Abstract
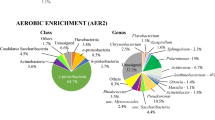
Diesel fuel storage tanks are not hostile environments for microorganisms and tend to form sludges in the water deposited at the bottom of the tanks. The lack of nutrient, carbon and energy limitations within these habitats boost the abundance and the metabolic activity of microorganisms providing microbial hotspots with high growing rates of diesel degradation (0.10 ± 0.021 d−1). Five different Phyla (Thermotogae, Spirochaetes, Firmicutes, Bacteroidetes Proteobacteria) were identified within the aqueous/sludge phase from in situ diesel storage tanks, by cultured independent molecular surveys using the 16S rDNA gene fragment. The identified dominant strains were Geotoga aestuarianus, Flavobacterium ceti, Spirochaeta thermophila, Propionispira arboris, Sporobacterium olearium and Dysgonomonas genera. The altitude where the storage tanks are located and the organic carbon concentration within the aqueous/sludge phases affected the bacterial diversity. Therefore, the more diverse the microbial communities are, the more probability of the presence of bacteria with capacity to metabolized diesel and eliminate organic matter. Despite, only phosphate showed an effect on the bacterial distribution within the storage tanks, there was an apparent lack of deterministic process in structuring microbial communities. Consequently, preventative protocols are a priority to avoid the microbial growth within diesel fuel storage tanks. A new focus of this worldwide problem within the oil industry would be to explore deeply the wide range of metabolic and adaptive capacities of these microorganisms. These microbial consortia are potential tools with new specific services to apply in bioremediation among others.
Graphical Abstract







Similar content being viewed by others
Abbreviations
- DGGE:
-
Denaturing gradient gel electrophoresis
- NGS:
-
Next generation sequencing
- MPN:
-
Most probably number technique
- DUB:
-
Diesel uncultured bacteria
- NJ:
-
Neighbor-joining method
- DCA:
-
Detrended correspondence analysis
- CCA:
-
Canonical correspondence analysis
References
Abena MTB, Chen G, Chen Z, Zheng X, Li S, Li T, Zhong W (2020) Microbial diversity changes and enrichment of potential petroleum hydrocarbon degraders in crude oil, diesel, and gasoline contaminated soil. Biotech 10:42. https://doi.org/10.1007/s13205-019-2027-7
Ahmadi N, Khosravi-Darani K, Mortazavian AM (2017) An overview of biotechnological production of propionic acid: From upstream to downstream processes. Electron J Biotechnol 28:67–75. https://doi.org/10.1007/s13205-019-2027-7
Auffret MD, Yergeau E, Labbé D, Fayolle-Guichard F, Greer CW (2015) Importance of Rhodococcus strains in a bacterial consortium degrading a mixture of hydrocarbons, gasoline, and diesel oil additives revealed by metatranscriptomic analysis. App Microbiol Biot 99:2419–2430. https://doi.org/10.1007/s00253-014-6159-8
Azam F (1998) Microbial control of oceanic carbon flux: the plot thickens. Science 280:694–696. https://doi.org/10.1126/science.280.5364.694
Bell TH, Yergeau E, Maynard C, Juck D, Whyte LG, Greer CW (2013) Predictable bacterial composition and hydrocarbon degradation in Arctic soils following diesel and nutrient disturbance. ISME 7:1200–1210. https://doi.org/10.1038/ismej.2013.1
Bautista LF, Vargas C, González N, Molina MC, Simarro R, Salmerón A, Murillo Y (2016) Assessment of biocides and ultrasound treatment to avoid bacterial growth in diesel fuel. Fuel Proc Technol 152:56–63. https://doi.org/10.1016/j.fuproc.2016.06.002
Bento FM, Gaylarde CC (2001) Biodeterioration of stored diesel: studies in Brazil. Int Biodeterior Biodegr 47:107–112. https://doi.org/10.1016/S0964-8305(00)00112-8
Bento FM, Beech IB, Gaylarde CC, Englert GE, Muller IL (2005) Degradation and corrosive activities of fungi in a diesel–mild steel–aqueous system. World J Microbiol Biotechnol 21:135–142. https://doi.org/10.1007/s11274-004-3042-2
