Abstract
The primary objective of this paper was to estimate how the mass of Silver fir (Abies alba Mill.) and Scots pine (Pinus sylvestris L.) deadwood in two decay classes affected biochemical processes and the accumulation of soil organic matter, as well as the extent of this impact. We evaluated deadwood mass, as well as the biological activity and influence of the distance from deadwood on biological activity and carbon (C) storage. The investigation was carried out in Magurski National Park, southern Poland, in four randomly selected study plots. The organic C and nitrogen contents and soil pH and texture were analysed. The study also included the determination of enzyme activities. The results obtained demonstrated that soil C storage and the biological activity of the soil were influenced by deadwood form, tree species and decay class. Significant differences in the C content in the soil between the logs of fir and pine trees were noted. With the advancement of the deadwood decomposition process, the C concentration and enzyme activity increased slightly. Lower C accumulation occurred further away from the deadwood. At a 100-cm distance from the deadwood, the influence of the logs was not evident. Higher C storage was found in the soil around the stumps than in the decaying logs. This study confirmed an important role of deadwood in forest ecosystems, i.e., maintaining species biodiversity and increasing C accumulation in the soil environment with a simultaneous increase in the biological activity of the soil.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Deadwood is a key component of carbon (C) pools in forest ecosystems (Caz 1993; Currie et al. 2003; Bantle et al. 2014). It affects soil development, C stores, nutrients and water and reduces erosion at the same time (Kupferschmidt et al. 2003; Pichler et al. 2011). According to Adamowicz et al. (2015), Polish forests have approximately 52 million m3 of wood that originates from dead trees. The amount of organic matter and soil C stocks are dependent on the vegetation, site conditions and forest management practices (Yin 1999; Debeljak 2006). Deadwood stock is influenced by forest age, and old-growth forests usually contain large quantities of C (Harmon and Hua 1991; Takahashi et al. 2000). In protected forest areas and in intensively managed forest ecosystems (old-growth Norway spruce mixed forest), stumps are usually present. These stumps generally represent up to 10–25% of the total biomass of living trees, and they store 15–20% of base cation content (Finér et al. 2003). The stumps are a major pool of soil organic matter and nutrients, and they play an important role in maintaining soil fertility (Wang et al. 2012). However, little is known regarding the effect of stumps on soil properties, particularly the biological activity (Kappes et al. 2007).
Deadwood serves as a habitat and resource for many insects and microbes (Davies et al. 2008; Persiani et al. 2010; Cocciufa et al. 2014), and it is a seedbed for plants (Narukawa et al. 2003; Bače et al. 2012). Soil enzymes produced by microbes play a key role in the biochemical process of organic matter decomposition (Kotroczó et al. 2014). Dehydrogenases (DHs) are enzymes responsible for the oxidation of organic matter through the movement of protons and electrons from a substrate to an acceptor. This process constitutes a part of the respiration of soil microorganisms, and it is controlled by soil type and water–air conditions (Kandeler 1996). DHs provide information about the state of the environment and the activity of microorganisms in the soil. These enzymes are related to the quantitative changes in microorganism populations, as only strictly intracellular enzymes can truly reflect microbial metabolic activity; compared with the degradation processes of extracellular soil enzymes, they are rapidly mineralized by other enzymes, i.e., proteases (Wolińska and Stępniewska 2012). β-glucosidase (BG) is an enzyme that participates in the decomposition of cellulose to glucose. This enzyme catalyses the hydrolysis of glucosides. Cellulose is quantitatively the most important organic compound in the biosphere; consequently, the product of its enzymatic hydrolysis is important as an energy source for soil microorganisms, being of significant importance in the C cycle (Sinsabaugh et al. 1991).
In contrast to forest succession, the effects of deadwood on biochemical soil properties, birds and insects have not been extensively studied (Olajuyigbe et al. 2011; Rinta-Kanto et al. 2016). Studies examining the differences between the effect of coniferous and broadleaf stumps on soil were presented by Lombardi et al. (2013). However, knowledge of the impact of deadwood on soil for selected broadleaf or coniferous tree species, such as pine and fir, is lacking. Scots pine (Pinus sylvestris L.) is the main forest-forming tree species in central-eastern Europe, while Silver fir (Abies alba Mill.), together with beech (Fagus sylvatica L.), is the dominant tree species in the Carpathian region. Due to the high adaptability of common pine to different environmental conditions and the high productivity of fir stands (Jaworski and Pach 2014), these tree species have high economic and ecological importance in Europe. Therefore, information on the influence of pine and fir deadwood on soil properties, specifically C stocks, is valuable for forest management. In this study, we examined the effect of pine and fir deadwood in two considerably different decay classes on the soil organic matter stock and activities of enzymes known to play an important role in the C cycle. The analysis included deadwood in an advanced stage of decomposition to reveal the final result of the decomposition impact on the accumulation of C and on the enzyme activity in the soil. Selected decay classes prevailed in the forest areas covered by this study. We estimated soil organic matter stabilization at different distances from decaying logs and stumps based on part of a tree trunk that protruded from the ground after a tree had fallen. We aimed to estimate how the deadwood of different species in various phases of decomposition affected the accumulation of soil organic matter and enzyme activity, as well as the extent of this impact. The following hypotheses were tested: (1) biological activity would be stimulated in the presence of strongly decayed deadwood; (2) the most pronounced effect of deadwood on the soil properties would be reflected in its immediate vicinity; and (3) fir and pine deadwood would have different impacts on the C storage and biological activity of the soil.
