime, possibly as a result of carryover effects of nighttime arousal into the daytime.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Inflammation is a hallmark of the pathophysiology of obstructive sleep apnea (OSA) and represents a pathway linking OSA to increased cardiovascular morbidity [1-3]. C-reactive protein (CRP) is an acute phase response protein implicated in broad range of cardiovascular diseases [4]. Through its effects on adhesion molecules and mononuclear cell recruitment and adhesion [5, 6], CRP plays an important role in atherosclerosis development [7].
Independent of age and body mass index (BMI), CRP levels are found to be elevated in OSA and associated with severity of apnea as indexed by the apnea/hypopnea index (AHI) [8-11], although not all studies demonstrate this relationship [12]. These studies of CRP and OSA have relied on a single blood sample obtained in the morning or afternoon hours to determine CRP levels, with few, if any, utilizing multiple blood sampling to examine the diurnal variation of CRP in OSA.
Studies in healthy individuals have not demonstrated a diurnal variation of CRP. Meier-Ewert et al., for example, determined CRP concentrations in hourly blood samples in 13 healthy individuals and found no rhythmic diurnal variation over a 24-h period [13]. Of relevance to OSA are studies examining the effects of sleep disruption and/or deprivation on CRP levels. In 43 otherwise healthy women, Okun et al. showed that elevated CRP levels (single samples obtained anywhere between the hours of 10:00 a.m. and 4:00 p.m.) were associated with self-reported sleep disruptions [14]. Laboratory studies of sleep deprivation report inconsistent findings. Frey et al. studied the effects of 40 h sleep deprivation in 19 healthy individuals; sampling CRP every 30 min revealed a significant decrease in CRP in morning levels (between the hours of 9:00 a.m. and 11:00 a.m.) and in late night levels (between the hours of 11:00 p.m. and 12:00 a.m.) [15]. Meier-Ewert et al. examined the effects of extended (88 h) and partial (4 h) sleep deprivation on CRP levels in ten healthy individuals; blood sampling every 60 to 90 min revealed that sleep deprivation was associated with marked elevation of daily averages of CRP levels [16]. In contrast to these studies, Dimitrov et al. using an 8-h sleep deprivation in 15 healthy individuals with blood sampling every 1.5 or 3 h showed no effects on CRP levels [17]. Thus, it is fair to say that the literature is not consistent concerning effects of sleep disruption on CRP.
The purpose of this study is to examine the diurnal pattern of CRP in individuals with OSA and to compare that pattern to that observed in otherwise healthy individuals with normal sleep.
Materials and methods
Forty-four women and men with untreated OSA and 23 healthy adults with no OSA were recruited by advertising and word of mouth referral. Participants were excluded if they reported a history of major medical illnesses (other than OSA and hypertension), current psychiatric diagnoses, or if they were receiving psychotropic medication. Patients who were receiving anti-hypertensive medications had their medications tapered for 3 weeks prior to participation. The protocol was approved by the University of California San Diego (UCSD) Human Subjects Committee. After description of the study, written informed consent was obtained.
Participants arrived at the UCSD General Clinical Research Center Gillin Laboratory of Sleep and Chronobiology (GCRC-GLSC) at 5:00 p.m., at which time, a venous catheter was inserted. Starting at 6:00 p.m., a blood sample was collected every 2 h for the next 24 h. Blood samples were collected in ethylenediaminetetraacetic acid, placed on ice, spun in a refrigerated centrifuge, and the plasma was stored at −80°C until assayed. CRP was assayed by using the high sensitivity Denka-Seiken assay [18]. The assay’s intra-assay coefficient of variation (CV) is <1.0%, the inter-assay CV is 1.6%, and the sensitivity is <0.05 mg/l. To minimize intra-assay variance, all 12 samples from a participant were analyzed in the same run.