Bryant J, Lamanna C, Morlon H, Kerkhoff AJ, Enquist BJ, Green JL (2008) Microbes on mountainsides: contrasting elevational patterns of bacterial and plant diversity. Proc Natl Acad Sci USA 105:11505–11511. https://doi.org/10.1073/pnas.0801920105
Bücker F, Barbosa CS, Quadros PD, Bueno MK, Fiori HC, Frazzon APG, Ferrao MF, Camargo FAO, Bento FM (2014) Fuel biodegradation and molecular characterization of microbial biofilms in stored diesel/biodiesel blend B10 and the effect of biocide. Int Biodet Biodegr 95:346–355. https://doi.org/10.1016/j.ibiod.2014.05.030
Clarke KR, Warwick RM (2001) Change in marine communities. An approach to statistical analysis and interpretation, 2nd edn. PRIMER-E, Ltd., Plymouth Marine Laboratory, Plymouth
Chaudhary DK, Kim J (2017) Flavobacterium olei sp. nov. a novel psychrotolerant bacterium isolated from oil-contaminated soil. Int J Syst Evol Microbiol 67:2211–2218. https://doi.org/10.1099/ijsem.0.001925
Chen YA, Liu PWG, Whang LM, Wu YJ, Cheng SS (2020) Effect of soil organic matter on petroleum hydrocarbon degradation in diesel/fuel oil-contaminated soil. J Biosci Bioeng 129:603–612. https://doi.org/10.1016/j.jbiosc.2019.12.001
Da Fonseca MMB, Minnicelli CF, Silva-Portela RDC, de Farias MF, dos Santos PRS, Fernandes GJT, Agnez-Lima LF (2019) Unlocking and functional profiling of the bacterial communities in diesel tanks upon additive treatment. Fuel 236:1311–1320. https://doi.org/10.1016/j.fuel.2018.09.107
De Azambuja AO, Bücker F, de Quadros PD, Zhalnina K, Dias R, Vacaro BB, Correa C, Ferrão MF, de Oliveira Camargo FA, Triplett E, Bento FM (2017) Microbial community composition in Brazilian stored diesel fuel of varying sulfur content, using high-throughput sequencing. Fuel 189:340–349. https://doi.org/10.1016/j.fuel.2016.10.108
Delgado-Baquerizo M, Reich PB, Khachane AN, Campbell CD, Thomas N, Freitag TE, Singh BK, Al-Soud WA, Sørensen S, Bardgett RD, Sing BK (2017) It is elemental: soil nutrient stoichiometry drives bacterial diversity. Environ Microbiol 19:1176–1188. https://doi.org/10.1111/1462-2920.13642
Dixit H, Lowry M, Mohsin U, Moond M, Kumar S, Chokriwal A, Gupta S (2018) Screening and identification of diesel oil degrading bacterial isolates from petroleum contaminated soil of Barmer. J Pharm Chem Biol Sci 6:34–40
Dorofeev AG, Nikolaev YA, Mardanov AV, Pimenov NV (2020) Role of phosphate-accumulating bacteria in biological phosphorus removal from wastewater. Appl Biochem Microbiol 56:1–14. https://doi.org/10.1134/S0003683820010056
Dutilh BE, Huynen MA, Bruno WJ, Snel B (2004) The consistent phylogenetic signal in genome trees revealed by reducing the impact of noise. J Mol Evol 58:527–539. https://doi.org/10.1007/s00239-003-2575-6
European Standard (2013) EN 14214:2013. Liquid petroleum products—fatty acid methyl esters (FAME) for use in diesel engines and heating applications—requirements and test methods. European Committee for Standardization, Brussels, Belgium
Fang D, Zhao G, Xu X, Zhang Q, Shen Q, Fang Z, Huang L, Ji F (2018) Microbial community structures and functions of wastewater treatment systems in plateau and cold regions. Bioresour Technol 249:684–693. https://doi.org/10.1016/j.biortech.2017.10.063
Fierer N, Bradford MA, Jackson RB (2007) Toward an ecological classification of soil bacteria. Ecology 88:1354–1364. https://doi.org/10.1890/05-1839
Fierer N, Jackson RB (2006) The diversity and biogeography of soil bacterial communities. Proc Natl Acad Sci USA 103:626–631. https://doi.org/10.1073/pnas.0507535103
Fierer N, McCain DM, Meir P, Zimmermann M, Rapp JM, Silman MR, Knigh R (2011) Microbes do not follow the elevational diversity patterns of plants and animals. Ecology 92:797–804. https://doi.org/10.1890/10-1170.1