Materials and methods
Study area
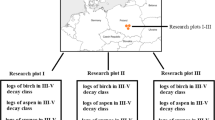
The investigation was conducted in Magurski National Park, southern Poland (Fig. 1, 21°46′56″E; 49°50′25″N). The mean annual precipitation in the study area ranges from 800 to 900 mm, and the average temperature is 6 °C. The elevation ranges from 300 to 600 m above sea level (a.s.l.). The research areas are characterized by exposure to a south-western slope of 15°–20° and the presence of a mountain forest habitat. Sample plots were located in an area with predominantly Magura tertiary sandstone and shale rocks, without the influence of glacial till or loess. The test area was dominated by Cambisols (WRB 2006) that developed from medium and fine-textured materials that had a favourable aggregate structure and a high content of clay minerals. All study sites had similar conditions. Four study plots were selected, including 60-year-old Silver fir stands (2 plots) and 50-year-old Scots pine stands (2 plots). In each study plot, 5 decaying logs and 2 stumps in two different decay classes (III and IV for decaying logs, 3 and 4 for stumps) were selected for analysis. Research plots were established around each deadwood 10 area (20 m × 50 m). The same forest management practices were used in each stand.
Deadwood measurements
Dead lying trees and stumps were analysed in the test areas. Whole logs, stems and fragments with a length greater than or equal to 1.00 m and a thin-end diameter of at least a 10.00 cm were considered. To ensure that dead lying trees were in contact with the soil surface since the beginning, windthrown trees were selected from the inventoried deadwood.
For stumps, the selection criterion was a maximum height of 1.30 m and a basal diameter that exceeded 10.00 cm. The number, size, form and decomposition classes of trees in the stand were investigated. The decay classes of the stumps were classified according to a six-stage system, whereas dead lying trees were evaluated based on five decay classes according to the classification of dead trees presented in Maser et al. (1979) (Table 1). An additional element used in the assessment of the physiological state of the dead trees was the extent to which they were covered by mosses and lichens based on a five-point scale: 0 (no coverage), 1 (coverage less than or equal to 25%), 2 (coverage between 26 and 50%), 3 (coverage between 51 and 75%) and 4 (coverage more than 75%). The dead tree inventory also included the determination of their basic biometric parameters. In the case of stumps, the basal diameter (DB) and height (H) were measured. The length (L) and half-length diameter (D1/2L), as the main attributes (Rondeux and Sanchez 2009), were defined for lying dead trees. The diameter measurements were conducted using a circumeter, with an accuracy of 0.1 cm. The tree height was measured using a Suunto altimeter, with an accuracy of 0.1 m.
The contribution of the varying degrees of coarse woody debris decomposition was determined by summing the length of the dead lying trees. In the case of dead lying trees that varied widely in the advancement of decomposition of the material in different sections, the highest accepted degree of decomposition was taken into account. The thickness of the stumps was used to assess the degree of stump decomposition. The volume (V) of coarse woody debris and stumps was calculated using Huber’s equation (Bruchwald 1999) according to the formula:
where V volume in m3, L length (dead lying trees), H height (stumps) and d diameter.
Soil investigations
Due to the inability to determine the tree species to which decaying logs and stumps with very advanced decomposition belonged (decay classes 5 and 6 for stumps, decay class V for logs) and the failure to yield soil micronutrients by deadwood in the first two decay classes (Stokland et al. 2004, Rondeux and Sanchez 2009), only the third and fourth decay classes of dead trees were taken into account in the analysis. Moreover, the results were representative of each study site, as stumps in decay classes 3 and 4 and dead lying trees in decay classes III and IV were the most numerous (Fig. 2).
Soil samples surrounding the stumps and decaying logs were collected from the A horizon (0–10 cm) after removing the organic horizon (O). The soil samples were collected at three different distances from the decaying logs (0–10, 50–100). Two soil samples associated with each stump were collected at 2 different distances (0–10 and 50 cm). In all cases, samples for research were collected from 4 sub-stands of the humus-mineral horizon (A), and they were thoroughly mixed to yield a composite soil sample. The soil samples were collected in September 2015. For the determination of enzymatic activity, one part of the fresh samples under natural moisture conditions was sieved through a sieve (Ø 2 mm) and stored at 4 °C before analysis. For the analysis of the physico-chemical properties, the samples were first air-dried at room temperature and then sieved.