Starting at 8:00 p.m. the following evening (i.e., upon completion of the 24-h blood sampling), participants were instrumented for standard polysomnography [19]. Lights were out at 10:00 p.m., and recording continued until 7:00 a.m. Polysomnography records were scored by trained sleep technicians for total sleep time, total minutes spent awake after sleep onset (WASO), and apneas and hypopneas. Apneas were defined as decrements in airflow of ≥90% from baseline for ≥10 s. Hypopneas were defined as decrements in airflow of ≥50% but <90% from baseline for ≥10 s. The numbers of apneas and hypopneas per hour of sleep were calculated to obtain the AHI. A diagnosis of OSA was given if AHI ≥10.
Statistical analysis
Means and standard deviations or frequencies were calculated for demographic and apnea-related variables. Subject characteristics data were compared between apneics and non-apneics by one-way analysis of variance for continuous variables and chi-squared tests for categorical variables. Box-plots and histograms were used to examine distributions of CRP levels. Line-plots of CRP levels over a 24-h period for each subject were also graphed to assess trends in marker levels. Based on these graphs, we used a linear mixed-effects model approach to characterize the time-course of CRP levels over the 24-h period. These models were fitted with restricted maximum likelihood methods [20]. This modeling strategy was adopted because it is a flexible approach that can account for correlations between repeated measures on an individual, include patient-specific intercept and slope terms (referred to as random effects), adjust for potentially confounding covariates (referred to as fixed-effects), and allow for unbalanced data (i.e., different individuals can have different numbers of observations). Regarding unbalanced data, four participants did not have all 12 CRP data points (one OSA patient had nine, one OSA patient had ten, one control had nine, and one control had 11).
The model included a random effect at the patient level (random intercept term in the model) to allow for variability in initial CRP levels between individuals. The following covariates (fixed-effects) were also included in the models: OSA status (present versus absent: reference was participant’s without OSA), time (12 time points at the 2-h intervals with the analysis starting at the 10:00 p.m. time point), and potential confounders (age, BMI, and gender). Two separate models were run, one with time as a continuous variable and the other with time as a categorical night versus day variable. A likelihood ratio test was used to test the significance of the interaction between time and OSA status to assess whether profiles of CRP levels over the day varied by OSA status. Residual plots and quantile–quantile plots were used to assess adequacy of fit of the models. Data were analyzed using the R statistics software package (http://cran.stat.ucla.edu). CRP values were log-transformed prior to analysis to better approximate a Gaussian distribution. Statistical significance was set at p < 0.05.
Results
Sample characteristics
Sample characteristics are presented in Table 1. The OSA group was older (F = 5.1, p < 0.05), had a greater BMI (F = 5.23, p < 0.5), as well as higher systolic (F = 15.1, p < 0.001) and diastolic (F = 4.43, p < 0.05) blood pressure. By definition, the AHI index was greater in those with OSA (F = 20.8, p < 0.001). OSA patients spent approximately 44.5 fewer minutes asleep (F = 13.3, p < 0.01). There was a marginally significant group difference for WASO (F = 3.54, p = 0.065), with OSA patients spending more time awake after the onset of sleep. Unadjusted mean 24-h CRP levels were marginally higher in sleep apneics (t = 1.91, p = 0.06). We also examined correlations between (log-transformed) CRP levels and AHI among all participants. Across the 12 individual CRP time points, correlations with AHI ranged from a low of r = 0.26 (p = 0.10) at the 8:00 p.m. time point to a high of r = 0.34 (p = 0.04) at the 4:00 p.m. time point. The mean CRP value correlated with AHI at r = 0.28, p ≤ 0.05.
Diurnal rhythm
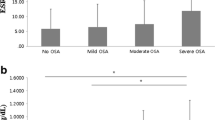
The CRP levels throughout the day are graphed in Figs. 1 and 2 according to group. Model results, including regression coefficients, slope, and p values, are presented in Table 2 for the two models that were fit. Model I was a mixed model starting time at 10:00 p.m. with time as a continuous variable. There were significant main effects of BMI (with higher BMI being associated with higher CRP levels; regression coefficient = 0.094, p < 0.001) and gender (with CRP levels being somewhat higher in women; regression coefficient = 0.667, p = 0.04) for the 24-h period. There was not a significant main effect (p = 0.27) or interaction (p = 0.13) for OSA group status.