González N, Simarro R, Molina MC, Bautista LF, Delgado L, Villa JA (2011) Effects of surfactants on PAH biodegradation by a bacterial consortium and on the dynamics of the bacterial community during the process. Biores Technol 102:9438–9446. https://doi.org/10.1016/j.biortech.2011.07.066
González-Benítez N, Molina MC, Arrayás M (2020) Empirical evidence and mathematical modelling of carbamazepine degradative kinetics by a wood-rotting microbial consortium. Waste Biomass Valor. https://doi.org/10.1007/s12649-020-01041-1
Groysman A (2017) Corrosion problems and solutions in oil refining and petrochemical industry. Springer International Publishing, Gewerbestrasse (ISBN 978-3-319-45254-8)
Gupta RS (2004) The phylogeny and signature sequences characteristics of Fibrobacteres, Chlorobi and Bacteroidetes. Crit Rev Microbiol 30:123–143. https://doi.org/10.1080/10408410490435133
Hulecki JC, Foght JM, Fedorak PM (2010) Storage of oil field-produced waters alters their chemical and microbiological characteristics. Microbiol Biotechnol 37:371–481. https://doi.org/10.1007/s10295-010-0693-x
Johnson DW (2018) The effects of storage on turbine engine fuels. In: Volkov K (ed) Flight physics—models, techniques and technologies. IntechOpen, London
Jost L (2006) Entropy and diversity. Oikos 113:363–375. https://doi.org/10.1111/j.2006.0030-1299.14714.x
Jukes TH, Cantor R (1969) Evolution of protein molecules. In: Munro H (ed) Mammalian protein metabolism. Academic Press, New York
Jung J, Philippot L, Park W (2016) Metagenomic and functional analyses of the consequences of reduction of bacterial diversity on soil functions and bioremediation in diesel-contaminated microcosms. Sci Rep 6:23012. https://doi.org/10.1038/srep23012
Kacagan M, Inan K, Belduz AO, Canakci S (2013) Flavobacterium anatoliense sp. nov. isolated from fresh water, and emended description of Flavobacterium ceti. Int J Syst Evol Microbiol 63:2075–2081. https://doi.org/10.1099/ijs.0.040394-0
Katoh K, Toh H (2008a) Recent developments in the MAFFT multiple sequence alignment program. Brief Bioinform 9:286–298. https://doi.org/10.1093/bib/bbn013
Katoh K, Toh H (2008b) Improved accuracy of multiple ncRNA alignment by incorporating structural information into a MAFFT-based framework. BMF Bioinform 9:212. https://doi.org/10.1186/1471-2105-9-212
Kawaharasaki M, Tanaka H, Kanagawa T, Nakamura K (1999) In situ identification of polyphosphate-accumulating bacteria in activated sludge by dual staining with rRNA-targeted oligonucleotide probes and 40,6-diamidino-2-phenylindol (DAPI) at a polyphosphate-probing concentration. Water Res 33:257–265. https://doi.org/10.1016/S0043-1354(98)00183-3
Klammer S, Knapp B, Insam H, Dell’Abate MT, Ros M (2008) Bacterial community patterns and thermal analyses of composts of various origins. Waste Manag Res 26:173–187. https://doi.org/10.1177/0734242X07084113
Kraková L, Šoltys K, Budiš J, Grivalský T, Ďuriš F, Pangallo D, Szemes T (2016) Investigation of bacterial and archaeal communities: novel protocols using modern sequencing by Illumina MiSeq and traditional DGGE-cloning. Extremophiles 20:795–808. https://doi.org/10.1007/s00792-016-0855-5
Kuzyakov Y, Blagodatskaya E (2015) Microbial hotspots and hot moments in soil: concept and review. Soil Biol Biochem 83:184–199. https://doi.org/10.1016/j.soilbio.2015.01.025
Lee JS, Ray RI, Little BJ (2010) An assessment of alternative diesel fuels: microbiological contamination and corrosion under storage conditions. Biofouling 26:623–635. https://doi.org/10.1080/08927014.2010.504984
Legendre P, Legendre L (2003) Numerical ecology, 2nd edn. Elsevier Science, Amsterdam
Liang B, Wang LY, Mbadinga SM, Liu JF, Yang SZ, Gu JD, Mu BZ (2015) Anaerolineaceae and Methanosaeta turned to be the dominant microorganisms in alkanes-dependent methanogenic culture after long-term of incubation. AMB Expr 5:37. https://doi.org/10.1186/s13568-015-0117-4