The soil particle-size distribution was estimated using laser diffraction (Analysette 22, Fritsch, Idar-Oberstein, Germany), and pH was determined in distilled water and 1 M KCl using the potentiometric method. The total nitrogen (N) and organic C (Ct) contents were measured using a LECO CNS True Mac Analyzer (Leco, St. Joseph, MI, USA), and the C/N ratio was calculated. The bulk density (BD) was determined with the use of Kopecky rings with a volume of 250 cm3, based on the dryer method (Ostrowska et al. 1991). The chemical properties of each sample were determined based on two replications. The DH (EC 1.1.1.1) and BG (EC 3.2.1.21) activities were determined based on three replications. DH activity was calculated by the reduction of 2,3,5 triphenyltetrazolium chloride (TTC) to triphenyl formazan (TPF) using Lenhard’s method according to the Casida procedure (Alef and Nannipieri 1995). The BG activity was determined following the method proposed by Eivazi and Tabatabai (1988) using p-nitrophenyl-β-D-glucopyranoside (PNG) as a substrate.
The obtained results were used to evaluate the C stored in the soil around the deadwood (various species in various decay classes). The C stored in the soil horizon (0–10 cm) was determined from the thickness and the bulk density of the horizon:
where CS carbon stored in soil (kg m−2), Ct soil carbon content (g kg−1), BD soil bulk density (g cm−3), T thickness of the soil horizon (10 cm), S surface (1 m2).
The background value for soil without deadwood was established to reference the obtained results. For this purpose, additional soil samples were collected from locations without deadwood. In all cases, the samples were collected from 10 sub-stands.
Statistical analysis
Principal component analysis (PCA) was used to evaluate the relationships between soil properties and deadwood characteristics. The variables used in the PCA included the chemical properties, enzyme activity in the soil, decay classes and the distance to the deadwood. The differences between the mean values of soil properties with respect to deadwood form and tree species were evaluated using the nonparametric U Mann–Whitney test. The impact of distance to the deadwood on the C accumulation and biological activity in the soil was tested using the Kruskal–Wallis test. The statistical significance of the results was verified at a significance level of α = 0.05. All statistical analyses were performed using Statistica 10 software (2010).
Results
Characterization of deadwood
At four study sites, 55 stumps (average 3.50 m3 per hectare) and 42 decaying logs (average 15.09 m3 per hectare) were recorded. The mean volume of Scots pine decaying logs reached 10.89 m3 ha−1 (ca. 1% of the volume of living trees). For Silver fir logs, the mean volume was evaluated as 17.63 m3 ha−1 (ca. 3% of the volume of living trees). The average diameter of the decaying logs and stumps did not exceed 15.00 and 35.00 cm, respectively (Table 2). The decay classes 3 and 4 of the fir and pine stumps and the decay classes III and IV of the fir and pine decaying logs were the most numerous (Fig. 2). A difference in the physiological conditions related to the coverage by mosses and lichens was observed between the fir and pine deadwood. Approximately 30% of the fir stumps and decaying logs were characterized by moss and lichen coverage at a level that exceeded 50%, whereas the share of logs with the same moss and lichen coverage did not reach the 10% level (Table 3).
Deadwood decay classes and soil properties
The C and N contents differed depending on the species of deadwood, the type of deadwood (tree stumps, decaying logs) and the deadwood decay classes (Tables 4, 5). The values of the mentioned soil characteristics were higher for stumps in decay class 4 and decaying logs in decay class IV in comparison to less decomposed deadwood (decay class III for decaying logs and decay class 3 for stumps); it is important to note that the differences were particularly pronounced in the soil in the immediate vicinity of the deadwood (Tables 4, 5).
The dependence of soil C storage on the decay classes was proven. Regardless of tree species, deadwood in the fourth decay class resulted in significantly higher accumulation of C than deadwood in the third decay class. In the soil around the fir deadwood, the C accumulation in the fourth decay class was 25% higher than in the soil around the fir deadwood in the third decay class. The increase in the C accumulation associated with the decay classes was lower in the soil around the pine deadwood (7%) (Table 7).
A projection of the variables on the factor plane clearly demonstrated correlations between the soil properties and the decay classes of the deadwood. In the case of logs, two main factors had a significant total impact (58.9%) on the variance of the variables. Factor 1 explained 41.02% of the variance of the examined properties, and factor 2 explained 17.9% of the variance (Fig. 3). Factor 1 was defined as “C storage”. The other factor was defined as the “biological activity of the soil”. DH activity was strongly correlated with the deadwood decay classes. BG activity was positively correlated with the N and C contents, and it was negatively correlated with pH and the clay content (Fig. 3a). In the case of stumps, the two factors had a significant total impact (71.8%) on the variance of the variables. Factor 1 explained 53.1% of the variance of the examined properties, while factor 2 explained 18.6% of the variance (Fig. 3b). BG activity was strongly correlated with the deadwood decay classes, as well as with the C and N contents. DH activity was negatively correlated with the C/N ratio.