Unadjusted 24-h CRP levels in OSA and healthy controls every 2 h starting at 10:00 p.m. (mean ± SEM). In an analysis adjusting for age, gender, and body mass index, patients with sleep apnea were found to have higher CRP levels during the daytime (8:00 a.m. until 8:00 p.m.) versus the nighttime (10:00 p.m. until 6:00 a.m.; p < 0.001); whereas, non-apneics showed no significant change in CRP levels during the 24 h
Model II, also starting with the 10:00 p.m. sample, had time as categorical variable, with night from 10:00 p.m. to 6:00 a.m. and day from 8:00 a.m. to 8:00 p.m. In addition to the same main effects for BMI and gender, a significant group by time interaction (regression coefficient = 0.081, p = 0.01) indicated that in the apneic group, mean CRP levels were higher during the day than at night (p < 0.001). There were no significant differences in day versus night CRP levels for the group with no sleep apnea.
We also conducted several sensitivity analyses to assess the adequacy of our fitted models. First, systolic and diastolic blood pressure, as well as smoking status were added to the mixed-effects models and were not significantly associated with CRP levels and did not change the observed association between OSA and 24-h CRP levels. In other sensitivity analyses, we also fitted cosinor models [21] which are a well-established method for modeling sleep-wake circadian rhythms. Residual plots and Akaike information criteria indicated that the cosinor models did not fit the CRP data well, as would be expected given the reasonably stable CRP levels during the 24-h period evidenced in line-plots, especially among non-apneics.
Discussion
CRP is an important risk factor and biomarker for cardiovascular diseases [4-7]. Prior studies using morning or afternoon sampling found CRP to be elevated in individuals with OSA compared to non-OSA adults [8-11]. We found 24-h mean CRP levels to be marginally higher in the patients with OSA. By obtaining samples every 2 h during the 24-h period, we were able to demonstrate a diurnal difference in CRP levels in OSA, with daytime levels being significantly higher than nighttime levels. We observed this interaction in an analytic model with time as a categorical night versus day variable and not an analytic model with time as a continuous variable. We suspect that the categorical analysis, when the two groups had the maximum difference in CRP levels, gave us the best power to discern the interaction. The p value of the interaction for the time as a continuous variable was p = 0.13, and with a larger sample-size, a significant interaction might have emerged.
OSA is characterized by repeated disruptions of sleep. More periodic sleep disruptions, as assessed by subjective report, have been associated with elevated CRP levels [14]. Studies of more significant sleep disruption, i.e., sleep deprivation, report conflicting findings on CRP levels, including a decrease [15], increase [16], and no change [17] in CRP levels. Consistent with one prior study to examine this issue, we found no evidence of diurnal variation of CRP in healthy individuals not experiencing disrupted sleep [13], raising the question whether it is the disturbed sleep associated with apneas and hypopneas in OSA patients or some other characteristic of apnea patients that is the responsible for the effect on CRP diurnal rhythm.
A limitation of this study is that we characterized night as starting at 10:00 p.m. (when the lights were turned off in the GCRC-GLSC) and ending at 6:00 a.m. This definition of night would not have universally applied to the night/day schedule for all of the participants.
What mechanisms might underlie the effect of OSA on inducing a diurnal rhythm of CRP? Sleep disruption and partial sleep deprivation are both associated with sympathetic activation [22, 23]. Sleep apneics show elevated norepinephrine levels as compared to non-apneics [24]. The repeated disruption of sleep as determined by the number of apneic events and/or the severity of nocturnal hypoxia are directly associated with the elevated norepinephrine levels [25-27]. Some of the earliest observations with catecholamines indicated that nighttime sympathetic over-activity in OSA carried over to the daytime when apnea events were not occurring [26, 28]. Elevations in catecholamines are associated with elevations in CRP levels via hepatocyte β-adrenergic receptors triggering CRP biosynthesis and release [29, 30]. Another possible mechanism is inflammatory cytokines. CRP synthesis in the liver is controlled by interleukin-6 (IL-6) as well as by tumor necrosis factor-alpha (TNF-α) [31]. A study by Entzian et al. in OSA reported no evidence of circadian rhythm change in ex vivo levels of IL-6 from cultured blood samples [32], although studies do indicate that circulating IL-6 levels are elevated in OSA [33]. Entzian et al. did report that the circadian rhythm of ex vivo TNF-α was disturbed in OSA such that the nocturnal physiologic peaks were absent but that an additional daytime peak was evident [32]. Thus, in addition to catecholamines, an additional potential mechanism of our findings is an effect of OSA on the diurnal rhythm of TNF-α.
