Abstract
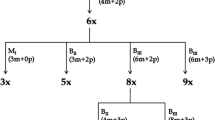
Apomixis is usually subdivided into parthenogenesis, apogamy, and apospory on the basis of different mechanisms of development and cytogenetic effects. Although most plants and animals undergo sexual reproduction to multiply, it is important to understand the role of apomixis in plant reproduction because of its useful applications in the breeding and propagation of some crops. Tetraploid Malus xiaojinensis Cheng et Jiang is a typical facultative apomictic plant species. Paternity and ploidy analyses of M. xiaojinensis seedlings were performed with flow cytometry in conjunction with microsatellite and single nucleotide polymorphism markers. The proportion of apomictic derived progeny of M. xiaojinensis × Malus baccata was 54.05, 53.96, and 43.55 % from 2008 to 2010, respectively. The progeny of M. xiaojinensis × M. baccata comprised hybrids of 2x, 3x, 4x, or 5x ploidy, whereas apomictic offspring were 2x, 3x, or 4x. Among the apomictic derived progeny, the ploidy of restituted apomictic progeny was 2x, 3x, or 4x, whereas that of nonrestituted apomictic progeny was 4x only. The proportions of the different types of apomictic derived progeny differed among the years 2008 to 2010. The ploidy of progeny from emasculated flowers also segregated into 2x, 3x, and 4x individuals. These results indicate that both parthenogenesis and unreduced meiotic diplospory play important roles in apomictic reproduction in M. xiaojinensis.


Similar content being viewed by others
References
Bicknell RA, Koltunow AM (2004) Understanding apomixes: recent advances and remaining conundrums. Plant Cell 16:228–245
Bisognin C, Seemuller E, Citterio S, Velasco R, Grando MS, Jarausch W (2009) Use of SSR markers to assess sexual vs. apomictic origin and ploidy level of breeding progeny derived from crosses of apple proliferation-resistant Malus sieboldii and its hybrids with Malus × domestica cultivars. Plant Breed 128:507–513
Boldrini KR, Adamowski EV, Silva N, Pagliarini MS, Valle CB (2011) Meiotic behavior in nonaploid accessions of Brachiaria humidicola (Poaceae) and implications for breeding. Genet Mol Res 10:169–176
Button J, Kochba J, Bornman CH (1974) Fine structure of and embryoid development from embryogenic ovular callus of “Shamouti” orange (Citrus sinensis Osb). J Exptl Bot 25:446–457
Cai X, Mu XJ, Zhu ZQ, Hua ZM, Huang YH, Sun QX (1997) Polyembryony and multiple seedlings in the apomictic plants. Acta Bot Sin 39:590–595
Chen RY, Song WQ, Li XL (1982) Wall degradation hypotonic method of preparing chromosome samples in plant and its significance in the cytogenetics. Acta Genet Sin 9:151–159
Cheng MH, Yang XH, Zeng WG (1984) A preliminary report on investigation and study of M. xiaojinensis—an apple stock resource. J Southwest University 6:38–43
Doyle JJ, Doyle JL (1990) Isolation of plant DNA from fresh tissue. Focus 12:13–15
Erwan AG, Aldo A, Dominique M, Bernard K (1996) Parthenogenesis and apospory in the Laminariales: a flow cytometry analysis. Eur J Phycol 31:369–380
Espinoza F, Daurelio LD, Pessino SC, Valle EM, Quarin CL (2006) Genetic characterization of Paspalum notatum accessions by AFLP markers. Plant Syst Evol 258:147–159
Harada T, Matsukawa K, Sato T, Ishikawa R, Niizeki M, Saito K (1993) DNA-RAPDs detect genetic variation and paternity in Malus. Euphytica 65:87–91
Huang YM, Chou HM, Hsieh TH, Wang JC, Chou WL (2006) Cryptic characteristics distinguish diploid and triploid varieties of Pteris fauriei (Pteridaceae). Can J Bot 84:261–268
Johri BM, Ambegaokar KB, Srivastava PS (1995) Comparative embryology of Angiosperms. Springer, New York, pp 338–339
Kamelina OP (1995) Synergid apogamety in the genus Tetradiclis Stev. (Tetradiclidaceae) and occurrence of this phenomenon flowering plants. Apomixis Newsl 8:32–34
Kroon GH, Zeilinga AE (1974) Apomixis and heterogamy in rose rootstocks (Rosa canina L.). Euphytica 23:345–352
Li YN (2001) Research of germplasm resources of Malus Mill. China Agriculture, Beijing, pp 207–210
Li YH, Han ZH, Xu XX (2004) Segregation patterns of AFLP markers in F1 hybrids of a cross between tetraploid and diploid species in the genus Malus. Plant Breed 123:316–320
Li LG, He P, Ou CQ, Li HF, Zhang ZH (2008) Culture and identification of seedlings from triploid apple. J Fruit Sci 25:400–403
Liu GJ, Zhang XZ, Ru GR, Bai QH (1997) 2n pollen investigation on apple germplasm. Hebei Fruit 32:9–11
Liu Y, Dong WX, Zhang L, Cheng XD (2006) Study on the development characteristics of different embryo sac types of Pingyi Tiancha (Malus hupehensis). J Fruit Sci 23:330–334