Distance from deadwood and soil properties
In the case of logs, the highest C content (79.0 g kg−1) was measured in the soil in the immediate vicinity of fir logs in the fourth decay class, whereas the lowest C content (31.4 g kg−1) was recorded in soil located 100 cm from pine logs in the third decay class (Table 4). The highest DH activity was recorded in the soil in the immediate vicinity of pine logs in the fourth decay class (33.27 μmol TPF kg−1 h−1), and the lowest activity was 10.65 μmol TPF kg−1 h−1, which was recorded in soil located 100 cm from fir logs in the third decay class (Table 4). High BG activity was observed in the case of soil collected 10 cm from decaying fir logs (Table 4). The C and N contents and the enzyme activity decreased with distance from the logs. At a distance of 50 cm from the logs, the C content was 20% lower compared with the soil in the immediate vicinity of the logs. The C content in soil located 100 cm from the logs was 30% lower compared with the soil in the immediate vicinity of the logs. The DH activity in soil collected 100 cm from a log was 40% lower compared with soil located 0–10 cm from a log. Statistically significant differences were observed for the C content, C/N ratio and DH activity in relation to the distance from a log. Clearer differences were noted in the case of soil collected from around decaying logs in the fourth decay class (Table 4). The enzymatic activity around stumps was distributed, in contrast to the decaying logs. The greater the distance from a stump, the higher the BG and DH activities. The DH activity increased by an average of 50% at a 50 cm distance from a stump, regardless of species and the decay class. The C content around the stumps was distributed similarly to that around the stems. The C content decreased with the distance from a stump depending on the decay class. In the case of soil around the fir and pine stumps in the 4th decay class, the C content decreased more than 50% compared with the soil collected in the immediate vicinity of the stumps. The C content decreased with distance from a stump (Table 5). PCA confirmed negative correlations between C stored in the soil and the distance from the deadwood (Fig. 3a, b).
Deadwood species and soil properties
The N and C contents reached slightly higher values in the fir than in the pine logs, whereas the opposite was observed for stumps (Tables 4, 5). Interestingly, statistically significant differences in the C content of dead lying trees and stumps were reported (Table 6).
No significant differences in the C content of the fir and pine stumps were noted, while tree species had a statistically significant impact on the DH activity in the soil around the stumps. Regardless of the decay classes of the decaying logs, significantly higher BG activity (an average of 15% in decay class III and 25% in decay class IV) was noted for fir logs compared with pine logs (Table 4). Soil collected from around the pine stumps was characterized by higher activities of DH and BG compared with the fir stumps (Tables 5, 6).
The C stored in the soil surrounding decomposed deadwood fir was approximately 25% higher (kg C m−2) than that around the pine deadwood. In decay class III, the C stored in the immediate vicinity of the fir and pine logs amounted to 4.92 and 4.54 kg C m−2, respectively. In decay class IV, the C stored in the immediate vicinity of the decaying fir logs was 7.06 kg C m−2 compared with 5.38 kg C m−2 for pine logs. Smaller differences in the C stored in the soil around fir and pine stumps were recorded (Table 7).
Discussion
The results obtained indicated that the soil C storage and biological activity of the soil were influenced differently by deadwood depending on its form, species, and decay class and by the distance from the deadwood. A strong stimulating effect of deadwood on the biological activity of the soil was reported. According to Lajtha et al. (2005), deadwood stimulates the availability of labile C. Our results confirmed that the amount of available C depends on the decay class of the deadwood. In our study, strong correlations between BG activity and the C content and between DH activity and the deadwood decay classes were noted (Fig. 3). The higher the decay class, the more C substrates contained in the soil, which stimulate the decomposition of soil organic matter. Veres et al. (2013) and Wolińska et al. (2015) proved that soil enzyme activities were ‘sensors’ of soil organic matter decomposition. The decomposition of soil organic matter depends on the environmental conditions such as temperature, humidity, pH and Eh, the presence of electron acceptors (e.g., O2, SO 2−,4 NO3 −, humic substances) and the accessibility of microorganisms and their enzymes (McLatchey and Reddy 1998; Wu and Blodau 2013). In this study, differences in the enzymatic activity between the soil around the fir and pine decaying logs were observed. Higher BG activity was measured in soil around the fir logs, while at the same time, high C storage occurred (Table 4). The difference in the enzymatic activity of the soil around the fir and pine logs could be associated with the characteristics of the test wood species, which were ultimately reflected in the amount and quality of the organic matter. Higher DH activity was measured in the soil around the pine deadwood, where higher quality organic matter was present (lower C/N ratio compared with soil around fir deadwood) (Tables 4, 5). The C/N ratio is often used to describe litter quality (Zhang et al. 2008; Handsson et al. 2011). According to Fontaine et al. (2003), both the amount and quality of soil organic matter is important because organic matter affects the supply of energy for microbial growth and enzyme production. In addition, the accumulation of organic matter and the biological activity in the soil can be affected by mosses and lichens. According to Sedia and Ehrenfeld (2006) and Delgado-Baquerizo et al. (2015), lichens and the bryophytic community have a strong impact on the deadwood decomposition rate and the soil enzyme activity. When the extent of decaying logs covered by lichens and mosses increased, a more advanced process of wood decay was observed (Harmon 1989; Kushnevskaya et al. 2007). In our results, a fivefold higher coverage of fir logs (>50% covered by mosses and lichens) relative to pine logs was observed. The increased coverage of fir logs by mosses and lichens can be explained by the substrate moisture conditions. High log humidity is conducive to colonization by mosses and lichens, and this increases the species richness (Rixen and Mulder 2005; Benavides et al. 2006). Moreover, log coverage by bryophytes was significantly correlated with decay class (Wierzgon and Fojcik 2014). This result appears to confirm those presented by Paletto and Tosi (2010), who demonstrated a higher moisture content for fir compared with pine dead lying trees and an increase in the value of parameters with the advancement of deadwood decay.