What might be the significance of greater daytime than nighttime CRP levels in OSA? Studies link elevations in daytime CRP with other markers of increased cardiovascular risk, including with greater nighttime blood pressure variability and with non-dipping of blood pressure [34, 35]. Studies in OSA show increased risk for cardiovascular events in the daytime, and this risk is attributed to several mechanisms, including local and systemic inflammation [36, 37]. Regardless, our data suggests that in order to better understand inflammatory variables in OSA, fine-grained chronobiological analyses with frequent blood sampling will be invaluable.
References
Benjamin JA, Lewis KE (2008) Sleep-disordered breathing and cardiovascular disease. Postgrad Med J 84(987):15–22. doi:10.1136/pgmj.2007.062836
Williams A, Scharf SM (2007) Obstructive sleep apnea, cardiovascular disease, and inflammation—is NF-κB the key? Sleep Breath 11(2):69–76. doi:10.1007/s11325-007-0106-1
Mills PJ, Dimsdale JE (2004) Sleep apnea: a model for studying cytokines, sleep, and sleep disruption. Brain Behav Immun 18(4):298–303. doi:10.1016/j.bbi.2003.10.004
Schwedler SB et al (2006) C-reactive protein: a family of proteins to regulate cardiovascular function. Am J Kidney Dis 47(2):212–222. doi:10.1053/j.ajkd.2005.10.028
Cottone S et al (2006) Relation of C-reactive protein to oxidative stress and to endothelial activation in essential hypertension. Am J Hypertens 19(3):313–318. doi:10.1016/j.amjhyper.2005.09.005
Zhang J et al (2006) C-reactive protein induced expression of adhesion molecules in cultured cerebral microvascular endothelial cells. Life Sci 78(26):2983–2988. doi:10.1016/j.lfs.2005.11.027
Teoh H et al (2008) Impaired endothelial function in C-reactive protein overexpressing mice. Atherosclerosis 201(2):318–325
Chung S et al (2009) Endothelial dysfunction and inflammatory reactions of elderly and middle-aged men with obstructive sleep apnea syndrome. Sleep Breath 13(1):11–17
Ye J et al (2007) Increased serum levels of C-reactive protein and matrix metalloproteinase-9 in obstructive sleep apnea syndrome. Chin Med J (Engl) 120(17):1482–1486
Punjabi NM, Beamer BA (2007) C-reactive protein is associated with sleep disordered breathing independent of adiposity. Sleep 30(1):29–34
Kapsimalis F et al (2008) Association of sleep apnea severity and obesity with insulin resistance, C-reactive protein, and leptin levels in male patients with obstructive sleep apnea. Lung 186(4):209–217. doi:10.1007/s00408-008-9082-x
Guilleminault C, Kirisoglu C, Ohayon MM (2004) C-reactive protein and sleep-disordered breathing. Sleep 27(8):1507–1511
Meier-Ewert HK et al (2001) Absence of diurnal variation of C-reactive protein concentrations in healthy human subjects. Clin Chem 47(3):426–430
Okun ML, Coussons-Read ME (2007) Sleep disruption during pregnancy: how does it influence serum cytokines? J Reprod Immunol 73(2):158–165. doi:10.1016/j.jri.2006.06.006
Frey DJ, Fleshner M, Wright KP Jr (2007) The effects of 40 hours of total sleep deprivation on inflammatory markers in healthy young adults. Brain Behav Immun 21(8):1050–1057. doi:10.1016/j.bbi.2007.04.003
Meier-Ewert HK et al (2004) Effect of sleep loss on C-reactive protein, an inflammatory marker of cardiovascular risk. J Am Coll Cardiol 43(4):678–683. doi:10.1016/j.jacc.2003.07.050
Dimitrov S et al (2006) Sleep enhances IL-6 trans-signaling in humans. FASEB J 20(12):2174–2176. doi:10.1096/fj.06-5754fje