Liu CH, Zhang QP, Yao JL, Chen CL (2007) Karyotype analysis and pollen mother cells meiosis observation in Eulaliopsis binata. Scientia Agriculture Sinica 40:27–33
Lubbers EL, Arthur L, Hanna WW, Ozias-Akins P (1994) Molecular markers shared by diverse apomictic Pennisetum species. Theor Appl Genet 89:636–642. doi:10.1007/BF00222459
Luo DG (1997) Inheritance of purple embryo marker trait and its utilization to apomixes research in maize. J Yunnan Agricultural University 12:114–118
Maheshwari P (1950) An introduction to the embryology of Angiosperms. McGraw-Hill, New York, pp 127–155
Morrion LA, Metzger RJ, Lukaszewski AJ (2004) Origin of the blue-aleurone gene in Sebesta Blue wheat genetic stocks and a protocol for it s use in apomixes screening. Crop Sci 44:2063–2067
Nakahara H, Nakamura Y (1973) Parthenogenesis, apogamy and apospory in Alaria crassifolia (Laminariales). Mar Biol 18:327–332
Olien WC (1987) Apomictic crabapples and their potential for research and fruit production. Hort Sci 22:541–546
Ozias-Akins P (2006) Apomixis: developmental characteristics and genetics. Crit Rev Plant Sci 25:199–214
Pichot C, Maataoui M, Radd S, Raddi P (2001) Surrogate mother for endangered Cupressus. Nat 412:39
Riccardo V, Andrey Z, Jason A et al (2010) The genome of the domesticated apple (Malus × domestica Borkh.). Nat Genet. doi:10.1038/ng.654
Risso-Pascotto C, Pagliarini MS, do Valle CB (2005) Multiple spindles and cellularization during microsporagenesis in an artificially induced tetraploid accession of Brachiaria ruzizensis (Gramineae). Plant Cell Rep 23:522–527
Sauer S, Gut IG (2002) Genotyping single nucleotide polymorphisms by matrix-assisted laser desorption/ionization time of flight mass spectrometry. J Chromatogr B 782:73–87
Savidan Y (2000) Apomixis: genetics and breeding. Plant Breed Rev 18:13–86
Silfverberg-Dilworth E, Matasci CL, Van de Weg WE, Van Kaauwen MPW, Walser M, Kodde LP, Soglio V, Gianfranceschi L, Durel CE, Costa F, Yamamoto T, Koller B, Gessler C, Patocchi A (2006) Microsatellite markers spanning the apple (Malus × domestica Borkh.) genome. Tree Genet Genomes 2:202–224
Takeru G, Toru M, Eiichi I, Mikio H, Toshiya Y, Tateki H, Fumio S, Masakazu K (2006) Overcoming hybrid lethality in a cross between Japanese pear and apple using gamma irradiation and confirmation of hybrid status using flow cytometry and SSR markers. Scientia Hort 109:43–47
Takuro K (1992) Apple culture in Japan. Chronica Hort 32:37–39
Talent N, Dickinson TA (2007) Apomixis and hybridization in Rosaceae subtribe Pyrinae Dumort.: a new tool promises new insights. Regnum Veg 147:301–316
Ur-Rahman H, James DJ, Hadonou AM, Caligari PDS (1997) The use of RAPD for verifying the apomictic status of seedlings of Malus species. Theor Appl Genet 95:1080–1083
Wieners RR, Fei SZ, Johnson RC (2006) Characterization of a USDA Kentucky bluegrass (Poa pratensis L.) core collection for reproductive mode and DNA content by flow cytometry. Genet Resour Crop Evol 53:1531–1541
Yan LY, Zhang XZ, Liu GJ (2000) Occurrence of unreduced gametes and ploidy restoration in haploid Capsicum annum L. J Hort Sci Biotech 75:195–197
Yang XH, Zhou ZQ (2009) Anatomical characters of apomixis development in the gynoecia of M. xiaojinensis. J Fruit Sci 26(1):1–5
Yang F, Zhang JE, Yi K, Liu Z, Rong ZX, Wang DM, Yan ZY (2010) Genetic trend of hybrid trees from the cross between Pingyi Tiancha (Malus hupehensis var. pingyiensis) and B9 (Malus cv. B9). J Fruit Sci 27(3):323–327
Zhang XZ, Liu GJ, Yan LY, Zhao YB, Chang RF, Shu HR (2003) Creating triploid germplasm via 2n gametes in Capsicum annuum L. J Hort Sci Biotech 78:84–88
Acknowledgments
This work was supported by the National High Technology Research and Development Program (no. NC2010BF0075), Non-Profit Industry Research and Special Program (no. 201203075), and National Apple's Industrial Technology System (no. CARS-28). Many thanks to Professor Guolu Liang and his colleagues at the Southwestern University of China, who provided valuable assistance with chromosome counting.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by R. Velasco
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
Meiotic behavior of microsporocytes of M. xiaojinensis at the tetrad stage. a, b Normal tetrads; c monad; d, e dyads; f unequal dyad; g, h triads; i–l polyads. Arrows indicate micronuclei (PDF 5933 kb)
Rights and permissions
About this article
Cite this article
Wang, L., Han, D., Gao, C. et al. Paternity and ploidy segregation of progenies derived from tetraploid Malus xiaojinensis . Tree Genetics & Genomes 8, 1469–1476 (2012). https://doi.org/10.1007/s11295-012-0535-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11295-012-0535-2