In this paper, we attempted to determine the impact of the distance from the deadwood (in the form of decaying logs and stumps) on soil properties. The highest C storage was recorded in the immediate vicinity of the decaying logs and stumps. Lower C accumulation occurred further away from the deadwood. At a 100-cm distance from the deadwood, the influence of the decaying logs was not evident (Table 4). The deadwood mass has been reported to be the main source of C (Caz 1993). The release of nutrients from deadwood can occur in several different ways. The flow of nutrients occurs through the mycelia of wood-decomposing fungi (Zimmerman et al. 1995), as well as through the mycelia of ectomycorrhizal fungi (Lepšová 2001). The products of gradual decomposition accrue in the zone nearest the tree trunk. Further away from the deadwood, little mixing occurs between decomposed organic substances and the surface of the soil. Peeled bark, chipped fragments of wood and the movement of the decomposition products inside the rhizomorphs of fungi support these observations. The mechanical shredding of deadwood in various decay classes by animals searching for insects was also a cause of the distribution of organic matter away from the deposition location. The C/N ratio further away from the deadwood (50 and 100 cm) was lower compared with the 0–10 cm distance from the deadwood. Poorly decomposed organic matter occurred closest to the deadwood, and organic substances were supplied from the direct decay of logs and stumps, as shown by the higher C/N ratios. The accumulation of organic matter and the biological activity of the soil decreased with distance from the logs. The opposite was noted in the case of stumps (Table 5). There was a clear decrease in pH and enzymatic activity around the stumps. Previous studies on the effect of individual trees on soil properties (Skeffington 1983; Boettcher and Kalisz 1990) tended to show a soil pH decrease close to the tree trunk. The investigations conducted by Gruba (2009) showed that the organic matter in soil around trees was crucial for the spatial distribution of soil properties. The activity of mycorrhizal fungi was also important. A significant source of easily degradable organic C is rhizodeposits, which include root cap and border cell loss, death and lysis of root cells, gaseous losses, and the passive and active releases of solutes (root exudates) (Hinsinger et al. 2009). In our study, the density of fine roots in the soil located 10 cm from the stump was presumably lower than in the soil located 50 cm from the stump, which may be the cause of increased activity of enzymes in the soil located further from the stumps. Regardless of the species and decay class, the enzyme activity was more than 50% higher in soil located 50 cm from the stump relative to soil located 10 cm from the stump.
In this study, there was a statistically significant difference in the C content in the soil around the logs of the fir and pine trees (Table 6). This may have been caused by the physical characteristics of the wood. According to Krzysik (1974), fir wood has a lower density (0.45 g cm3) compared with pine (0.55 g cm3). At the same time, fir wood is less saturated with resin compounds. The resinous compounds pose mechanical obstacles that hinder the spread of fungi in wood, and they have a toxic effect on fungi and inhibit their development. These features result in fir wood decomposing more rapidly compared with pine wood. There was a slight increase in the C content in the area surrounding the decaying logs in decay class IV in relation to dead lying trees in decay class III (Table 7). It is highly probable that the difference resulted from the changes in deadwood density, which were associated with the advancement of log decay. As the decomposition of the wood increased, the wood density decreased. This phenomenon has been reported to be most noticeable between the fourth and fifth decay classes (Sandström et al. 2007; Paletto and Tosi 2010). Lombardi et al. (2013) observed differences in the cellulose and lignin levels in most decayed wood samples. It may be hypothesized that the presence of cerambycid frass and fungi in the xylem increased the C concentration, as shown in previous studies (Torres 1994). During the assessment of the degree of decomposition of deadwood in the field, we found that decay class III was characterized by larger hard fragments with a circular shape and fragmented bark. The logs classified in decay class IV presented small pieces, an oval shape and no bark, which were associated with the change in the density of the wood. During the evaluation of the wood decay classes in the field, we observed that in the same decay class, the fir and pine wood differed with respect to hardness. Pine wood was harder (higher density) compared to fir wood. There were also significant differences in the C content in the soil around the stumps and decaying logs. There was higher C storage in the soil around the stumps (Table 7). Stumps are associated with an extensive root system that fills the space around the trunks. According to Janssens et al. (2002), roots are the key component of the underground part of forest ecosystems and the primary source of soil organic matter. Forest litter and roots deliver organic matter that contains various components, such as soluble sugars, organic acids, celluloses and lignin (Baldrian and Šnajdr 2011).