Roberts WL et al (2001) Evaluation of nine automated high-sensitivity C-reactive protein methods: implications for clinical and epidemiological applications. Part 2. Clin Chem 47(3):418–425
von Kanel R et al (2007) Association between polysomnographic measures of disrupted sleep and prothrombotic factors. Chest 131(3):733–739. doi:10.1378/chest.06-2006
Diggle PJ, Liang K, Zeger S (1996) Analysis of longitudinal data. Clarendon Press, Oxford
Marler MR et al (2006) The sigmoidally transformed cosine curve: a mathematical model for circadian rhythms with symmetric non-sinusoidal shapes. Stat Med 25(22):3893–3904
Markov D, Goldman M (2006) Normal sleep and circadian rhythms: neurobiologic mechanisms underlying sleep and wakefulness. Psychiatr Clin North Am 29(4):841–853 (abstract vii)
Irwin M et al (1999) Effects of sleep and sleep deprivation on catecholamine and interleukin-2 levels in humans: clinical implications. J Clin Endocrinol Metab 84(6):1979–1985
Coy TV et al (1996) Sleep apnoea and sympathetic nervous system activity: a review. J Sleep Res 5(1):42–50
Ziegler MG et al (2001) Effect of continuous positive airway pressure and placebo treatment on sympathetic nervous activity in patients with obstructive sleep apnea. Chest 120(3):887–893
Fletcher EC (2003) Sympathetic over activity in the etiology of hypertension of obstructive sleep apnea. Sleep 26(1):15–19
Dimsdale JE et al (1997) Sympathetic nervous system alterations in sleep apnea. The relative importance of respiratory disturbance, hypoxia, and sleep quality. Chest 111(3):639–642
Peled N et al (1998) Contributions of hypoxia and respiratory disturbance index to sympathetic activation and blood pressure in obstructive sleep apnea syndrome. Am J Hypertens 11(11 Pt 1):1284–1289
Aso Y et al (2006) High serum high-sensitivity C-reactive protein concentrations are associated with relative cardiac sympathetic overactivity during the early morning period in type 2 diabetic patients with metabolic syndrome. Metabolism 55(8):1014–1021
Wessel J et al (2007) C-reactive protein, an ‘intermediate phenotype’ for inflammation: human twin studies reveal heritability, association with blood pressure and the metabolic syndrome, and the influence of common polymorphism at catecholaminergic/beta-adrener. J Hypertens 25(2):329–343
Baumann H, Gauldie J (1994) The acute phase response. Immunol Today 15(2):74–80
Entzian P et al (1996) Obstructive sleep apnea syndrome and circadian rhythms of hormones and cytokines. Am J Respir Crit Care Med 153(3):1080–1086
Vgontzas AN et al (2005) IL-6 and its circadian secretion in humans. Neuroimmunomodulation 12(3):131–140
Tsioufis C et al (2008) Disturbed circadian blood pressure rhythm and C-reactive protein in essential hypertension. J Hum Hypertens 22(7):501–508
Abramson JL et al (2006) Relation of C-reactive protein and tumor necrosis factor-alpha to ambulatory blood pressure variability in healthy adults. Am J Cardiol 98(5):649–652
Zamarron C, Garcia Paz V, Riveiro A (2008) Obstructive sleep apnea syndrome is a systemic disease. Current evidence. Eur J Intern Med 19(6):390–398
Merritt SL (2004) Sleep-disordered breathing and the association with cardiovascular risk. Prog Cardiovasc Nurs 19(1):19–27
Acknowledgements
This work was supported in part NIH grants HL073355, HL44915, HL36005, and CA23100.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Mills, P.J., Natarajan, L., von Känel, R. et al. Diurnal variability of C-reactive protein in obstructive sleep apnea. Sleep Breath 13, 415–420 (2009). https://doi.org/10.1007/s11325-009-0268-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-009-0268-0