Liu Z, Ge Y, Xu J, Gao C, Ma C, Xu P (2016) Efficient production of propionic acid through high density culture with recycling cells of Propionibacterium acidipropionici. Bioresour Technol 216:856–861. https://doi.org/10.1016/j.biortech.2016.06.023
Lomans BP, Van der Drift C, Pol A, Op den Camp HJM (2002) Microbial cycling of volatile organic sulfur compounds. Cell Mol Life Sci 59:575–588. https://doi.org/10.1007/s00018-002-8450-6
Madden TL, Tatusov RL, Zhang J (1996) Applications of network BLAST server. Meth Enzymol 266:131–141. https://doi.org/10.1016/S0076-6879(96)66011-X
Magot M, Fardeau ML, Arnauld O, Lanau C, Ollivier B, Thomas P, Patel BKC (1997) Spirochaeta smaragdinae sp. nov. a new mesophilic strictly anaerobic spirochete from an oil field. FEMS Microbiol Lett 155:185–191. https://doi.org/10.1111/j.1574-6968.1997.tb13876.x
Martin-Sanchez PM, Gorbushina AA, Toepel J (2018) Quantification of microbial load in diesel storage tanks using culture- and qPCR-based approaches. Int Biodet Biodegr 126:216–223. https://doi.org/10.1016/j.ibiod.2016.04.009
Marques CR (2018) Extremophilic microfactories: applications in metal and radionuclide bioremediation. Frontiers Microbiol. https://doi.org/10.3389/fmicb.2018.01191
Mechichi T, Labat M, Garcia JL, Thomas P, Patel BK (1999) Sporobacterium olearium gen. nov. sp. nov. a new methanethiol-producing bacterium that degrades aromatic compounds, isolated from an olive mill wastewater treatment digester. Int J Syst Evol Microbiol 49:1741–1748. https://doi.org/10.1099/00207713-49-4-1741
Molina MC, González N, Bautista LF, Sanz R, Simarro R, Sánchez I, Sanz JL (2009) Isolation and genetic identification of PAH degrading bacteria from a microbial consortium. Biodegradation 20:789–800. https://doi.org/10.1007/s10532-009-9267-x
Naeem S, Li S (1997) Biodiversity enhances ecosystem reliability. Nature 390:507–509. https://doi.org/10.1038/37348
Núñez A, Moreno DA (2020) The differential vertical distribution of the airborne biological particles reveals an atmospheric reservoir of microbial pathogens and aeroallergens. Microb Ecol. https://doi.org/10.1007/s00248-020-01505-w
Ollivier B, Cayol JL (2005) The fermentative, iron-reducing, and nitrate-reducing microorganisms. In: Ollivier B, Magot M (eds) Petroleum microbiology. ASM press, Washington, pp 71–88
Pedros-Alió C (2006) Microbial diversity: can it be determined? Trends Microbiol 14:257–263. https://doi.org/10.1016/j.tim.2006.04.007
Rowland HAL, Boothman C, Pancost R, Gault AG, Polya DA, Lloyd JR (2009) The role of indigenous microorganisms in the biodegradation of naturally occurring petroleum, the reduction of iron, and the mobilization of arsenite from west Bengal aquifer sediments. J Environ Qual 38:1598–1607. https://doi.org/10.2134/jeq2008.0223
Ryazanova LP, Suzina NE, Kulakovskaya TV, Kulaev IS (2009) Phosphate accumulation of Acetobacter xylinum. Arch Microbiol 191:467–471. https://doi.org/10.1007/s00203-009-0470-2
Semenova EM, Grouzdev DS, Tourova TP, Nazina TN (2019) Physiology and genomic characteristics of Geotoga petraea, a bacterium isolated from a low-temperature petroleum reservoir (Russia). Microbiology 88:662–670. https://doi.org/10.1134/S0026261719060171
Shahi A, Aydin S, Ince B, Ince O (2016) Reconstruction of bacterial community structure and variation for enhanced petroleum hydrocarbons degradation through biostimulation of oil contaminated soil. Chem Eng J 306:60–66. https://doi.org/10.1016/j.cej.2016.07.016
Sharma S, Compant S, Ballhausen MB, Ruppel S, Franke P (2020) The interaction between Rhizoglomus irregulare and hyphae attached phosphate solubilizing bacteria increases plant biomass of Solanum lycopersicum. Microbiol Res. https://doi.org/10.1016/j.micres.2020.126556