The results obtained confirmed an important role of deadwood in forest ecosystems through enhancement of C accumulation in the soil environment with a simultaneous increase in the biological activity of the soil. Logs and stumps in advanced decay stages most strongly influenced the soil properties. Knowledge of the mechanisms and factors affecting the dynamics of organic C accumulation in forest soils can be useful in reducing climate change impacts on forest ecosystem health. Deadwood in forests should be protected because it can serve as a cheap and sustainable method for protecting and increasing the C stocks in soil. A better understanding of deadwood decomposition processes will complement information on the global C cycle.
Conclusions
The results obtained demonstrated that soil C accumulation and the enzyme activity in the soil were influenced by deadwood form, tree species and decay class. A higher accumulation of C was measured in the soil around the stumps. The stumps were associated with an extensive root system, which filled the space around the trunks. There were significant differences in the C content in the soil around the logs of fir and pine trees. Biological activity was stimulated in the presence of strongly decayed deadwood. The C concentration increased slightly from decay classes 3–4 for stumps and from decay classes III to IV for decaying logs. The highest accumulation of C was observed in the immediate vicinity of decaying logs and stumps. A lower C accumulation in the soil was observed further away from the deadwood.
References
Adamowicz K, Jaszczak R, Kuźmiński R, Łabędzki A, Łakomy P, Mazur A, Starosta-Grala M, Szramka H, Turski M, Zientarski J (2015) An attempt at valuation of wood from dead trees in Polish forests. Acta Sci Pol Silv Colendar Rat Ind Lignar 14:5–13
Alef K, Nannipieri P (1995) Enzyme activities. In: Nannipieri P, Alef K (eds) Methods in applied soil microbiology and biochemistry. Academic Press, London, pp 311–375
Bače RA, Svoboda M, Pouska V, Janda P, Červenka J (2012) Natural regeneration in Central-European subalpine spruce forests: which logs are suitable for seedling recruitment? For Ecol Manag 266:254–262
Baldrian P, Šnajdr J (2011) Lignocellulose-degrading enzymes in soil. In: Shukla G, Varma A (eds) Soil enzymology. Springer, Berlin, pp 167–186
Bantle A, Borken W, Ellerbrock RH, Shulze ED, Weisser WW, Matzner E (2014) Quantity and quality of dissolved organic carbon released from coarse woody debris of different tree species in the early phase of decomposition. For Ecol Manag 329:287–294
Benavides JC, Duque MAJ, Duivenvoorden JF, Cleef AM (2006) Species richness and distribution of understorey bryophytes in different forest types in Colombian Amazonia. J Bryol 28:182–189
Boettcher SE, Kalisz PJ (1990) Single tree influence on soil properties in the mountains of eastern Kentucky. Ecology 71:1365–1372
Bruchwald A (1999) Dendrometria. SGGW, Warszawa
Caz CL (1993) Woody debris in the forests of British Columbia: a review of the literature and current research. B.C. Ministry of Forests Land Management Report No. 78
Cocciufa C, Gerth W, Luiselli L, Redolfi de Zan L, Cerretti P, Carpaneto GM (2014) Survey of saproxylic beetle assemblages at different forest plots in central Italy. B Insectol 67:295–306
Currie WS, Yanai RD, Piatek KB, Prescott CE, Goodale CL (2003) Processes affecting carbon storage in the forest floor and in downed woody debris. In: Heath LS, Birdsey RA, Lal R, Kimble JM (eds) The potential of U.S. forest soils to sequester carbon and mitigate the greenhouse effect. CRC Press, Boca Raton
Davies ZG, Tyler C, Stewart GB, Pullin AS (2008) Are current management recommendations for conserving saproxylic invertebrates effective? Biodivers Conserv 17:209–234
Debeljak M (2006) Coarse woody debris in virgin and managed forest. Ecol Indic 6:733–742
Delgado-Baquerizo M, Gallardo A, Covelo F, Prado-Comesaña A, Ochoa V, Maestre FT (2015) Differences in thallus chemistry are related to species-specific effects of biocrust-forming lichens on soil nutrients and microbial communities. Funct Ecol 29:1087–1098
Eivazi F, Tabatabai MA (1988) Glucosidases and galactosidases in soils. Soil Biol Biochem 20:601–606
Finér L, Mannerkoski H, Piirainen S, Starr M (2003) Carbon and nitrogen pools in an old-growth Norway spruce mixed forest in eastern Finland and changes associated with clear-cutting. For Ecol Manag 174:51–63
