Abstract
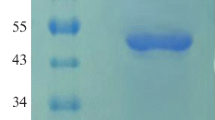
A novel neutral phytase gene (phyC) from Bacillus licheniformis was cloned and expressed in Pichia pastoris under the control of AOX1 promoter. The gene is 1,146 bp in size and encodes a polypeptide of 381 amino acids. The recombinant PhyCm (rePhyCm), driven by the Saccharomyces cerevisiae α-mating factor, was secreted into culture medium. After 0.5% methanol induction for 96 h, the activity of rePhyCm in culture supernatant reached 0.23 U/ml. The optimum temperature and pH of purified rePhyCm were 60°C and 7.5, respectively. The rePhyCm was stable in a wide pH range of 5.0–9.0, especially for alkaline pH. The residual activities of rePhyCm retained over 80% after being incubated at pH 5.0–9.0, 37°C for 1 h in the presence of 1 mM CaCl2. Interestingly, supplemental Ca2+ upgraded both the thermostability and pH stability of rePhyCm. Substrate specificity of rePhyCm, effects of metal ions and chemicals on phytase activity were also investigated in current study.




Similar content being viewed by others
References
Baruah K, Sahu NP, Pal AK, Debnath D (2004) Dietary phytase: an ideal approach for a cost-effective and low-polluting aquafeed. NAGA, World Fish Center Q 27:15–19
Boling SD, Douglas MW, Johnson ML, Wang X, Parsons CM, Koelkebeck KW, Zimmerman RA (2000) The effects of dietary available phosphorus levels and phytase on performance of young and older laying hens. Poult Sci 79:224–230
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cao L, Wang WM, Yang CT, Yang Y, Diana J, Yakupitiyage A, Luo Z, Li DP (2007) Application of microbial phytase in fish feed. Enzyme Microb Technol 40:497–507
Choi YM, Suh HJ, Kim JM (2001) Purification and properties of extracellular phytase from Bacillus sp. KHU-10. J Protein Chem 20:287–292
Fu DW, Huang HQ, Luo HY, Wang YR, Yang PL, Meng K, Bai YG, Wu NF, Yao B (2008a) A highly pH-stable phytase from Yersinia kristeensenii: cloning, expression, and characterization. Enzyme Microb Technol 42:499–505
Fu SJ, Sun JY, Qian LC, Li ZY (2008b) Bacillus phytases: present scenario and future perspectives. Appl Biochem Biotechnol 151:1–8
Fu SJ, Sun JY, Qian LC (2008c) Effect of Ca2+ on beta-propeller phytases. Protein Pept Lett 15:39–42
Gulati HK, Chadha BS, Saini HS (2007) Production and characterization of thermostable alkaline phytase from Bacillus laevolacticus isolated from rhizosphere soil. J Ind Microbiol Biot 34:91–98
Guo MJ, Hang HF, Zhu TC, Zhuang YP, Chu J, Zhang SL (2008) Effect of glycosylation on biochemical characterization of recombinant phytase expressed in Pichia pastoris. Enzyme Microb Technol 42:340–345
Ha NC, Oh BC, Shin S, Kim HJ, Oh TK, Kim YO, Choi KY, Oh BH (2000) Crystal structures of a novel, thermostable phytase in partially and fully calcium-loaded states. Nat Struct Biol 7:147–153
Hang HF, Ye XH, Guo MJ, Chu J, Zhuang YP, Zhang M, Zhang SL (2009) A simple fermentation strategy for high-level production of recombinant phytase by Pichia pastoris using glucose as the growth substrate. Enzyme Microb Technol 44:185–188
Harland BF, Oberleas D (1999) Phytic acid complex in feed ingredients. In: Coelho MB, Kornegay ET (eds) Phytase in animal nutrition and waste management. BASF Corp, Mount Olive, pp 69–76
Heinonen JK, Lahti RJ (1981) A new and convenient colorimetric determination of inorganic orthophosphate and its application to the assay of inorganic pyrophosphatase. Anal Biochem 113:313–317
Kerovuo J, Lappalainen I, Reinikainen T (2000) The metal dependence of Bacillus subtilis phytase. Biochem Biophys Res Commun 268:365–369
Kim YO, Kim HK, Bae KS, Yu JH, Oh TK (1998) Purification and properties of a thermostable phytase from Bacillus sp. DS11. Enzyme Microb Technol 22:2–7
Konietzny U, Greiner R (2004) Bacterial phytase: potential application, in vivo function and regulation of its synthesis. Braz J Microbiol 35:11–18
Kumar S, Dudley J, Nei M, Tamura K (2008) MEGA: a biologist-centric software for evolutionary analysis of DNA and protein sequences. Brief Bioinform 9:299–306
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lee J, Choi Y, Lee PC, Kang S, Bok J, Cho J (2007) Recombinant production of Penicillium oxalicum PJ3 phytase in Pichia Pastoris. World J Microbiol Biotechnol 23:443–446