Striebich RC, Smart CE, Gunasekera TS, Mueller SS, Strobel EM, McNichols BW, Ruiz ON (2014) Characterization of the F-76 diesel and Jet-A aviation fuel hydrocarbon degradation profiles of Pseudomonas aeruginosa and Marinobacter hydrocarbonoclasticus. Int Biodet Biodeg 93:33–43. https://doi.org/10.1016/j.ibiod.2014.04.024
Sun J, Daniel R, Wagner-Döbler I, Zeng AP (2003) Is autoinducer-2 a universal signal for interspecies communication: a comparative genomic and phylogenetic analysis of the synthesis and signal transduction pathways? BMC Evol Biol 4:36. https://doi.org/10.1186/1471-2148-4-36
Swofford DL (2002) PAUP and other methods: phylogenetic analysis using parsimony. Version 4.0b10. Sinauer Associates, Sunderland
Tayabali A, Coleman G, Crosthwait J, Nguyen KC, Zhang Y, Shwed P (2017) Composition and pathogenic potential of a microbial bioremediation product used for crude oil degradation. PLoS ONE 12:e0171911. https://doi.org/10.1371/journal.pone.0171911
van Ommen KF, Geesey GG (1999) Localization, identification of populations of phosphatase-active bacterial cells associated with activated sludge flocs. Microbial Ecol 38:201–214. https://doi.org/10.1007/s002489900170
van Veen HW, Abee T, Korstee GJJ, Konings WN, Zehnder AJB (1994) Translocation of metal phosphate via the phosphate inorganic transport system of Escherichia coli. Biochemistry 33:1766–1770. https://doi.org/10.1021/bi00173a020
Varjani SJ (2017) Microbial degradation of petroleum hydrocarbons. Biores Technol 223:277–286. https://doi.org/10.1016/j.biortech.2016.10.037
Viejo RM (2009) Resilience in intertidal rocky shore assemblages across the stress gradient created by emersion times. Mar Ecol Prog Ser 390:55–65. https://doi.org/10.3354/meps08171
Wang LY, Sun XB, Liu JF, Gu JD, Mu BZ (2014) Comparison of bacterial community in aqueous and oil phases of the water-flooded petroleum reservoir using pyrosequencing and clone library approaches. Appl Microbiol Biotechnol 98:4209–4221. https://doi.org/10.1007/s00253-013-5472-y
White J, Gilbertm J, Hill G, Hill E, Huse SM, Weightman AJ, Mahenthiralingam E (2011) Culture-independent analysis of bacterial fuel contamination provides insight into the level of concordance with the standard industry practice of aerobic cultivation. Appl Environ Microbiol 77:4527–4538. https://doi.org/10.1128/AEM.02317-10
Wilhelm L, Besemer K, Fragner L, Peter H, Weckwerth W, Battin TJ (2015) Altitudinal patterns of diversity and functional traits of metabolically active microorganisms in stream biofilms. ISME J 9:2454–2464. https://doi.org/10.1038/ismej.2015.56
Wolf M, Müller T, Dandekar T, Pollack JD (2004) Phylogeny of Firmicutes with special reference to Mycoplasma mollicutes. as inferred from phosphoglycerate kinase amino acid sequence data. Int J Syst Evol Microbiol 54:871–875. https://doi.org/10.1099/ijs.0.02868-0
Wu L, Wen C, Qin Y, Yin H, Tu Q, van Nostrand JD, Yuan T, Yuan M, Deng Y, Zhou J (2015) Phasing amplicon sequencing on Illumina Miseq for robust environmental microbial community analysis. BMC Microbiol 15:125. https://doi.org/10.1186/s12866-015-0450-4
Yang SS, Pang JW, Guo WQ, Yang XY, Wu ZY, Ren NQ, Zhao ZQ (2017) Biological phosphorus removal in an extended ASM2 model: Roles of extracellular polymeric substances and kinetic modeling. Bioresour Technol 232:412–416. https://doi.org/10.1016/j.biortech.2017.01.048
Acknowledgements
Authors are deeply indebted to reviewers for their constructive criticism and helpful about the manuscript. Thanks, are given to Clara Santos for helping during the experimental phase of the work.
Funding
This work was financed by Repsol, S.A.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
González-Benítez, N., Bautista, L.F., Simarro, R. et al. Bacterial diversity in aqueous/sludge phases within diesel fuel storage tanks. World J Microbiol Biotechnol 36, 180 (2020). https://doi.org/10.1007/s11274-020-02956-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-020-02956-6