Fontaine S, Marotti A, Abbadie L (2003) The priming effect of organic matter: a question of microbial competition. Soil Biol Biochem 35:837–843
Gruba P (2009) The influence of trees on spatial variability of pH in top horizons of forest soil. Sylwan 153:332–337
Handsson K, Olsson BA, Olsson M, Johansson U, Kleja DB (2011) Differences in soil properties in adiacent stands of Scots pine, Norway spruce and Siver birch in SW Sweden. For Ecol Manag 262:522–530
Harmon ME (1989) Effects of Bark Fragmentation on Plant Succession on Conifer Logs in the Picea-Tsuga Forests of Olympic National Park, Washington. Am Midl Nat 121:112–124
Harmon ME, Hua C (1991) Coarse woody debris dynamics in two old-growth ecosystems. -comparing a deciduous forest in China and a conifer forest in Oregon. Bioscience 41:604–610
Hinsinger P, Bengough A, Vetterlein D, Young I (2009) Rhizosphere: biophysics, biogeochemistry and ecological relevance. Plant Soil 321:117–152
Janssens JA, Sampson DA, Curiel-Yuste J, Carrara A, Cenlemans R (2002) The carbon cost of fine root turnover in a Scots pine forest. For Ecol Manag 168:231–240
Jaworski A, Pach M (2014) A comparison of lower montane natural forest (Abies, Fagus, Picea) in Oszast Reserve and spruce monocultures in the Żywiecki Beskid and Śląski Beskid. For Res Pap 75:13–23
Kandeler E (1996) Nitrate. In: Schinner F, Öhlinger R, Kandeler E, Margesin R (eds) Methods in soil biology. Springer, Berlin, pp 408–410
Kappes H, Catalano C, Topp W (2007) Coarse woody debris ameliorates chemical and biotic soil parameters of acidified broad-leaved forests. Appl Soil Ecol 36:190–198
Kotroczó Z, Veres Z, Fekete J, Krakomperger Z, Tóth JA, Lajtha K, Tóthmérisz B (2014) Soil enzyme activity in response to long-term organic matter manipulation. Soil Biol Biochem 70:237–243
Krzysik F (1974) Nauka o drewnie. PWN. Warszawa. pp. 1–653
Kupferschmidt AD, Brang P, Schönenberger W, Bugmann H (2003) Decay of Picea abies snags stands on steep mountain slopes. For Chron 79:1–6
Kushnevskaya H, Mirin D, Shorohova E (2007) Patterns of epixylic vegetation on spruce logs in late-successional boreal forests. For Ecol Manag 250:25–33
Lajtha K, Crow SE, Yano Y, Kaushal SS, Sulzman EW, Sollins P, Spears JDH (2005) Detrial controls on soil solution N and dissolved organic matter in soils: a field experiment. Biogeochemistry 76:261–281
Lepšová A (2001) Ectomycorrhizal system of naturally established Norway spruce [Picea abies (L.) Karst] seedlings from different microhabitats—forest floor and coarse woody debris. Silva Gabreta 7:223–234
Lombardi F, Cherubini P, Tognetti R, Cocozza C, Lasserre B, Marchetti M (2013) Investigating biochemical processes to assess deadwood decay of beech and silver fir in Mediterranean mountain forests. Ann For Sci 70:101–111
Maser C, Anderson RG, Cromak K, Williams JT, Martin RE (1979) Dead and down woody material. In: Thomas JW (ed) Wildlife habitats in managed forests: the blue mountains of Oregon and Washington. USDA forest service agricultural handbook, vol 553. USDA Forest Service, Pacific Northwest Forest and Range Experiment Station, Portland, pp 78–95
McLatchey GP, Reddy KR (1998) Regulation of Organic Matter Decomposition and Nutrient Release in A Wetland soil. J Environ Qual 27:1268–1274
Narukawa Y, Iida S, Tanouchi H (2003) State of fallen logs and the occurrence of conifer seedlings and saplings in boreal and subalpine old-growth forests in Japan. Ecol Res 18:267–277
Olajuyigbe SO, Tobina B, Gardinera P, Nieuwenhuisa M (2011) Stocks and decay dynamics of above- and belowground coarse woody debris in managed Sitka spruce forests in Ireland. For Ecol Manag 262:1109–1118
Ostrowska A, Gawliński S, Szczubiałka Z (1991) Methods of analysis and assessment of soil and plant properties. Environmental Protection Institute, Warszawa, p 334
Paletto A, Tosi V (2010) Deadwood density variation with decay class in seven tree species of the Italian Alps. Scand J For Res 25:164–173
Persiani AM, Audisio P, Lunghini D, Maggi O, Granito VM, Biscaccianti AB, Chiavetta U, Marchetti M (2010) Linking taxonomical and functional biodiversity of saproxylic fungi and beetles in broad-leaved forests in southern Italy with varying management histories. Plant Biosyst 144:250–261