Maenz DD, Classen HL (1998) Phytase activity in the small intestinal brush border membrane of the chicken. Poult Sci 77:557–563
Mukesh P, Suma S, Singaracharya MA, Lakshmipathi V (2004) Isolation of phytate-hydrolysing microbial strains from traditional waste water of rice fermentation and liquid cattle feeds. World J Microbiol Biotechnol 20:531–534
Mullaney EJ, Ullah AH (2003) The term phytase comprises several different classes of enzymes. Biochem Biophys Res Commun 312:179–184
Mullaney EJ, Locovare H, Sethumadhavan K, Boone S, Lei XG, Ullah AHJ (2010) Site-directed mutagenesis of disulfide bridges in Aspergillus niger NRRL 3135 phytase (PhyA), their expression in Pichia pastoris and catalytic characterization. Appl Microbiol Biotechnol 87:1367–1372
Oh BC, Chang BS, Park KH, Ha NC, Kim HK, Oh BH, Oh TK (2001) Calcium-dependent catalytic activity of a novel phytase from Bacillus amyloliquefaciens DS11. Biochemistry 40:9669–9676
Oh BC, Choi WC, Park S, Kim YO, Oh TK (2004) Biochemical properties and substrate specificities of alkaline and histidine acid phytases. Appl Microbiol Biotechnol 63:362–372
Park SC, Choi YW, Oh TK (1999) Comparative enzymatic hydrolysis of phytate in various animal feedstuff with two different phytases. J Vet Med Sci 61:1257–1259
Reddy NR, Sathe SK, Salunkhe DK (1982) Phytates in legumes and cereals. Advances in Food Research, Logan, pp 281–290
Roy T, Mondal S, Ray AM (2009) Phytase-producing bacteria in the digestive tracts of some freshwater fish. Aquac Res 40:344–353
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Sebastian S, Touchburn SP, Chavez ER (1998) Implications of phytic acid and supplemental microbial phytase in poultry nutrition: a review. World Poult Sci J 54:27–47
Shimizu M (1992) Purification and characterization of phytase from Bacillus subtilis (natto) N-77. Biosci Biotechnol Biochem 56:1266–1269
Song GY, Wang XY, Wang M (2005) Influence of disulfide bonds on the conformational changes and activities of refolded phytase. Protein Pept Lett 12:533–535
Soni SK, Khire JM (2007) Production and partial characterization of two types of phytase from Aspergillus niger NCIM 563 under submerged fermentation conditions. World J Microbiol Biotechnol 23:1585–1593
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Tian YS, Peng RH, Xu J, Zhao W, Gao F, Fu XY, Xiong AS, Yao QH (2010) Mutations in two amino acids in phyI1s from Aspergillus niger 113 improve its phytase activity. World J Microbiol Biotechnol 26:903–907
Tye AJ, Siu FKY, Leung TYC, Lim BL (2002) Molecular cloning and the biochemical characterization of two novel phytases from B. subtilis 168 and B. licheniformis. Appl Microbiol Biotechnol 59:190–197
Ullah AHJ, Mullaney EJ (1996) Disulfide bonds are necessary for structure and activity in Aspergillus ficuum phytase. Biochem Biophys Res Commun 227:311–317
Van Etten RL, Davidson R, Stevis PE, MacArthur H, Moore DL (1991) Covalent structure, disulfide bonding, and identification of reactive surface and active site residues of human prostatic acid phosphatase. J Biol Chem 266:2313–2319
Veith B, Herzberg C, Steckel S, Feesche J, Maurer KH, Ehrenreich P, Bäumer S, Henne A, Liesegang H, Merkl R, Ehrenreich A, Gottschalk G (2004) The complete genome sequence of Bacillus licheniformis DSM13, an organism with great industrial potential. J Mol Microbiol Biotechnol 7:204–211
Wang XY, Meng FG, Zhou HM (2004) The role of disulfide bonds in the conformational stability and catalytic activity of phytase. Biochem Cell Biol 82:329–334
Acknowledgments
This work was supported by the National High-Tech Research and Development Plan (2007AA100601), and was also supported by the Science and Technology Department of Zhejiang Province of China (2006C12036) and the Shandong Science and Technology Development Project of People’s Republic of China (2009GG10009001). The authors gratefully acknowledge Miss Shirley for her useful comments on this work and critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Q., Fu, SJ., Sun, JY. et al. Characterization of a thermostable alkaline phytase from Bacillus licheniformis ZJ-6 in Pichia pastoris . World J Microbiol Biotechnol 27, 1247–1253 (2011). https://doi.org/10.1007/s11274-010-0574-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-010-0574-5