Pichler V, Homolák M, Skierucha W, Pichlerová M, Ramírez D, Gregor J, Jaloviar P (2011) Variability of moisture in coarse woody debris from several ecologically important tree species of the Temperate Zone of Europe. Ecohydrology 5:424–434
Rinta-Kanto JM, Sinkko H, Rajala T, Abu Al-Soud W, Sørensen SJ, Tamminen MV, Timonen S (2016) Natural decay process sffects the abundance and community tructure of Bactreia and Archea in Picea abies logs. Microb Ecol 1:231
Rixen C, Mulder CPH (2005) Improved water retention links high species richness with increased productivity in arctic tundra moss communities. Oecologia 2005:287–299
Rondeux J, Sanchez C (2009) Review of indicators and field methods for monitoring biodiversity within national forest inventories Core variable: deadwood. Environ Monit Assess 164:617–630
Sandström F, Petersson H, Kruys N, Ståhl G (2007) Biomass conversion factors (density and carbon concentration) by decay classes for dead wood of Pinus sylvestris, Picea abies and Betula spp. in boreal forests of Sweden. For Ecol Manag 243:19–27
Sedia EG, Ehrenfeld JG (2006) Differential effects of lichens and mosses on soil enzyme activity and litter decomposition. Biol Fertil Soils 43:177–189
Sinsabaugh RL, Antabus RK, Linkins A (1991) An enzymic approach to the analysis of microbial activity during plant litter deciomposition. Agric Ecosyst Environ 34:43–54
Skeffington RA (1983) Soil properties under three species of tree in southern England in relation to acid deposition in throughfall. In: Ulrich B, Pankrath J (eds) Effect of accumulation of air pollutants in forest ecosystems. Reidel publishing Company, Dordrecht, pp 219–231
StatSoft, Inc. (2010 STATISTICA (version 10.0). Computer software
Stokland JN, Tomter SM, Söderberg U (2004) Development of dead wood indicators for biodiversity monitoring: experiences from Scandinavia. EFI Proceedings pp. 207–228
Takahashi M, Sakai Y, Ootomo R, Shiozaki M (2000) Establishment of tree seedlings and water-soluble nutrients in coarse woody debris in an old-growth Picea-Abies forest in Hokkaido, northern Japan. Can J For Res 30:1148–1155
Torres JA (1994) Wood decomposition of Cyrilla racemiflora in tropical montane forest. Biotopica 26:124–140
Veres Z, Kotroczó Z, Magyaros K, Tóth JA, Tóthmérész B (2013) Dehydrogenase activity in a litter manipulation experiment in temperate forest soil. Acta Silv Lignaria Hung 9:25–33
Wang Q, Xiao F, Wang S, Xu G (2012) Response of selected soil biological properties to stump presence and age in a managed subtropical forest ecosystem. Appl Soil Ecol 57:59–64
Wierzgoń M, Fojcik B (2014) Martwe drewno jako ostoja różnorodności mszaków w lesie gospodarczym. Studia i Materiały CEPL w Rogowie 41:212–222
Wolińska A, Stępniewska Z (2012) Dehydrogenase activity in the Soil Environment. In: Canuto RA (ed) Dehydrogenases. InTech, Rijeka
Wolińska A, Stępniewska Z, Pytlak A (2015) The effect of environmental factors on total soil DNA content and dehydrogenase activity. Arch Biol Sci 67:493–501
WRB (World Reference Base for Soil Resource). (2006) FAO, ISRIC and ISSS
Wu Y, Blodau C (2013) PEATBOG: a biogeochemical model for analyzing coupled carbon and nitrogen dynamics in northern peatlands. Geosci Model Dev 6:1173–1207
Yin X (1999) The decay of forest woody debris: numerical modeling and implications based on some 300 data cases from North America. Oecologia 121:81–98
Zhang D, Hui D, Luo Y, Zhou G (2008) Rates of litter decomposition in terrestrial ecosystems: global patterns and controlling factors. J Plant Ecol 1:85–93
Zimmerman JK, Pulliam WM, Lodge DJ, Quinones-Orfila V, Fetcher N, Guzman-Grajales S, Parrotta JA, Asbury CE, Walker LR, Waide RB (1995) Nitrogen immobilization by decomposing woody debris and the recovery of tropical wet forest from hurricane damage. Oikos 72:314–322
Acknowledgements
The authors would like to thank the management staff of the Magurski National Park for enabling this research. The investigation was financed by the Ministry of Science and Higher Education of the Republic of Poland.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Błońska, E., Kacprzyk, M. & Spólnik, A. Effect of deadwood of different tree species in various stages of decomposition on biochemical soil properties and carbon storage. Ecol Res 32, 193–203 (2017). https://doi.org/10.1007/s11284-016-1430-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11284-016-1430-3






